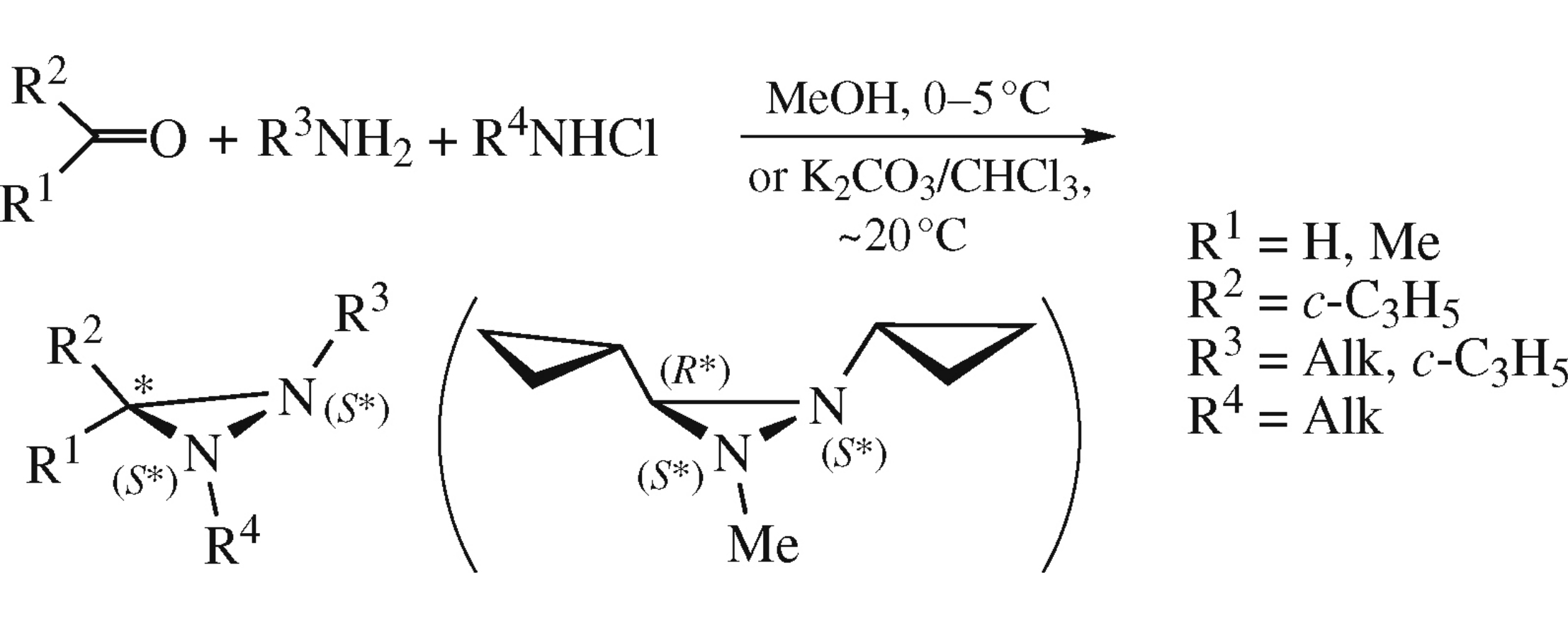
Abstract
A facile, diastereoselective method for the synthesis of diaziridines with C- and N-cyclopropyl substituents has been developed, N-cyclopropyldiaziridines being for the first time synthesized. The method is based on one-pot three-component condensation of cyclopropyl-containing carbonyl compounds, primary aliphatic amines including cyclopropylamine, and N-chloroalkylamines in organic solvents in the presence of bases under mild conditions.
References
1.

Borretto E., Lazzarato L., Spallotta F., Cencioni C., D’Alessandra Y., Gaetano C., Fruttero R., Gasco A.
ACS Medicinal Chemistry Letters,
2013
2.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
3.

Fershtat L.L., Makhova N.N.
ChemMedChem,
2017
4.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
5.
Kulikov A.S., Larin A.A., Fershtat L.L., Anikina L.V., Pukhov S.A., Klochkov S.G., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
6.

Auclair A.L., Martel J.C., Assié M.B., Bardin L., Heusler P., Cussac D., Marien M., Newman-Tancredi A., O'Connor J.A., Depoortère R.
Neuropharmacology,
2013
7.
![Enantiopure Cyclopropane-Bearing Pyridyldiazabicyclo[3.3.0]octanes as Selective α4β2-nAChR Ligands](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Onajole O.K., Eaton J.B., Lukas R.J., Brunner D., Thiede L., Caldarone B.J., Kozikowski A.P.
ACS Medicinal Chemistry Letters,
2014
8.

Talele T.T.
Journal of Medicinal Chemistry,
2016
9.

Lutfy K., Cowan A.
Current Neuropharmacology,
2004
10.

Domagala J.M.
Journal of Antimicrobial Chemotherapy,
1994
11.

YOSHIDA M., EZAKI M., HASHIMOTO M., YAMASHITA M., SHIGEMATSU N., OKUHARA M., KOHSAKA M., HORIKOSHI K.
Journal of Antibiotics,
1990
12.

Ren J., Milton J., Weaver K.L., Short S.A., Stuart D.I., Stammers D.K.
Structure,
2000
13.

Yamamoto Y., Gaynor R.B.
Trends in Biochemical Sciences,
2004
14.

Bart G., Schluger J.H., Borg L., Ho A., Bidlack J.M., Kreek M.J.
Neuropsychopharmacology,
2005
15.

Tomilov Y.V., Menchikov L.G., Novikov R.A., Ivanova O.A., Trushkov I.V.
Russian Chemical Reviews,
2018
16.

Paget C.J., Davis C.S.
Journal of Medicinal Chemistry,
1964
17.

Kostyanovskii R.G., Shustov G.V., Nabiev O.G., Denisenko S.N., Sukhanova S.A., Lavretskaya É.F.
Pharmaceutical Chemistry Journal,
1986
18.

Baichurina A.Z., Semina I.I., Garaev R.S.
Bulletin of Experimental Biology and Medicine,
1996
19.
10.1016/j.mencom.2018.09.015_sbref0015d
Makhova
Patent WO 2013/111117,
2013
20.
10.1016/j.mencom.2018.09.015_sbref0015e
Makhova
Patent WO 2013/111118,
2013
21.
10.1016/j.mencom.2018.09.015_sbref0015f
Makhova
Patent WO 2013.121334,
2013
22.

23.

Prokopov A.A., Kotlova L.I., Berlyand A.S.
Pharmaceutical Chemistry Journal,
2007
24.
![Synthesis and structure of 1-[ω-(3,3-dialkyldiaziridin-1-yl)alkyl]-3, 3-dialkyldiaziridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Petukhova V.Y., Strelenko Y.A., Lyssenko K.A., Makhova N.N.
Russian Chemical Bulletin,
2007
25.

Prokopov A.A., Kotlova L.I., Berlyand A.S.
Pharmaceutical Chemistry Journal,
2006
26.

Prokopov A.A., Kotlova L.I., Berlyand A.S.
Pharmaceutical Chemistry Journal,
2005
27.
Kuznetsov V.V., Shevtsov A.V., Pleshchev M.I., Strelenko Y.A., Makhova N.N.
Mendeleev Communications,
2016
28.
![1,5- Diazabicyclo[3.1.0]hexanes and 1,6-diazabicyclo[4.1.0]heptanes: A new method for the synthesis, quantum-chemical calculations, and X-ray diffraction study](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kuznetsov V.V.
Russian Chemical Bulletin,
2003
29.

Modarelli D.A., Morgan S., Platz M.S.
Journal of the American Chemical Society,
1992
30.

De Meijere A., Erden I., Weber W., Kaufmann D.
Journal of Organic Chemistry,
1988
31.

Rosenberg M.G., Brinker U.H.
Journal of Organic Chemistry,
2001
32.
Makhova N.N., Petukhova V.Y., Kuznetsov V.V.
Arkivoc,
2008
33.
Petukhova V.Y., Pleshchev M.I., Fershtat L.L., Kuznetsov V.V., Kachala V.V., Makhova N.N.
Mendeleev Communications,
2012
34.

Makhova N.N., Shevtsov A.V., Petukhova V.Y.
Russian Chemical Reviews,
2011
35.
Pleshchev M.I., Petukhova V.Y., Kuznetsov V.V., Khakimov D.V., Pivina T.S., Struchkova M.I., Nelyubina Y.V., Makhova N.N.
Mendeleev Communications,
2013
36.
![CAN-mediated new, regioselective one-pot access to bicyclic cationic structures with 2,3-dihydro-1H-pyrazolo[1,2-a]pyrazol-4-ium core](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Pleshchev M.I., Das Gupta N.V., Kuznetsov V.V., Fedyanin I.V., Kachala V.V., Makhova N.N.
Tetrahedron,
2015
37.
![Conformational and Bonding Properties of 3,3-Dimethyl- and 6,6-Dimethyl-1,5-diazabicyclo[3.1.0]hexane: A Case Study Employing the Monte Carlo Method in Gas Electron Diffraction](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Vishnevskiy Y.V., Schwabedissen J., Rykov A.N., Kuznetsov V.V., Makhova N.N.
Journal of Physical Chemistry A,
2015
38.

Kuznetsov V.V., Marochkin I.I., Goloveshkin A.S., Makhova N.N., Shishkov I.F.
Structural Chemistry,
2017
39.

Chagarovskiy A.O., Vasin V.S., Kuznetsov V.V., Ivanova O.A., Rybakov V.B., Shumsky A.N., Makhova N.N., Trushkov I.V.
Angewandte Chemie - International Edition,
2018
40.

Altova E.P., Kuznetsov V.V., Marochkin I.I., Rykov A.N., Makhova N.N., Shishkov I.F.
Structural Chemistry,
2018
41.

Trapp O., Schurig V., Kostyanovsky R.G.
Chemistry - A European Journal,
2004
42.

Jung M., Schurig V.
Journal of the American Chemical Society,
1992
43.
10.1016/j.mencom.2018.09.015_sbref0045c
Lehn
Top. Curr. Chem.,
1970
44.

Mannschreck A., Radeglia R., Gründemann E., Ohme R.
European Journal of Inorganic Chemistry,
1967