Abstract
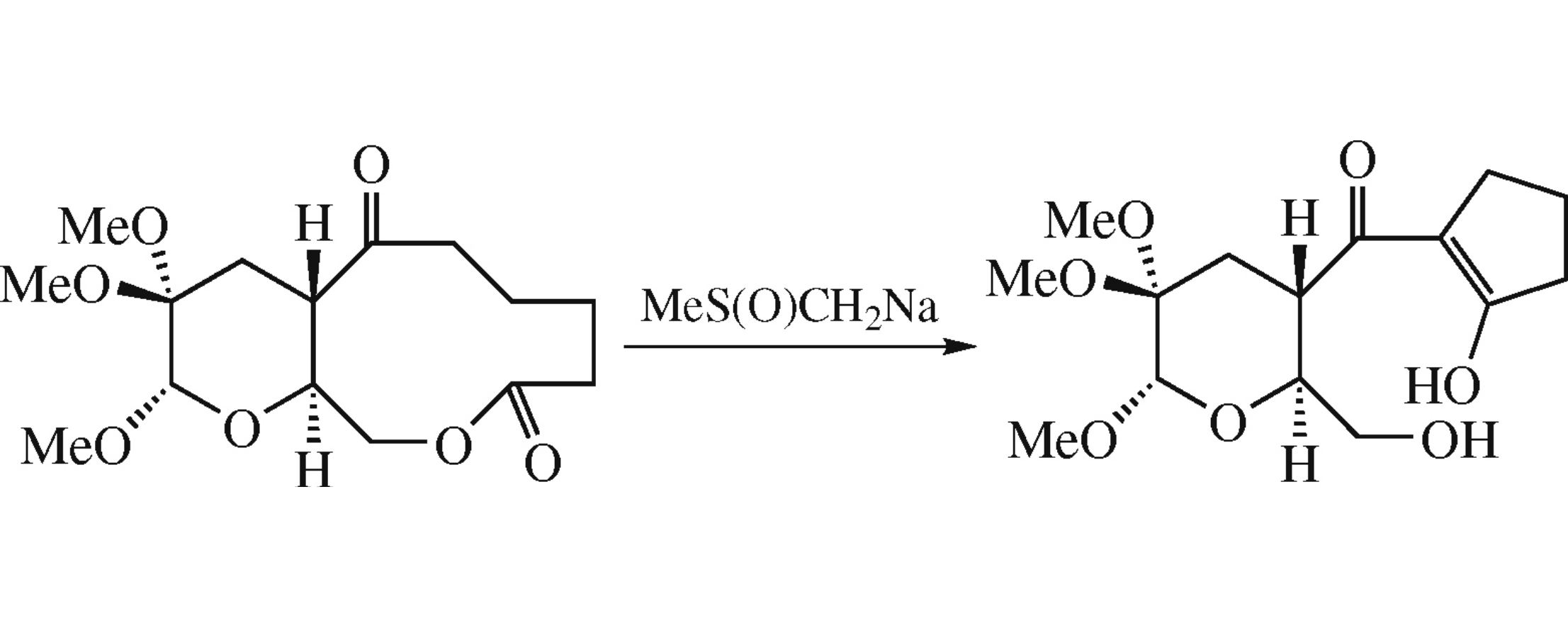
Action of bases on levoglucosenone-derived 6-oxononan-9-olide and 7-oxodecan-10-olide causes the transannular reaction of the aldol type affording stable 2-acylcyclopent-1-en-1-ol and 2-acylcyclohex-1-en-1-ol, respectively. The lower homologue, 5-oxooctan-8-olide derivative, under the similar conditions gives a complex mixture of products, which may be explained by the poor stability of the intermediate 2-acyl-cyclobut-1-en-1-ol.
References
1.

Isobe M., Fukami N., Nishikawa T., Goto T.
Heterocycles,
1987
2.

Miftakhov M.S., Valeev F.A., Gaisina I.N.
Russian Chemical Reviews,
1994
3.
10.1016/j.mencom.2018.09.009_sbref0005c
Levoglucosenone and Levoglucosans: Chemistry and Applications,
1994
4.
Miftakhov M.S., Valeev F.A., Gaisina I.N., Tolstikov G.A.
Mendeleev Communications,
1994
5.

Bamba M., Nishikawa T., Isobe M.
Tetrahedron,
1998
6.

M. Sarotti A., M. Zanardi M., A. Spanevello R.
Current Organic Synthesis,
2012
7.
10.1016/j.mencom.2018.09.009_sbref0010d
Comba
Eur. J. Org. Chem.,
2017
8.
Biktagirov I.M., Faizullina L.K., Salikhov S.M., Galin F.Z., Valeev F.A.
Mendeleev Communications,
2017
9.
Sharipov B.T., Pershin A.A., Valeev F.A.
Mendeleev Communications,
2017
10.
10.1016/j.mencom.2018.09.009_sbref0010g
Valeev
Russ. J. Org. Chem.,
1996
11.

Koseki K., Ebata T., Kawakami H., Matsushita H., Naoi Y., Itoh K.
Heterocycles,
1990
12.

Matsumoto K., Ebata T., Koseki K., Okano K., Kawakami H., Matsushita H.
Bulletin of the Chemical Society of Japan,
1995
13.

Flourat A.L., Peru A.A., Teixeira A.R., Brunissen F., Allais F.
Green Chemistry,
2015
14.

Valeev F.A., Gorobets E.V., Tsypysheva I.P., Singizova G.S., Kalimullina L.K., Safarov M.G., Shitikova O.V., Miftakhov M.S.
Chemistry of Natural Compounds,
2003
15.

Ok T., Jeon A., Lee J., Lim J.H., Hong C.S., Lee H.
Journal of Organic Chemistry,
2007
16.
![Preparation of chiral cyclopropanecarboxylic acids and 3-oxabicyclo[3.1.0]hexane-2-ones from levoglucosenone](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Samet A.V., Lutov D.N., Konyushkin L.D., Strelenko Y.A., Semenov V.V.
Tetrahedron Asymmetry,
2008
17.

Stockton K.P., Merritt C.J., Sumby C.J., Greatrex B.W.
European Journal of Organic Chemistry,
2015
18.

Peru A., Flourat A., Gunawan C., Raverty W., Jevric M., Greatrex B., Allais F.
Molecules,
2016
19.

Ostermeier M., Schobert R.
Journal of Organic Chemistry,
2014
20.

Khalilova Y.A., Tagirov A.R., Doronina O.Y., Spirikhin L.V., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2014
21.

Khalilova Y.A., Spirikhin L.V., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2014
22.

Dräger G., Kirschning A., Thiericke R., Zerlin M.
Natural Product Reports,
1996
23.
![Proximity Effects. I. 6-Aminocyclodecanol and 11-Azabicyclo[4.4.1]-1-undecene from 6-Aminocyclodecanone](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Cope A.C., Cotter R.J., Roller G.G.
Journal of the American Chemical Society,
1955
24.

Karim M.R., Sampson P.
Tetrahedron Letters,
1988
25.

Strenge A., Rademacher P.
European Journal of Organic Chemistry,
1999
26.

Reyes E., Uria U., Carrillo L., Vicario J.L.
Tetrahedron,
2014
27.

Honeychuck R.V., Bonnesen P.V., Farahi J., Hersh W.H.
Journal of Organic Chemistry,
1987
28.
![The Relative Stabilities of cis and trans Isomers. V. The Bicyclo[5.2.0]nonanes. An Extension of the Conformational Rule1,2](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Allinger N.L., Nakazaki M., Zalkow V.
Journal of the American Chemical Society,
1959
29.
![Benz[cd]indolderivate](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
30.
31.

Smith A.B., Levenberg P.A., Jerris P.J., Scarborough R.M., Wovkulich P.M.
Journal of the American Chemical Society,
1981
32.

Luche J.L., Petrier C., Dupuy C.
Tetrahedron Letters,
1984
33.

Biktagirov I.M., Faizullina L.K., Salikhov S.M., Safarov M.G., Valeev F.A.
Russian Journal of Organic Chemistry,
2015
34.
Lüttringhaus A., Prinzbach H.
1959
35.

Leonard N.J., Morrow D.F., Rogers M.T.
Journal of the American Chemical Society,
1957
36.

Silva L.F.
Tetrahedron,
2002
37.

Fadel A., Canet J., Salaün J.
Synlett,
1990
38.

Bloomfield J.J., Owsley D.C., Ainsworth C., Robertson R.E.
Journal of Organic Chemistry,
1975