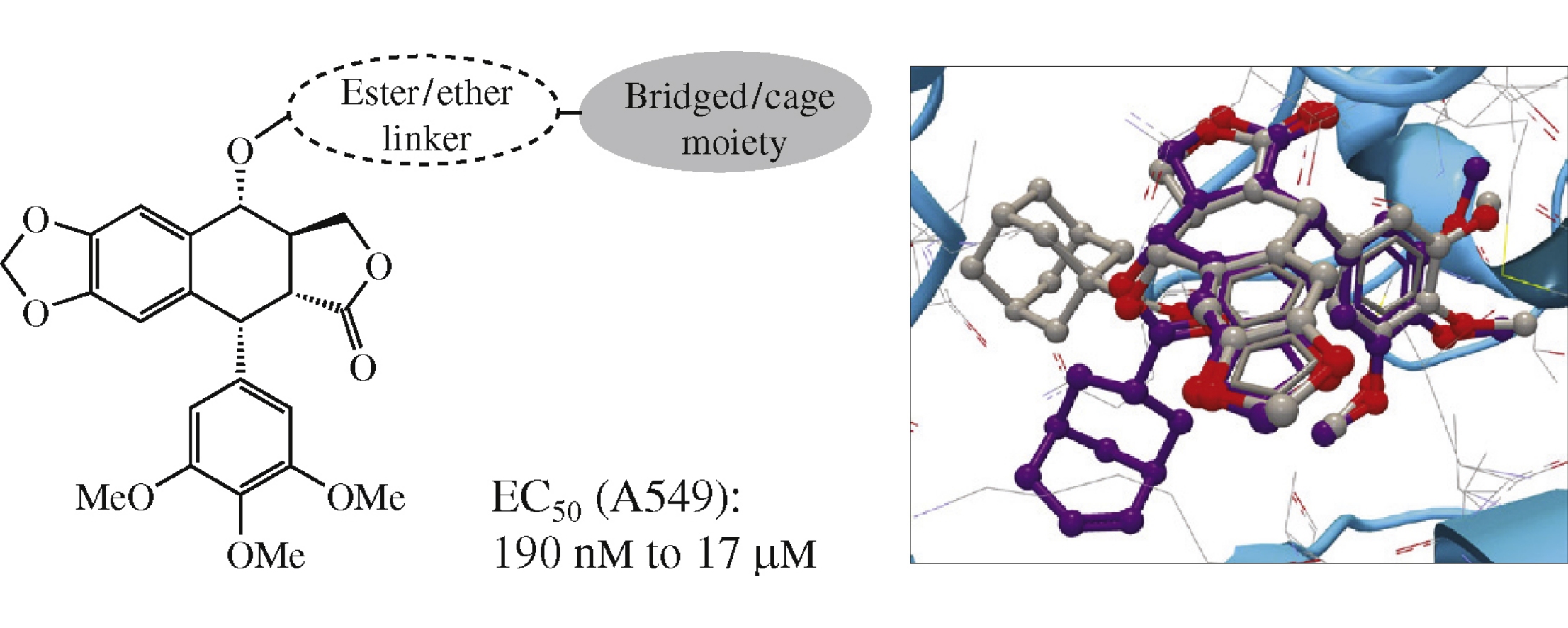
Abstract
New podophyllotoxin C4-derivatives with bridged and cage moieties were synthesized by the Steglich esterification of podophyllotoxin with polycyclic carboxylic acids or by etherication with (adamantan-1-yl)methanol in the presence of BF3·Et2O with the following separation of diastereomers. Most of the target compounds inhibited the growth of human lung carcinoma A549 cells, induced apoptosis, stimulated shortening of microtubules or induced their unusual curling and involution. The activity depends on the slightest differences in the structures of alicyclic moieties and the type of the bond between podophyllotoxin and alicyclic group.
References
1.

Yu X., Che Z., Xu H.
Chemistry - A European Journal,
2017
2.

Liu Y., Tian J., Qian K., Zhao X., Morris-Natschke S.L., Yang L., Nan X., Tian X., Lee K.
Medicinal Research Reviews,
2014
3.

Kamal A., Ali Hussaini S.M., Rahim A., Riyaz S.
Expert Opinion on Therapeutic Patents,
2015
4.

López-Pérez J.L., del Olmo E., de Pascual-Teresa B., Abad A., San Feliciano A.
Bioorganic and Medicinal Chemistry Letters,
2004
5.

Abad A., López-Pérez J.L., del Olmo E., García-Fernández L.F., Francesch A., Trigili C., Barasoain I., Andreu J.M., Díaz J.F., San Feliciano A.
Journal of Medicinal Chemistry,
2012
6.

Sauer W.H., Schwarz M.K.
Journal of Chemical Information and Computer Sciences,
2003
7.
GVK Biosciences Pvt. Ltd Proprietary Databases, https://www.gostardb.com.
8.
B.T. Yajie, X. Li, Patent CN 102875565, 2013.
9.
Zefirova O.N., Nurieva E.V., Lemcke H., Ivanov A.A., Zyk N.V., Weiss D.G., Kuznetsov S.A., Zefirov N.S.
Mendeleev Communications,
2008
10.

Sasaki T., Eguchi S., Toru T.
Journal of Organic Chemistry,
1970
11.
L. Tafesse, N. Tsuno, X. Zhou, Patent ES 2583146, 2016.
12.
![Synthesis and Reactions of 3- and 3,7-Substituted Bicyclo[3.3.1]nonanes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Liu J., Gauger G., Kovacic P.
Journal of Organic Chemistry,
1973
13.

Zilla M.K., Nayak D., Vishwakarma R.A., Sharma P.R., Goswami A., Ali A.
European Journal of Medicinal Chemistry,
2014
14.

Mosmann T.
Journal of Immunological Methods,
1983
15.
Nurieva E.V., Zefirov N.A., Mamaeva A.V., Grishin Y.K., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2017
16.
Vasilenko D.A., Averina E.B., Zefirov N.A., Wobith B., Grishin Y.K., Rybakov V.B., Zefirova O.N., Kuznetsova T.S., Kuznetsov S.A., Zefirov N.S.
Mendeleev Communications,
2017
17.

Beloglazkina A.A., Wobith B., Barskaia E.S., Zefirov N.A., Majouga A.G., Beloglazkina E.K., Zyk N.V., Kuznetsov S.A., Zefirova O.N.
Medicinal Chemistry Research,
2016
18.

Zefirova O.N., Lemcke H., Lantow M., Nurieva E.V., Wobith B., Onishchenko G.E., Hoenen A., Griffiths G., Zefirov N.S., Kuznetsov S.A.
ChemBioChem,
2013
19.

Al-Haddad A., Shonn M.A., Redlich B., Blocker A., Burkhardt J.K., Yu H., Hammer J.A., Weiss D.G., Steffen W., Griffiths G., Kuznetsov S.A.
Molecular Biology of the Cell,
2001
20.

Potten C.
British Journal of Cancer,
1996
21.

Zefirova O.N., Nurieva E.V., Wobith B., Gogol V.V., Zefirov N.A., Ogonkov A.V., Shishov D.V., Zefirov N.S., Kuznetsov S.A.
Molecular Diversity,
2017
22.

Nurieva E.V., Zefirov N.A., Mamaeva A.V., Wobith B., Kuznetsov S.A., Zefirova O.N.
Russian Chemical Bulletin,
2018