Abstract
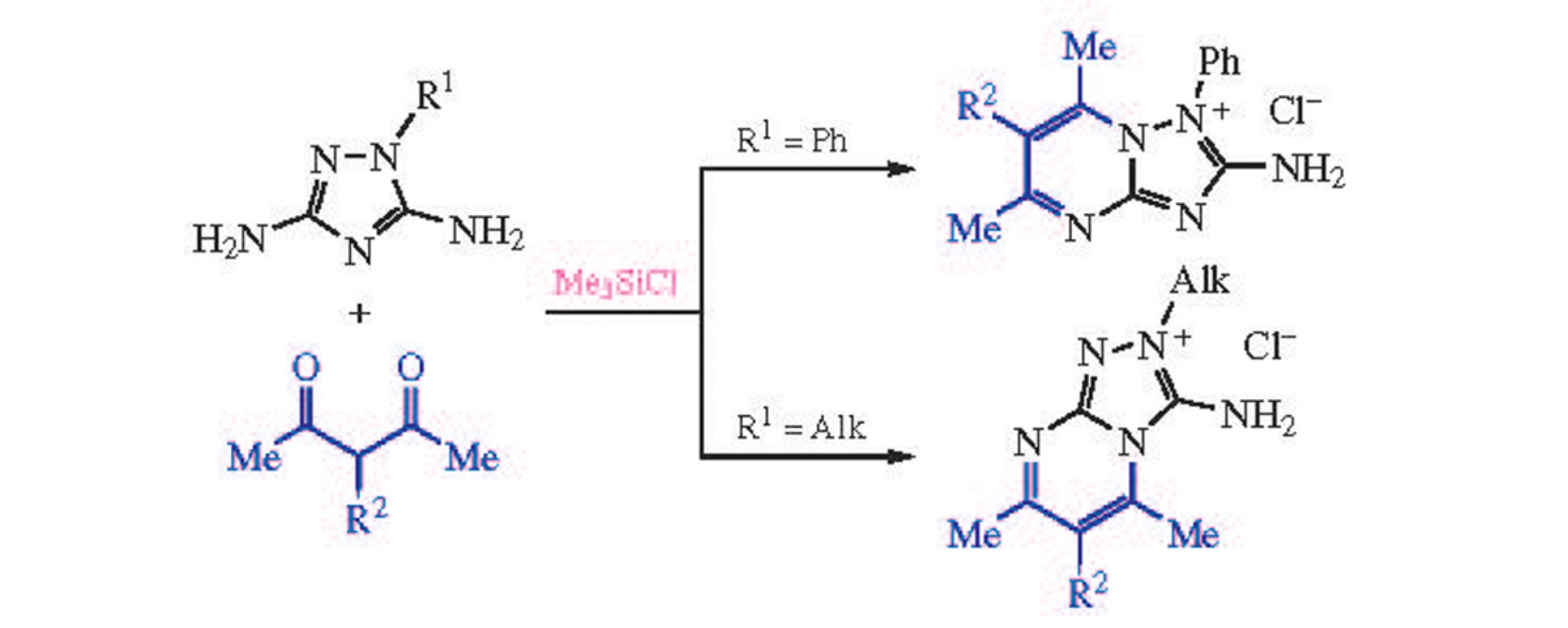
An efficient synthesis of 2-amino-1-R-[1,2,4]triazolo[1,5-a]-pyrimidinium or 3-amino-2-R-[1,2,4]triazolo[4,3-a]pyrimidi- nium chloride derivatives by heterocyclization of 3,5-diamino-1-R-1,2,4-triazoles (R=Alk or Ar) with pentane-2,4-diones was developed. The process is promoted by chlorotrimethyl- silane which plays the dual role of carbonyl-activating agent and water scavenger.
References
1.
![Recent Progress in 1,2,4-Triazolo[1,5-a]pyrimidine Chemistry](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fischer G.
Advances in Heterocyclic Chemistry,
2007
2.
![Synthesis of [1,2,4]triazolo[1,5-a]pyrimidine (microreview)](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Fizer M., Slivka M.
Chemistry of Heterocyclic Compounds,
2016
3.
![Application of 1,2,4-triazolo[1,5 -a ]pyrimidines for the design of coordination compounds with interesting structures and new biological properties](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Łakomska I., Fandzloch M.
Coordination Chemistry Reviews,
2016
4.
![Leishmanicidal and Trypanocidal Activity of Metal Complexes with 1,2,4-Triazolo[1,5-a]pyrimidines: Insights on their Therapeutic Potential against Leishmaniasis and Chagas Disease.](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
Salas J.M., B. Caballero A., Esteban-Parra G.M., Méndez-Arriaga J.M.
Current Medicinal Chemistry,
2017
5.
Prezent M.A., Daeva E.D., Baranin S.V., Zavarzin I.V.
Mendeleev Communications,
2017
6.

Saloutin V.I., Goryaeva M.V., Khudina O.G., Ivanova A.E., Burgart Y.V.
Russian Chemical Bulletin,
2016
7.

Kim S., Kim H., Lee J., Jin W.J., Hwang S.J., Kim H., Ha H., Lee Z.H.
Biochemical Pharmacology,
2013
8.

Feng Y., Gao Y., Zhang Y., Dong L., Li J.
Weed Science,
2016
9.

Guan A., Liu C., Yang X., Dekeyser M.
Chemical Reviews,
2014
10.

Zhu X., Zhang M., Liu J., Ge J., Yang G.
Journal of Agricultural and Food Chemistry,
2015
11.

Coteron J.M., Marco M., Esquivias J., Deng X., White K.L., White J., Koltun M., El Mazouni F., Kokkonda S., Katneni K., Bhamidipati R., Shackleford D.M., Angulo-Barturen I., Ferrer S.B., Jiménez-Díaz M.B., et. al.
Journal of Medicinal Chemistry,
2011
12.

Jakobsen J.C., Nielsen E.E., Feinberg J., Katakam K.K., Fobian K., Hauser G., Poropat G., Djurisic S., Weiss K.H., Bjelakovic M., Bjelakovic G., Klingenberg S.L., Liu J.P., Nikolova D., Koretz R.L., et. al.
Cochrane Database of Systematic Reviews,
2017
13.
![Novel agonists of benzodiazepine receptors: Design, synthesis, binding assay and pharmacological evaluation of 1,2,4-triazolo[1,5-a]pyrimidinone and 3-amino-1,2,4-triazole derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Faizi M., Dabirian S., Tajali H., Ahmadi F., Zavareh E.R., Shahhosseini S., Tabatabai S.A.
Bioorganic and Medicinal Chemistry,
2015
14.

Gomez L., Massari M.E., Vickers T., Freestone G., Vernier W., Ly K., Xu R., McCarrick M., Marrone T., Metz M., Yan Y.G., Yoder Z.W., Lemus R., Broadbent N.J., Barido R., et. al.
Journal of Medicinal Chemistry,
2017
15.
![Partially hydrogenated 2-amino[1,2,4]triazolo[1,5- a ]pyrimidines as synthons for the preparation of polycondensed heterocycles: reaction with chlorocarboxylic acid chlorides](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chernyshev V.M., Pyatakov D.A., Sokolov A.N., Astakhov A.V., Gladkov E.S., Shishkina S.V., Shishkin O.V.
Tetrahedron,
2014
16.
![Partially hydrogenated 2-amino[1,2,4]triazolo[1,5-a]pyrimidines as synthons for the preparation of polycondensed heterocycles: reaction with α-bromoketones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chernyshev V.M., Pyatakov D.A., Astakhov A.V., Sokolov A.N., Fakhrutdinov A.N., Rybakov V.B., Chernyshev V.V.
Tetrahedron,
2015
17.
![Diversity Oriented Synthesis of Polycyclic Heterocycles through the Condensation of 2-Amino[1,2,4]triazolo[1,5-a]pyrimidines with 1,3-Diketones.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Pyatakov D.A., Sokolov A.N., Astakhov A.V., Chernenko A.Y., Fakhrutdinov A.N., Rybakov V.B., Chernyshev V.V., Chernyshev V.M.
Journal of Organic Chemistry,
2015
18.

Pyatakov D.A., Astakhov A.V., Sokolov A.N., Fakhrutdinov A.N., Fitch A.N., Rybakov V.B., Chernyshev V.V., Chernyshev V.M.
Tetrahedron Letters,
2017
19.
![Efficient and regioselective one-step synthesis of 7-aryl-5-methyl- and 5-aryl-7-methyl-2-amino-[1,2,4]triazolo[1,5-a]pyrimidine derivatives](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Massari S., Desantis J., Nannetti G., Sabatini S., Tortorella S., Goracci L., Cecchetti V., Loregian A., Tabarrini O.
Organic and Biomolecular Chemistry,
2017
20.
10.1016/j.mencom.2018.07.034_bib0150
Sedash
RSCAdv,
2012
21.

Chernyshev V.M., Vlasova A.G., Astakhov A.V., Shishkina S.V., Shishkin O.V.
Journal of Organic Chemistry,
2014
22.
![Reaction of 1-substituted 3,5-diamino-1,2,4-triazoles with β-keto esters: synthesis and new rearrangement of mesoionic 3-amino-2H-[1,2,4]triazolo-[4,3-a]pyrimidin-5-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chernyshev V.M., Astakhov A.V., Starikova Z.A.
Tetrahedron,
2010
23.
![Synthesis of mesoionic 1,2,4-triazolo[4,3-a]pyrimidin-5-ones and subsituted formamidines from diethyl (5-1-r-1h-1,2,4-triazol -3-yl)aminomethylenemalonates](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Astakhov A.V., Chernyshev V.M.
Chemistry of Heterocyclic Compounds,
2010
24.
![Synthesis of [1,2,4]triazolo[4,3-a]pyrimidin-5(1H)-ones by the Condensation of 3-Alkylamino-5-amino-1-phenyl[1,2,4]triazoles with β-Keto Esters or Diethyl Ethoxymethylenemalonate](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Astakhov A.V., Zubatyuk R.I., Abagyan R.S., Chernyshev V.M.
Chemistry of Heterocyclic Compounds,
2014
25.
Azev Y.A., Kodess M.I., Ezhikova M.A., Ermakova O.S., Berseneva V.S., Bakulev V.A.
Mendeleev Communications,
2017
26.
10.1016/j.mencom.2018.07.034_bib0130
Pashinnik
Ukr. Khim. Zh.,
1973
27.
![Synthesis and rearrangement of 3-Amino-2-Benzyl[1,2,4]Triazolo[4,3-a]Pyrimidinium Salts](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Astakhov A.V., Chernyshev V.M.
Chemistry of Heterocyclic Compounds,
2012
28.

Dilman A.D., Ioffe S.L.
Chemical Reviews,
2003
29.

Volochnyuk D., Ryabukhin S., Plaskon A., Grygorenko O.
Synthesis,
2009