Abstract
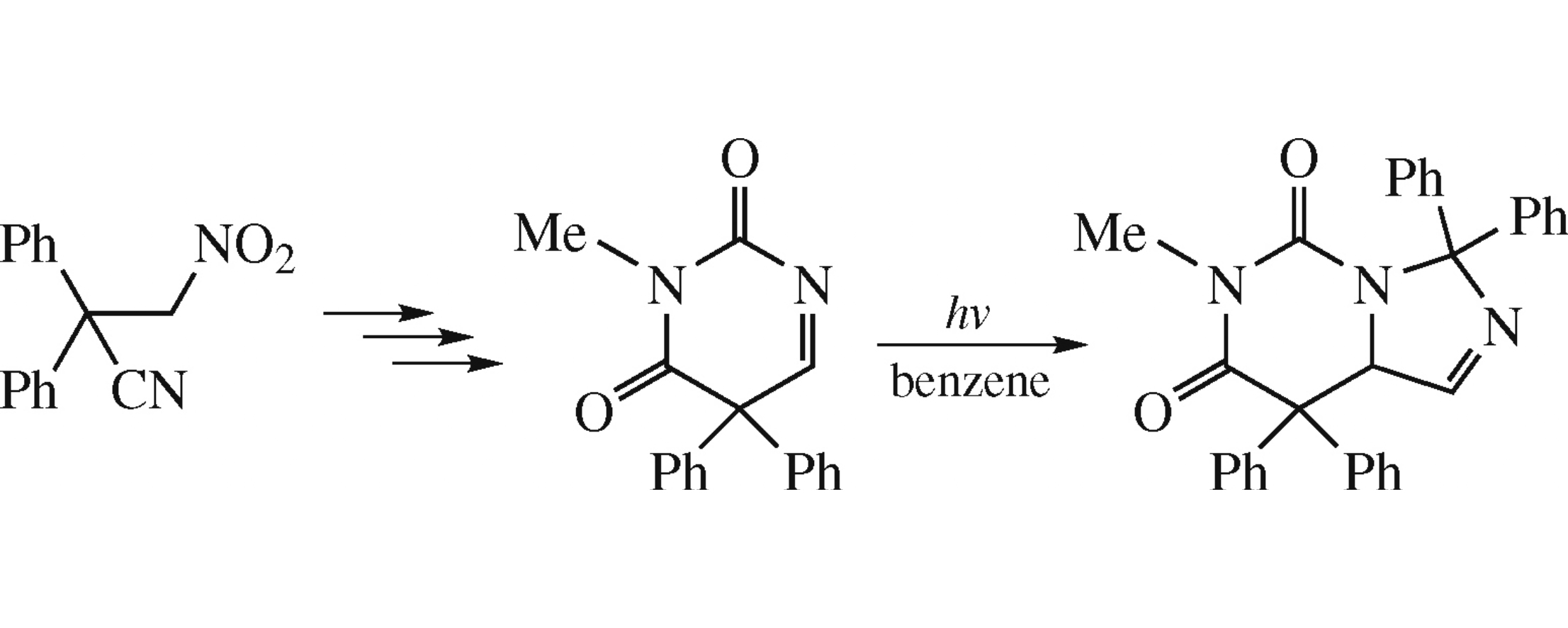
The herein obtained 3-methyl-5,5-diphenylpyrimidine-2,4-(3H,5H)-dione forms upon photolysis an unusual bicyclic product, namely, 6-methyl-3,3,8,8-tetraphenyl-8,8a-dihydroimidazo[1.5-c]pyrimidine-5,7(3H,6H)-dione in 21% yield. The transformation starts with elimination of carbon monoxide and methyl isocyanate from the molecule of substrate via a ‘Type A’ photochemical process giving 3,3-diphenylazirine. Then 1,3-dipole species generated from the latter is trapped by the starting compound via a [3+2] cycloaddition.
References
1.

Zimmerman H.E., Swenton J.S.
Journal of the American Chemical Society,
1967
2.

Schuster D.I., Patel D.J.
Journal of the American Chemical Society,
1968
3.

Arigoni D., Bosshard H., Bruderer H., Büchi G., Jeger O., Krebaum L.J.
Helvetica Chimica Acta,
1957
4.

Barton D.H., de Mayo P., Shafiq M.
Journal of the Chemical Society (Resumed),
1958
5.
10.1016/j.mencom.2018.09.016_sbref0015a
Zimmerman
Abstracts of Papers, 17th National Organic Symposium of the American Chemical Society,
1961
6.

Zimmerman H.E., Schuster D.I.
Journal of the American Chemical Society,
1962
7.

Zimmerman H.E., Wilson J.W.
Journal of the American Chemical Society,
1964
8.

Zimmerman H.E., Rieke R.D., Scheffer J.R.
Journal of the American Chemical Society,
1967
9.

Zimmerman H.E., Mitkin O.D.
Journal of Organic Chemistry,
2007
10.

Zimmerman H.E., Mitkin O.D.
Journal of the American Chemical Society,
2006
11.

Zimmerman H.E., Pushechnikov A.
Organic Letters,
2004
12.

Zimmerman H.E., Shorunov S.
Journal of Organic Chemistry,
2009
13.

Gross Z., Hoz S.
Journal of the American Chemical Society,
1988
14.

Matsuo J., Tanaki Y., Ishibashi H.
Tetrahedron Letters,
2007
15.
10.1016/j.mencom.2018.09.016_sbref0045a
Sheldrick
Acta Crystallogr.,
2015
16.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009