Abstract
Recent achievements in the charge-transfer chemistry of 1,2,5-chalcogenadiazoles, 1,2,3-dichalcogenazoles (chalcogen is S, Se, or Te), their fused congeners and hybrids are discussed with special emphasis on the synthesis and structural and functional characterization of radical anions, radical-anion salts, charge-transfer complexes, neutral and charged donor–acceptor complexes, and metal coordination compounds.
References
1.
10.1016/j.mencom.2018.09.001_sbref0005a
Chivers
A Guide to Chalcogen–Nitrogen Chemistry,
2005
2.
10.1016/j.mencom.2018.09.001_sbref0005b
Zibarev
Selenium and Tellurium Chemistry: From Small Molecules to Biomolecules and Materials,
2011
3.

Neto B.A., Carvalho P.H., Correa J.R.
Accounts of Chemical Research,
2015
4.

Takaluoma T.T., Laasonen K., Laitinen R.S.
Inorganic Chemistry,
2013
5.

Banister A.J., Gorrell I.B.
Advanced Materials,
1998
6.

Labes M.M., Love P., Nichols L.F.
Chemical Reviews,
1979
7.

Postulka L., Winter S.M., Mihailov A.G., Mailman A., Assoud A., Robertson C.M., Wolf B., Lang M., Oakley R.T.
Journal of the American Chemical Society,
2016
8.

Winter S.M., Hill S., Oakley R.T.
Journal of the American Chemical Society,
2015
9.

Wong J.W., Mailman A., Winter S.M., Robertson C.M., Holmberg R.J., Murugesu M., Dube P.A., Oakley R.T.
Chemical Communications,
2014
10.
![Third-Order Nonlinear Optical Properties and Electroabsorption Spectra of an Organic Biradical, [Naphtho[2,1-d:6,5-d′]bis([1,2,3]dithiazole)]](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Takauji K., Suizu R., Awaga K., Kishida H., Nakamura A.
Journal of Physical Chemistry C,
2014
11.

Winter S.M., Balo A.R., Roberts R.J., Lekin K., Assoud A., Dube P.A., Oakley R.T.
Chemical Communications,
2013
12.

Winter S.M., Oakley R.T., Kovalev A.E., Hill S.
Physical Review B,
2012
13.

14.
10.1016/j.mencom.2018.09.001_sbref0015h
Stable Radicals: Fundamentals and Applied Aspects of Odd Electron Compounds,
2010
15.
10.1016/j.mencom.2018.09.001_sbref0015i
Rawson
Handbook of Chalcogen Chemistry: New Perspectives in Sulfur, Selenium and Tellurium,
2007
16.

Awaga K., Tanaka T., Shirai T., Umezono Y., Fujita W.
Comptes Rendus Chimie,
2007
17.

Saito G., Yoshida Y.
Bulletin of the Chemical Society of Japan,
2007
18.

Lonchakov A., Rakitin O., Gritsan N., Zibarev A.
Molecules,
2013
19.

Makarov A.Y., Chulanova E.A., Semenov N.A., Pushkarevsky N.A., Lonchakov A.V., Bogomyakov A.S., Irtegova I.G., Vasilieva N.V., Lork E., Gritsan N.P., Konchenko S.N., Ovcharenko V.I., Zibarev A.V.
Polyhedron,
2014
20.
Novel long-lived π-heterocyclic radical anion: a hybrid of 1,2,5-thiadiazo- and 1,2,3-dithiazolidyls
Chulanova E.A., Irtegova I.G., Vasilieva N.V., Bagryanskaya I.Y., Gritsan N.P., Zibarev A.V.
Mendeleev Communications,
2015
21.

Konstantinova L.S., Baranovsky I.V., Pritchina E.A., Mikhailov M.S., Bagryanskaya I.Y., Semenov N.A., Irtegova I.G., Salnikov G.E., Lyssenko K.A., Gritsan N.P., Zibarev A.V., Rakitin O.A.
Chemistry - A European Journal,
2017
22.

Chulanova E.A., Pritchina E.A., Malaspina L.A., Grabowsky S., Mostaghimi F., Beckmann J., Bagryanskaya I.Y., Shakhova M.V., Konstantinova L.S., Rakitin O.A., Gritsan N.P., Zibarev A.V.
Chemistry - A European Journal,
2016
23.

Semenov N.A., Gorbunov D.E., Shakhova M.V., Salnikov G.E., Bagryanskaya I.Y., Korolev V.V., Beckmann J., Gritsan N.P., Zibarev A.V.
Chemistry - A European Journal,
2018
24.
10.1016/j.mencom.2018.09.001_sbref0020g
Pushkarevsky
Chem. Eur. J.,
2018
25.

Hou J., Wang Y., Eguchi K., Nanjo C., Takaoka T., Sainoo Y., Awaga K., Komeda T.
Applied Surface Science,
2018
26.

Hamdoush M., Skvortsov I.A., Mikhailov M.S., Pakhomov G., Stuzhin P.A.
Journal of Fluorine Chemistry,
2017
27.
![DFT Study of molecular and electronic structure of magnesium (II) tetra(1,2,5-chalcogenadiazolo) porphyrazines, [TXDPzMg] (X = O, S, Se, Te)](/storage/images/resized/jZQkhOOcY9wrGwZm7XGRwMe8H7fThQu3KamISuun_small_thumb.webp)
Zhabanov Y.A., Tverdova N.V., Giricheva N.I., Girichev G.V., Stuzhin P.A.
Journal of Porphyrins and Phthalocyanines,
2017
28.
Mikhailov M.S., Hamdoush M., Islyaikin M.K., Koifman O.I., Stuzhin P.A.
Arkivoc,
2017
29.

Svec J., Zimcik P., Novakova L., Rakitin O.A., Amelichev S.A., Stuzhin P.A., Novakova V.
European Journal of Organic Chemistry,
2014
30.

Stuzhin P.A., Mikhailov M.S., Yurina E.S., Bazanov M.I., Koifman O.I., Pakhomov G.L., Travkin V.V., Sinelshchikova A.A.
Chemical Communications,
2012
31.

Miyoshi Y., Takahashi K., Fujimoto T., Yoshikawa H., Matsushita M.M., Ouchi Y., Kepenekian M., Robert V., Donzello M.P., Ercolani C., Awaga K.
Inorganic Chemistry,
2011
32.

33.
![Salts of Sterically Hindered Chalcogen-Varied Herz Cations Including Those with [Te3 Cl14 ]2- and [Te4 Cl18 ]2- Anions](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Makarov A.Y., Bagryanskaya I.Y., Volkova Y.M., Shakirov M.M., Zibarev A.V.
European Journal of Inorganic Chemistry,
2018
34.
Volkova Y.M., Makarov A.Y., Zikirin S.B., Genaev A.M., Bagryanskaya I.Y., Zibarev A.V.
Mendeleev Communications,
2017
35.

Constantinides C.P., Koutentis P.A.
Advances in Heterocyclic Chemistry,
2016
36.

Smithson C.S., MacDonald D.J., Matt Letvenuk T., Carello C.E., Jennings M., Lough A.J., Britten J., Decken A., Preuss K.E.
Dalton Transactions,
2016
37.

Makarov A.Y., Blockhuys F., Bagryanskaya I.Y., Gatilov Y.V., Shakirov M.M., Zibarev A.V.
Inorganic Chemistry,
2013
38.

Rakitin O.A.
Russian Chemical Reviews,
2011
39.

Pivtsov A.V., Kulik L.V., Makarov A.Y., Blockhuys F.
Physical Chemistry Chemical Physics,
2011
40.

Gritsan N.P., Makarov A.Y., Zibarev A.V.
Applied Magnetic Resonance,
2011
41.

Rawson J.M., Alberola A., Whalley A.
Journal of Materials Chemistry A,
2006
42.
![From Cyclopentanone Oximes to Bis[1,2,3]dithiazolo-s-indacenes, Cyclopenta[c][1,2]thiazine, Pentathiepino-, Tetrathiino-, and Thienocyclopenta[1,2,3]dithiazoles as a Rich Source of New Materials](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Macho S., Miguel D., Gómez T., Rodríguez T., Torroba T.
Journal of Organic Chemistry,
2005
43.

Koutentis P.A., Rees C.W.
Journal of the Chemical Society Perkin Transactions 1,
2002
44.
10.1016/j.mencom.2018.09.001_bib0045
IUPAC Compendium of Chemical Terminology (The Gold Book),
1997
45.

Konstantinova L., Baranovsky I., Irtegova I., Bagryanskaya I., Shundrin L., Zibarev A., Rakitin O.
Molecules,
2016
46.

Konstantinova L.S., Knyazeva E.A., Rakitin O.A.
Organic Preparations and Procedures International,
2014
47.

Neto B.A., Lapis A.A., da Silva Júnior E.N., Dupont J.
European Journal of Organic Chemistry,
2012
48.
10.1016/j.mencom.2018.09.001_sbref0050d
Todres
Chalcogenadiazoles: Chemistry and Applications,
2012
49.

Cozzolino A.F., Elder P.J., Vargas-Baca I.
Coordination Chemistry Reviews,
2011
50.
10.1016/j.mencom.2018.09.001_sbref0050f
Koutentis
2008
51.
10.1016/j.mencom.2018.09.001_sbref0050g
Rakitin
2008
52.

Konstantinova L.S., Rakitin O.A.
Russian Chemical Reviews,
2008
53.
Shundrin L.A., Irtegova I.G., Avrorov P.A., Mikhailovskaya T.F., Makarov A.G., Makarov A.Y., Zibarev A.V.
Arkivoc,
2017
54.
![Selenadiazolo[3,4-b]pyrazines: Synthesis from 3,4-Diamino-1,2,5-selenadiazole and Generation of Persistent Radical Anions](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Konstantinova L.S., Bobkova I.E., Nelyubina Y.V., Chulanova E.A., Irtegova I.G., Vasilieva N.V., Camacho P.S., Ashbrook S.E., Hua G., Slawin A.M., Woollins J.D., Zibarev A.V., Rakitin O.A.
European Journal of Organic Chemistry,
2015
55.

Vasilieva N.V., Irtegova I.G., Gritsan N.P., Lonchakov A.V., Makarov A.Y., Shundrin L.A., Zibarev A.V.
Journal of Physical Organic Chemistry,
2010
56.
Konstantinova L.S., Bol'shakov O.I., Baranovsky I.V., Bogacheva A.M., Strunyasheva V.V., Rakitin O.A.
Mendeleev Communications,
2015
57.

Konstantinova L., Knyazeva E., Rakitin O.
Molecules,
2015
58.

Mikhailov M.S., Stuzhin P.A.
Macroheterocycles,
2015
59.

Konstantinova L.S., Knyazeva E.A., Obruchnikova N.V., Gatilov Y.V., Zibarev A.V., Rakitin O.A.
Tetrahedron Letters,
2013
60.
Ul-Haq A., Donzello M.P., Stuzhin P.A.
Mendeleev Communications,
2007
61.
![Selenium for sulfur substitution in a thiazyl ring: identification of the phenyl-selenathiadiazolylium cation, [PhCNSeSN]+](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Less R.J., Rawson J.M., Jones M.
Polyhedron,
2001
62.
![Dye-sensitized solar cells: Investigation of D-A-Π-A organic sensitizers based on [1,2,5]selenadiazolo[3,4-c]pyridine](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Knyazeva E.A., Wu W., Chmovzh T.N., Robertson N., Woollins J.D., Rakitin O.A.
Solar Energy,
2017
63.

Knyazeva E.A., Rakitin O.A.
Russian Chemical Reviews,
2016
64.

Bashirov D.A., Sukhikh T.S., Kuratieva N.V., Chulanova E.A., Yushina I.V., Gritsan N.P., Konchenko S.N., Zibarev A.V.
RSC Advances,
2014
65.

Pushkarevsky N.A., Lonchakov A.V., Semenov N.A., Lork E., Buravov L.I., Konstantinova L.S., Silber G.T., Robertson N., Gritsan N.P., Rakitin O.A., Woollins J.D., Yagubskii E.B., Beckmann J., Zibarev A.V.
Synthetic Metals,
2012
66.

Gritsan N.P., Zibarev A.V.
Russian Chemical Bulletin,
2011
67.

Cozzolino A.F., Vargas-Baca I., Mansour S., Mahmoudkhani A.H.
Journal of the American Chemical Society,
2005
68.

Cozzolino A.F., Britten J.F., Vargas-Baca I.
Crystal Growth and Design,
2005
69.

Cozzolino A.F., Elder P.J., Lee L.M., Vargas-Baca I.
Canadian Journal of Chemistry,
2013
70.

Eichstaedt K., Wasilewska A., Wicher B., Gdaniec M., Połoński T.
Crystal Growth and Design,
2016
71.

Lee L.M., Elder P.J., Dube P.A., Greedan J.E., Jenkins H.A., Britten J.F., Vargas-Baca I.
CrystEngComm,
2013
72.

Bashirov D.A., Sukhikh T.S., Kuratieva N.V., Naumov D.Y., Konchenko S.N., Semenov N.A., Zibarev A.V.
Polyhedron,
2012
73.

Milios C.J., Ioannou P.V., Raptopoulou C.P., Papaefstathiou G.S.
Polyhedron,
2009
74.

Semenov N.A., Bagryanskaya I.Y., Alekseev A.V., Gatilov Y.V., Lork E., Mews R., Roeschentaler G.-., Zibarev A.V.
Journal of Structural Chemistry,
2010
75.

Shur V.B., Tikhonova I.A.
Russian Chemical Bulletin,
2003
76.

Haneline M.R., Taylor R.E., Gabbaï F.P.
Chemistry - A European Journal,
2003
77.

Tugashov K.I., Gribanyov D.A., Dolgushin F.M., Smol'yakov A.F., Peregudov A.S., Minacheva M.K., Strunin B.N., Tikhonova I.A., Shur V.B.
Journal of Organometallic Chemistry,
2013
78.

Tugashov K.I., Gribanyov D.A., Dolgushin F.M., Smol’yakov A.F., Peregudov A.S., Minacheva M.K., Tikhonova I.A., Shur V.B.
Organometallics,
2016
79.

Stephan D.W.
Accounts of Chemical Research,
2014
80.

Stephan D.W., Erker G.
Angewandte Chemie - International Edition,
2015
81.

Stephan D.W.
Organic and Biomolecular Chemistry,
2008
82.
![3D molecular network and magnetic ordering, formed by multi-dentate magnetic couplers, bis(benzene)chromium(i) and [1,2,5]thiadiazolo[3,4-: C] [1,2,5]thiadiazolidyl](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Shuku Y., Hirai Y., Semenov N.A., Kadilenko E., Gritsan N.P., Zibarev A.V., Rakitin O.A., Awaga K.
Dalton Transactions,
2018
83.
![Synthesis and Properties of the Heterospin (S1 = S2 = 1/2) Radical-Ion Salt Bis(mesitylene)molybdenum(I) [1,2,5]Thiadiazolo[3,4- c ][1,2,5]thiadiazolidyl](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
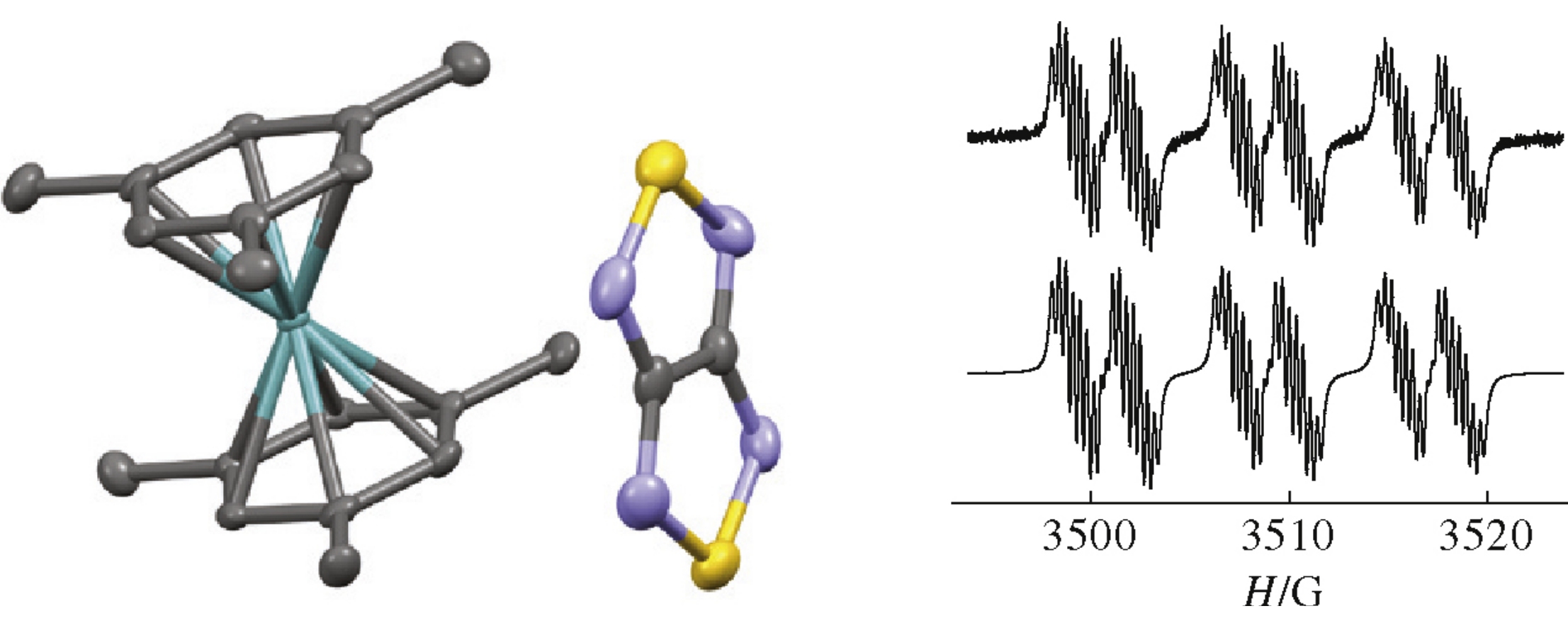
Pushkarevsky N.A., Semenov N.A., Dmitriev A.A., Kuratieva N.V., Bogomyakov A.S., Irtegova I.G., Vasilieva N.V., Bode B.E., Gritsan N.P., Konstantinova L.S., Woollins J.D., Rakitin O.A., Konchenko S.N., Ovcharenko V.I., Zibarev A.V., et. al.
Inorganic Chemistry,
2015
84.
![Bis(toluene)chromium(I) [1,2,5]Thiadiazolo[3,4-c ][1,2,5]thiadiazolidyl and [1,2,5]Thiadiazolo[3,4-b ]pyrazinidyl: New heterospin (S 1 = S 2 = 1/2) radical-ion salts](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Semenov N.A., Pushkarevsky N.A., Suturina E.A., Chulanova E.A., Kuratieva N.V., Bogomyakov A.S., Irtegova I.G., Vasilieva N.V., Konstantinova L.S., Gritsan N.P., Rakitin O.A., Ovcharenko V.I., Konchenko S.N., Zibarev A.V.
Inorganic Chemistry,
2013
85.
![Heterospin π-Heterocyclic Radical-Anion Salt: Synthesis, Structure, and Magnetic Properties of Decamethylchromocenium [1,2,5]Thiadiazolo[3,4-c][1,2,5]thiadiazolidyl](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Semenov N.A., Pushkarevsky N.A., Lonchakov A.V., Bogomyakov A.S., Pritchina E.A., Suturina E.A., Gritsan N.P., Konchenko S.N., Mews R., Ovcharenko V.I., Zibarev A.V.
Inorganic Chemistry,
2010
86.
Konchenko S.N., Gritsan N.P., Lonchakov A.V., Radius U., Zibarev A.V.
Mendeleev Communications,
2009
87.
![Cobaltocenium [1,2,5]Thiadiazolo[3,4-c][1,2,5]thiadiazolidyl: Synthesis, Structure, and Magnetic Properties](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Konchenko S.N., Gritsan N.P., Lonchakov A.V., Irtegova I.G., Mews R., Ovcharenko V.I., Radius U., Zibarev A.V.
European Journal of Inorganic Chemistry,
2008
88.
![Diamagnetic π-Dimers of the [1,2,5]Thiadiazolo[3,4-c][1,2,5]thiadiazolidyl Radical Anion in the Crystalline State: Preparation and X-ray Crystal Structure of a [(Me2N)2CC(NMe2)2]2+[(C2N4S2)2]2– Salt](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gritsan N.P., Lonchakov A.V., Lork E., Mews R., Pritchina E.A., Zibarev A.V.
European Journal of Inorganic Chemistry,
2008
89.
![[1,2,5]Selenadiazolo[3,4-c][1,2,5]thiadiazole and [1,2,5]Selenadiazolo[3,4-c][1,2,5]thiadiazolidyl – A Synthetic, Structural, and Theoretical Study](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bagryanskaya I.Y., Gatilov Y.V., Gritsan N.P., Ikorskii V.N., Irtegova I.G., Lonchakov A.V., Lork E., Mews R., Ovcharenko V.I., Semenov N.A., Vasilieva N.V., Zibarev A.V.
European Journal of Inorganic Chemistry,
2007
90.
![Early Alkali Metal (Li, Na, K) and Tris(dimethylamino)sulfonium (TAS) Salts of [1,2,5]Thiadiazolo[3,4-c][1,2,5]thiadiazolidyl Radical Anion: Rational Syntheses, Structures and Magnetic Properties](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ikorskii V.N., Irtegova I.G., Lork E., Makarov A.Y., Mews R., Ovcharenko V.I., Zibarev A.V.
European Journal of Inorganic Chemistry,
2006
91.
![[1,2,5]Thiadiazolo[3,4-c][1,2,5]thiadiazolidyl: A Long-Lived Radical Anion and Its Stable Salts](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Makarov A.Y., Irtegova I.G., Vasilieva N.V., Bagryanskaya I.Y., Borrmann T., Gatilov Y.V., Lork E., Mews R., Stohrer W., Zibarev A.V.
Inorganic Chemistry,
2005
92.
E.A. Radiush, Diploma Thesis, Novosibirsk State University, Novosibirsk, 2018.
93.
10.1016/j.mencom.2018.09.001_sbref0110b
Radiush
4th European Crystallography School (ECS4),
2017
94.
Lork E., Mews R., Zibarev A.V.
Mendeleev Communications,
2009
95.

Fedorov D.G., Koseki S., Schmidt M.W., Gordon M.S.
International Reviews in Physical Chemistry,
2003
96.
10.1016/j.mencom.2018.09.001_sbref0120b
Marian
2001
97.
10.1016/j.mencom.2018.09.001_sbref0120c
Kahn
Molecular Magnetism,
1993
98.

McConnell I mechanism promotes ferromagnetic interactions between π-stacked Ni(ii)–thiazyl complexes
Fatila E.M., Clérac R., Jennings M., Preuss K.E.
Chemical Communications,
2013
99.

Hirel C., Luneau D., Pécaut J., Öhrström L., Bussière G., Reber C.
Chemistry - A European Journal,
2002
100.
101.
![[TDNQ][CoCp*2] and [TDNQ]3[CoCp2]2; Radical Anions of a 1,2,5-Thiadiazolo-naphthoquinone](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Morgan I.S., Jennings M., Vindigni A., Clérac R., Preuss K.E.
Crystal Growth and Design,
2011
102.

Preuss K.E.
Polyhedron,
2014
103.
![The First Lanthanide Complexes with a Redox-Active Sulfur Diimide Ligand: Synthesis and Characterization of [LnCp*2(RN=)2S], Ln=Sm, Eu, Yb; R=SiMe3](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Klementyeva S.V., Gritsan N.P., Khusniyarov M.M., Witt A., Dmitriev A.A., Suturina E.A., Hill N.D., Roemmele T.L., Gamer M.T., Boeré R.T., Roesky P.W., Zibarev A.V., Konchenko S.N.
Chemistry - A European Journal,
2016
104.

105.

McKinnon J.J., Spackman M.A., Mitchell A.S.
Acta Crystallographica Section B Structural Science,
2004
106.

Pushkarevsky N.A., Petrov P.A., Grigoriev D.S., Smolentsev A.I., Lee L.M., Kleemiss F., Salnikov G.E., Konchenko S.N., Vargas-Baca I., Grabowsky S., Beckmann J., Zibarev A.V.
Chemistry - A European Journal,
2017
107.

Semenov N.A., Lonchakov A.V., Gritsan N.P., Zibarev A.V.
Russian Chemical Bulletin,
2015
108.

Semenov N.A., Lonchakov A.V., Pushkarevsky N.A., Suturina E.A., Korolev V.V., Lork E., Vasiliev V.G., Konchenko S.N., Beckmann J., Gritsan N.P., Zibarev A.V.
Organometallics,
2014
109.

Semenov N.A., Pushkarevsky N.A., Beckmann J., Finke P., Lork E., Mews R., Bagryanskaya I.Y., Gatilov Y.V., Konchenko S.N., Vasiliev V.G., Zibarev A.V.
European Journal of Inorganic Chemistry,
2012
110.

Suturina E.A., Semenov N.A., Lonchakov A.V., Bagryanskaya I.Y., Gatilov Y.V., Irtegova I.G., Vasilieva N.V., Lork E., Mews R., Gritsan N.P., Zibarev A.V.
Journal of Physical Chemistry A,
2011
111.

De Vleeschouwer F., Denayer M., Pinter B., Geerlings P., De Proft F.
Journal of Computational Chemistry,
2017
112.

Garrett G.E., Gibson G.L., Straus R.N., Seferos D.S., Taylor M.S.
Journal of the American Chemical Society,
2015
113.

Ciancaleoni G., Santi C., Ragni M., Braga A.L.
Dalton Transactions,
2015
114.

Molina P., Zapata F., Caballero A.
Chemical Reviews,
2017
115.

116.

Gale P.A., Busschaert N., Haynes C.J., Karagiannidis L.E., Kirby I.L.
Chemical Society Reviews,
2014
117.

Saravanan C., Easwaramoorthi S., Hsiow C., Wang K., Hayashi M., Wang L.
Organic Letters,
2013
118.

119.

Martínez-Máñez R., Sancenón F.
Chemical Reviews,
2003
120.

Beer P.D., Gale P.A.
Angewandte Chemie - International Edition,
2001
121.

Sukhikh T.S., Bashirov D.A., Ogienko D.S., Kuratieva N.V., Sherin P.S., Rakhmanova M.I., Chulanova E.A., Gritsan N.P., Konchenko S.N., Zibarev A.V.
RSC Advances,
2016
122.

Sukhikh T.S., Bashirov D.A., Kuratieva N.V., Smolentsev A.I., Bogomyakov A.S., Burilov V.A., Mustafina A.R., Zibarev A.V., Konchenko S.N.
Dalton Transactions,
2015
123.

Sukhikh T.S., Ogienko D.S., Bashirov D.A., Kuratieva N.V., Komarov V.Y., Rakhmanova M.I., Konchenko S.N.
Journal of Coordination Chemistry,
2016
124.

Sukhikh T.S., Bashirov D.A., Kolybalov D.S., Andreeva A.Y., Smolentsev A.I., Kuratieva N.V., Burilov V.A., Mustafina A.R., Kozlova S.G., Konchenko S.N.
Polyhedron,
2017
125.

Sukhikh T.S., Komarov V.Y., Konchenko S.N., Benassi E.
Polyhedron,
2018
126.

Sukhikh T.S., Bashirov D.A., Shuvaev S., Komarov V.Y., Kuratieva N.V., Konchenko S.N., Benassi E.
Polyhedron,
2018
127.

Sullivan D.J., Clérac R., Jennings M., Lough A.J., Preuss K.E.
Chemical Communications,
2012
128.

Sánchez-Sanz G., Trujillo C.
Journal of Physical Chemistry A,
2018
129.

Garrett G.E., Carrera E.I., Seferos D.S., Taylor M.S.
Chemical Communications,
2016
130.

Scheiner S.
Chemistry - A European Journal,
2016
131.

Alikhani E., Fuster F., Madebene B., Grabowski S.J.
Physical Chemistry Chemical Physics,
2014
132.

Bauzá A., Alkorta I., Frontera A., Elguero J.
Journal of Chemical Theory and Computation,
2013
133.

Cavallo G., Metrangolo P., Milani R., Pilati T., Priimagi A., Resnati G., Terraneo G.
Chemical Reviews,
2016
134.
10.1016/j.mencom.2018.09.001_sbref0185a
Gamez
Chem. Commun.,
2011
135.

Mukherjee A.J., Zade S.S., Singh H.B., Sunoj R.B.
Chemical Reviews,
2010
136.
10.1016/j.mencom.2018.09.001_sbref0185c
Handbook of Chalcogen Chemistry. New Perspectives in Sulfur, Selenium and Tellurium,
2007
137.

Roy D., Sunoj R.B.
Journal of Physical Chemistry A,
2006
138.

Bleiholder C., Werz D.B., Köppel H., Gleiter R.
Journal of the American Chemical Society,
2006
139.

Tiecco M., Testaferri L., Santi C., Tomassini C., Santoro S., Marini F., Bagnoli L., Temperini A., Costantino F.
European Journal of Organic Chemistry,
2006
140.

Bachrach S.M., Demoin D.W., Luk M., Miller J.V.
Journal of Physical Chemistry A,
2004
141.

Tiecco M., Testaferri L., Santi C., Tomassini C., Marini F., Bagnoli L., Temperini A.
Angewandte Chemie,
2003
142.

Tiecco M., Testaferri L., Santi C., Tomassini C., Marini F., Bagnoli L., Temperini A.
Chemistry - A European Journal,
2002
143.

Konstantinova L.S., Knyazeva E.A., Obruchnikova N.V., Vasilieva N.V., Irtegova I.G., Nelyubina Y.V., Bagryanskaya I.Y., Shundrin L.A., Sosnovskaya Z.Y., Zibarev A.V., Rakitin O.A.
Tetrahedron,
2014
144.

Konstantinova L.S., Knyazeva E.A., Gatilov Y.V., Zlotin S.G., Rakitin O.A.
Russian Chemical Bulletin,
2018
145.

Zhivonitko V.V., Makarov A.Y., Bagryanskaya I.Y., Gatilov Y.V., Shakirov M.M., Zibarev A.V.
European Journal of Inorganic Chemistry,
2005
146.
10.1016/j.mencom.2018.09.001_bib0200
Smart Materials,
2008
147.
N. D. D. Hill, R. T. Boere, 99th Canadian Chemistry Conference and Exhibition, 2016, Abstract 00809.
148.

Kaleta K., Ruhmann M., Theilmann O., Roy S., Beweries T., Arndt P., Villinger A., Jemmis E.D., Schulz A., Rosenthal U.
European Journal of Inorganic Chemistry,
2011
149.

Bestari K., Oakley R.T., Cordes A.W.
Canadian Journal of Chemistry,
1991
150.

Hunter J.A., King B., Lindsell W.E., Neish M.A.
Journal of the Chemical Society Dalton Transactions,
1980
151.

Brands G., Golloch A.
Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences,
1982
152.

153.
![Preparation and Structure of [Li2Se(NtBu)3]2, Containing the Novel Se(NtBu)32- Anion](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chivers T., Parvez M., Schatte G.
Inorganic Chemistry,
1996
154.
![Tellurium-Nitrogen Double Bonds and a Novel Te3N3 Ring: Formation and Structures of [(tBuNH)(tBuN)3Te2]Cl, [tBuNTeNtBu]2, and [tBuNTe]3](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chivers T., Gao X., Parvez M.
Journal of the American Chemical Society,
1995
155.

Roesky H.W., Schmieder W., Isenberg W., Sheldrick W.S., Sheldrick G.M.
Chemische Berichte,
1982
156.

Kuyper J., Vrieze K.
Journal of the Chemical Society Chemical Communications,
1976