Abstract
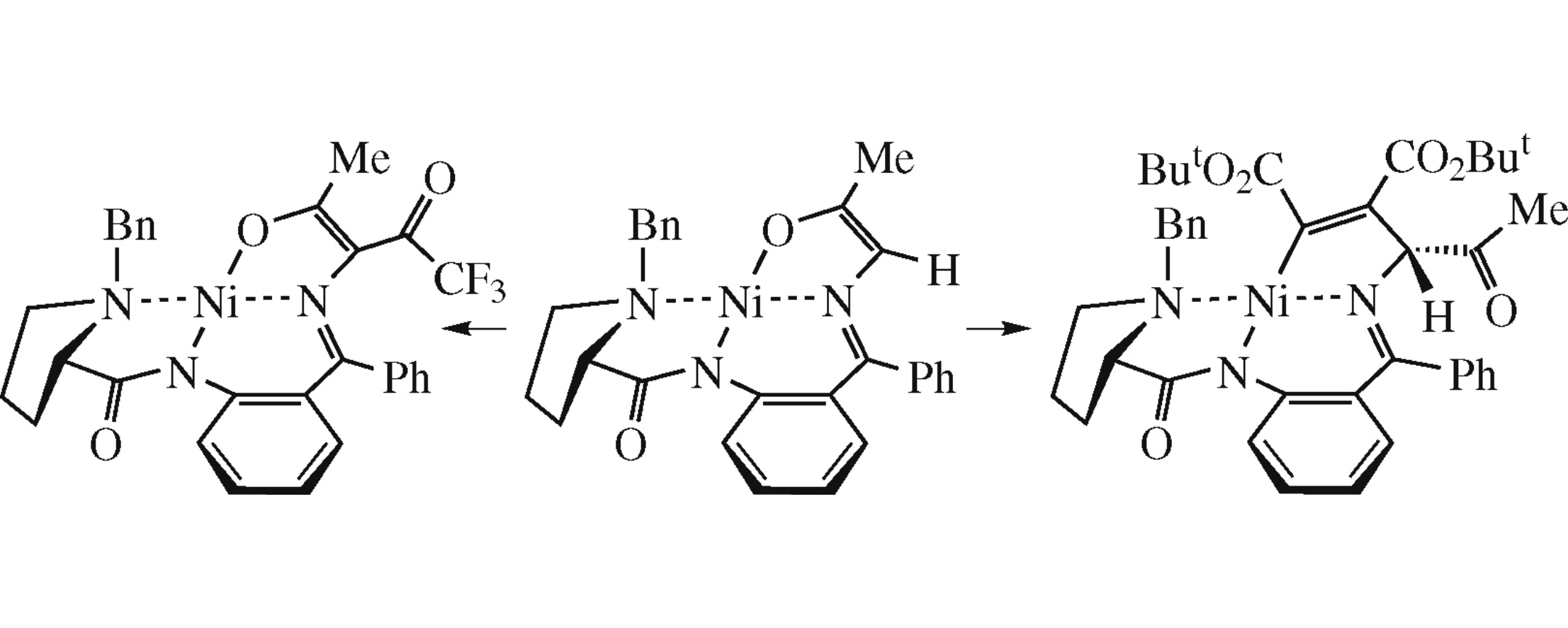
A representative within a new class of chiral enol NiII complexes derived from a Schiff base of aminoacetone and (S)-2-N-(N-benzylprolinoylamino)benzophenone was prepared, and its performance in nucleophilic addition was estimated. The complex was inert towards aldehydes and activated C=C bonds but reacted with carboxylic anhydrides and di-tert-butyl acetylenedicarboxylate. An unusual Michael addition intermediate stabilized by the Ni–C bond was discovered in the latter reaction.
References
1.
10.1016/j.mencom.2018.09.003_sbref0005a
Rouhi
C&EN,
2003
2.

Michaudel Q., Ishihara Y., Baran P.S.
Accounts of Chemical Research,
2015
3.

Busacca C.A., Fandrick D.R., Song J.J., Senanayake C.H.
Advanced Synthesis and Catalysis,
2011
4.
10.1016/j.mencom.2018.09.003_sbref0005d
Waser
Asymmetric Organocatalysis in Natural Product Syntheses,
2012
5.

Gröger H.
European Journal of Organic Chemistry,
2016
6.

Buckingham F., Gouverneur V.
Chemical Science,
2016
7.

Ermert J., Coenen H.H.
Journal of Labelled Compounds and Radiopharmaceuticals,
2013
8.
10.1016/j.mencom.2018.09.003_sbref0010c
Hanson
The Organic Chemistry of Isotopic Labelling,
2011
9.

Neumann C.N., Ritter T.
Accounts of Chemical Research,
2017
10.

Belokon Y.N., Bulychev A.G., Vitt S.V., Struchkov Y.T., Batsanov A.S., Timofeeva T.V., Tsyryapkin V.A., Ryzhov M.G., Lysova L.A.
Journal of the American Chemical Society,
1985
11.

Belokon Y.N., Bakhmutov V.I., Chernoglazova N.I., Kochetkov K.A., Vitt S.V., Garbalinskaya N.S., Belikov V.M.
Journal of the Chemical Society Perkin Transactions 1,
1988
12.
Belokon Y.N., Kochetkov K.A., Borkin D.A.
Mendeleev Communications,
2003
13.

Beloko? Y.N., Bulychev A.G., Pavlov V.A., Fedorova E.B., Tsyryapkin V.A., Bakhmutov V.A., Belikov V.M.
Journal of the Chemical Society Perkin Transactions 1,
1988
14.

Belokon' Y.N., Sagyan A.S., Djamgaryan S.M., Bakhmutov V.I., Belikov V.M.
Tetrahedron,
1988
15.

Belokoń Y.N., Popkov A.N., Chernoglazova N.I., Saporovskaya M.B., Bakhmutov V.I., Belikov V.M.
Journal of the Chemical Society Chemical Communications,
1988
16.
![Improved procedures for the synthesis of (S)-2-[N-(N′-benzylprolyl)amino]benzophenone (BPB) and Ni(II) complexes of Schiff's bases derived from BPB and amino acids](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Belokon' Y.N., Tararov V.I., Maleev V.I., Savel'eva T.F., Ryzhov M.G.
Tetrahedron Asymmetry,
1998
17.

Ko K., Wagner S., Yang S., Furkert D.P., Brimble M.A.
Journal of Organic Chemistry,
2015
18.

Zhang H., Yang B., Yang Z., Lu H., Li G.
Journal of Organic Chemistry,
2016
19.
![Synthesis, Ni(II) Schiff base complexation and structural analysis of fluorinated analogs of the ligand (S)-2-[N-(N′-benzylprolyl)amino]benzophenone (BPB)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Tatum N.J., Yufit D.S., Cobb S.L., Coxon C.R.
Journal of Fluorine Chemistry,
2015
20.

Bremerich M., Bolm C., Raabe G., Soloshonok V.A.
European Journal of Organic Chemistry,
2017
21.

Jörres M., Aceña J.L., Soloshonok V.A., Bolm C.
ChemCatChem,
2015
22.

Le Chevalier Isaad A., Barbetti F., Rovero P., D'Ursi A.M., Chelli M., Chorev M., Papini A.M.
European Journal of Organic Chemistry,
2008
23.

Lin D., Wang J., Zhang X., Zhou S., Lian J., Jiang H., Liu H.
Chemical Communications,
2013
24.
10.1016/j.mencom.2018.09.003_sbref0020h
Saghyan
J. Chem. Eng. Appl.,
2016
25.

Sorochinsky A.E., Aceña J.L., Moriwaki H., Sato T., Soloshonok V.A.
Amino Acids,
2013
26.

Sorochinsky A.E., Aceña J.L., Moriwaki H., Sato T., Soloshonok V.
Amino Acids,
2013
27.

Wang Y., Song X., Wang J., Moriwaki H., Soloshonok V.A., Liu H.
Amino Acids,
2017
28.
29.
![Stereospecificity in the [bis(ethylenediamine)-N-methyl-(S)-alaninatecobalt] (2+) and [bis(ethylenediamine)-(S)-prolinatecobalt] (2+) ions](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Buckingham D.A., Dekkers J., Sargeson A.M., Wein M.
Inorganic Chemistry,
1973