Abstract
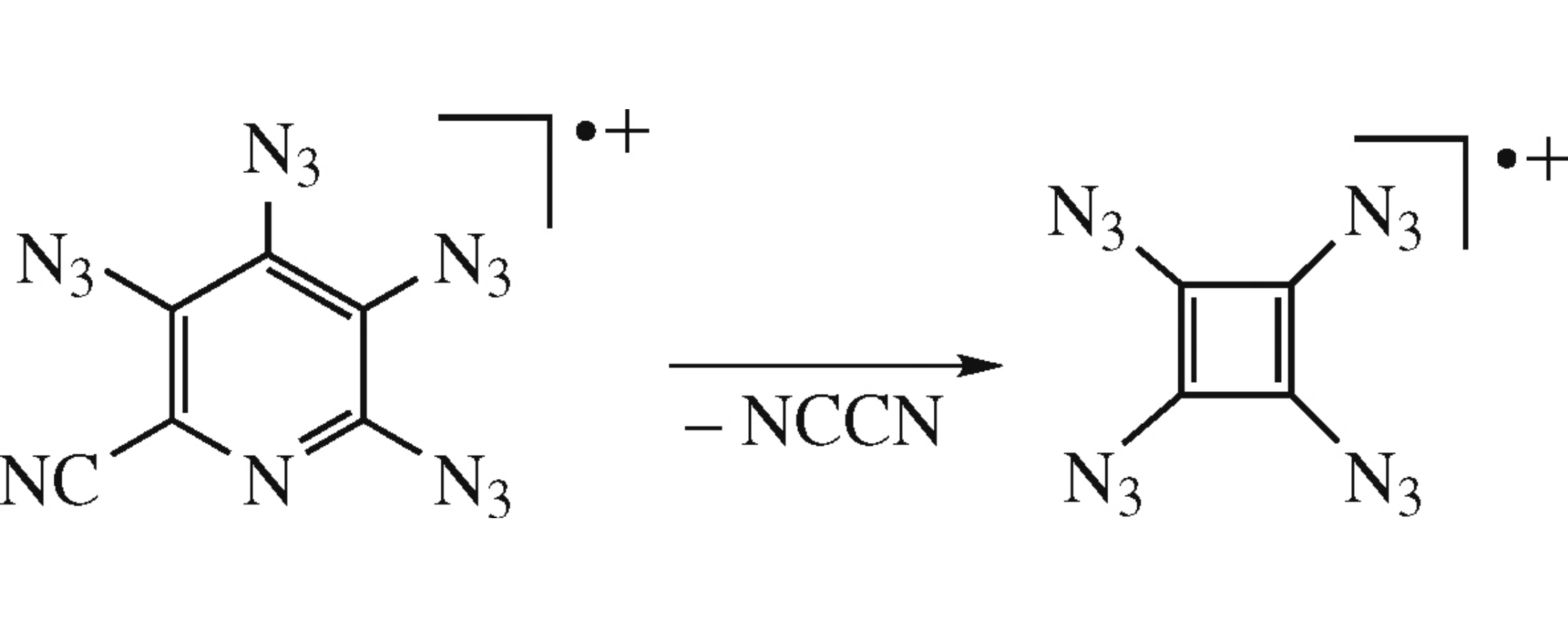
Upon electron impact, tetraazidopyridine-2-carbonitrile releases cyanogen to form tetraazidocyclobutadiene radical cation as the first representative of azido-substituted cyclo-butadiene derivatives.
References
1.
10.1016/j.mencom.2018.09.005_sbref0005a
Williams
Chem. Commun.,
1967
2.

Dickinson R.J., Williams D.H.
Journal of the Chemical Society Perkin Transactions 2,
1972
3.
10.1016/j.mencom.2018.09.005_sbref0005c
1971
4.

Yim M.K., Choe J.C.
Journal of Physical Chemistry A,
2011
5.
10.1016/j.mencom.2018.09.005_bib0010
Molenaar-Langeveld
J. Mass Spectrom.,
1979
6.
Chapyshev S.V., Korchagin D.V., Chernyak A.V., Aldoshin S.M.
Mendeleev Communications,
2017
7.

Chapyshev S.V., Korchagin D.V., Ganin Y.V., Ushakov E.N.
Chemistry of Heterocyclic Compounds,
2017
8.

Mikhailov Y.M., Chapyshev S.V., Nedel’ko V.V.
Russian Chemical Bulletin,
2009
9.

Nedel’ko V.V., Korsunskii B.L., Larikova T.S., Mikhailov Y.M., Chapyshev S.V., Chukanov N.V.
Russian Journal of Physical Chemistry B,
2011
10.

Chapyshev S.V., Chernyak A.V., Ushakov E.N.
Magnetic Resonance in Chemistry,
2016
11.

Nedel’ko V.V., Korsunskii B.L., Larikova T.S., Chapyshev S.V., Chukanov N.V., Yuantsze S.
Russian Journal of Physical Chemistry B,
2016
12.

Chapyshev S.
Molecules,
2015
13.
![Nitrene–Nitrene Rearrangement under Thermal, Photochemical, and Electron‐Impact Conditions: The 2‐Azidopyridines/Tetrazolo[1,5‐ a ]pyridines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Wentrup C., Braybrook C., Liu S., Tzschucke C.C., Dargelos A., Bégué D.
European Journal of Organic Chemistry,
2016
14.
10.1016/j.mencom.2018.09.005_bib0050
Zeller
The Chemistry of Quinonoid Compounds,
1974
15.

Neidlein R., Leidholdt R.
Chemische Berichte,
1986
16.

Esselman B.J., McMahon R.J.
Journal of Physical Chemistry A,
2011
17.
10.1016/j.mencom.2018.09.005_sbref0060b
Rayne
Comput. Theor. Chem.,
2012
18.

Menke J.L., Patterson E.V., McMahon R.J.
Journal of Physical Chemistry A,
2010
19.

Nazari F., Doroodi Z.
International Journal of Quantum Chemistry,
2009
20.

Eckert-Maksić M., Lischka H., Maksić Z.B., Vazdar M.
Journal of Physical Chemistry A,
2009
21.

Menke J.L., McMahon R.J.
Canadian Journal of Chemistry,
2011