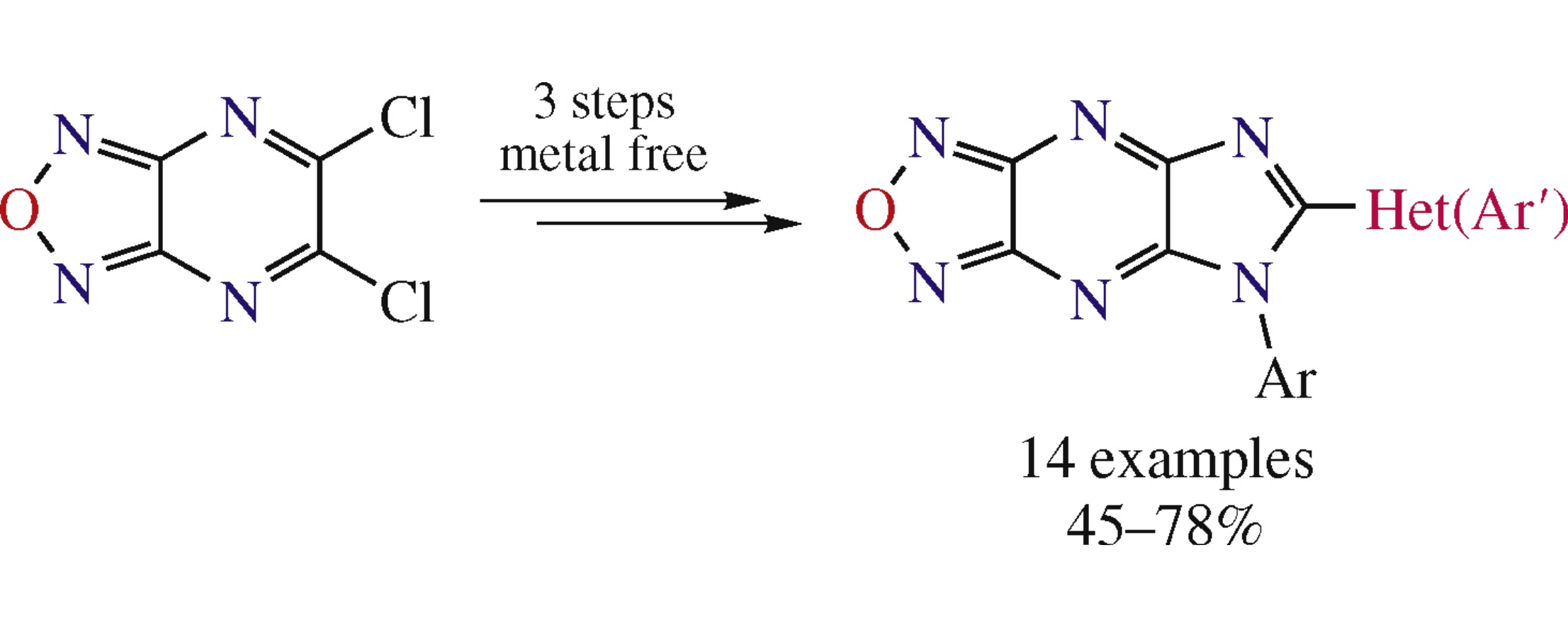
Abstract
New 5-aryl-5H-imidazo[4,5-b][1,2,5]oxadiazolo[3,4-e]pyrazines with the 6-positioned electron-rich (het)aryl substituents have been obtained from 5,6-diamino[1,2,5]oxadiazolo[3,4-b]pyrazines through the effective two-step strategy: the starting diamino compounds were treated with triethyl orthoformate, followed by the reaction with a π-excessive arene in thepresence of trifluoroacetic acid. The structures of target products were for the first time confirmed by X-ray diffraction analysis.
References
1.
H.A. Gelbard, S. Dewhurst, V.S. Goodfellow, T. Wiemann, S.B. Ravula, C.J. Loweth, US Patent 20130203755 A1, 2013.
2.
M. Sheridan, J.R. Heal, W.D.O. Hamilton, I. Pike, WO Patent 2012080729 A2, 2012.
3.
N. Skelton, S. Gradl, J.F. Blake, J.M. Graham, I.W. Gunavardana, M. Hentemann, A.L. Marlow, T.P. Tang, WO Patent 2013078254 A1, 2013.
4.
O. Hisashi, I. Toshihiro, M. Masayuki, T. Toshiki, US Patent 20020055014 A1, 2002.
5.
C.S. Kim, Y.G. Kim, Y.J. Cho, H.J. Kwon, B.O. Kim, S.M. Kim, WO Patent 2011055911 A1, 2011.
6.
E. Fuchs, N. Langer, O. Molt, K. Dormann, C. Schildknecht, S. Watanabe, G. Wagenblast, C. Lennartz, T. Schäfer, H. Wolleb, T.M. Figueira Duarte, S. Metz, P. Murer, WO Patent 2011073149 A1, 2011.
7.
L. Mataranga-Popa, R. Czerwieniec, A. Bolz, WO Patent 2015121241 A1, 2015.
8.
A. Aspuru-Guzik, R. Gomez-Bombarelli, T.D. Hirzel, J. Aguilera-Iparragurre, P. Adams, D. Maclaurin, D.K. Duvenaud, WO Patent 2015175678 A1, 2015.
9.

Hama N., Aoki T., Miwa S., Yamazaki M., Sato T., Chida N.
Organic Letters,
2010
10.
Y. Yasutomo, M. Kensuke, I. Toshihiro, JP Patent 2013258381 A, 2013.
11.
![Tricyclic Heteroaromatic Systems. Synthesis and A1 and A2a Adenosine Binding Activities of Some 1-Aryl-1,4-dihydro-3-methyl[1]benzopyrano[2,3-c]pyrazol-4-ones, 1-Aryl-4,9-dihydro-3-methyl-1H-pyrazolo[3,4-b]quinolin-4-ones, and 1-Aryl-1H-imidazo[4,5-b]quinoxalines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Catarzi D., Cecchi L., Colotta V., Filacchioni G., Martini C., Tacchi P., Lucacchini A.
Journal of Medicinal Chemistry,
1995
12.
10.1016/j.mencom.2018.09.002_bib0060
El-Sharief
Elem.,
2000
13.
![Metabolite Analogs. V. Preparation of Some Substituted Pyrazines and Imidazo [b]pyrazines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Muehlmann F.L., Day A.R.
Journal of the American Chemical Society,
1956
14.
![The chemistry of furazano-[3,4-b]pyrazine. 7. Properties of 5,6-diamino- and 5,6-dihydrazino-furazano[3,4-b]pyrazine](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Starchenkov I.B., Andrianov V.G., Mishnev A.F.
Chemistry of Heterocyclic Compounds,
1999
15.
10.1016/j.mencom.2018.09.002_bib0075
Molnár
ARKIVOC,
2010
16.
![Synthesis of Heterocyclic Quinones Containing Bridgehead Nitrogen Atom from 2-Aminonaphtho[2,3-d]thiazole-4,9-dione.](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Fandy R.F.
Heterocyclic Communications,
2000
17.
![Chemistry of furazano[3,4-b]pyrazine. 4. 5,6-Dichlorofurazano[3,4-b]pyrazine in cyclization reactions](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Starchenkov I.B., Andrianov V.G.
Chemistry of Heterocyclic Compounds,
1997
18.

Li C., Chen L., Steinhuebel D., Goodman A.
Tetrahedron Letters,
2016
19.

Groll K., Manolikakes S.M., du Jourdin X.M., Jaric M., Bredihhin A., Karaghiosoff K., Carell T., Knochel P.
Angewandte Chemie - International Edition,
2013
20.
![Synthesis of Imidazo[4,5-b]pyridines and Imidazo[4,5-b]pyrazines by Palladium Catalyzed Amidation of 2-Chloro-3-amino-heterocycles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rosenberg A.J., Zhao J., Clark D.A.
Organic Letters,
2012
21.
![An Improved Synthesis of Imidazo[4,5-b]pyridines and Imidazo[4,5-b]pyrazines by Palladium Catalyzed Amidation using Xantphos in a 1,4-Dioxane:tert-Amyl Alcohol Solvent System](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Rosenberg A.J., Ahmed I., Wilson R.J., Williams T.M., Kaminsky L., Clark D.A.
Advanced Synthesis and Catalysis,
2014
22.

Digwal C.S., Yadav U., Ramya P.V., Sana S., Swain B., Kamal A.
Journal of Organic Chemistry,
2017