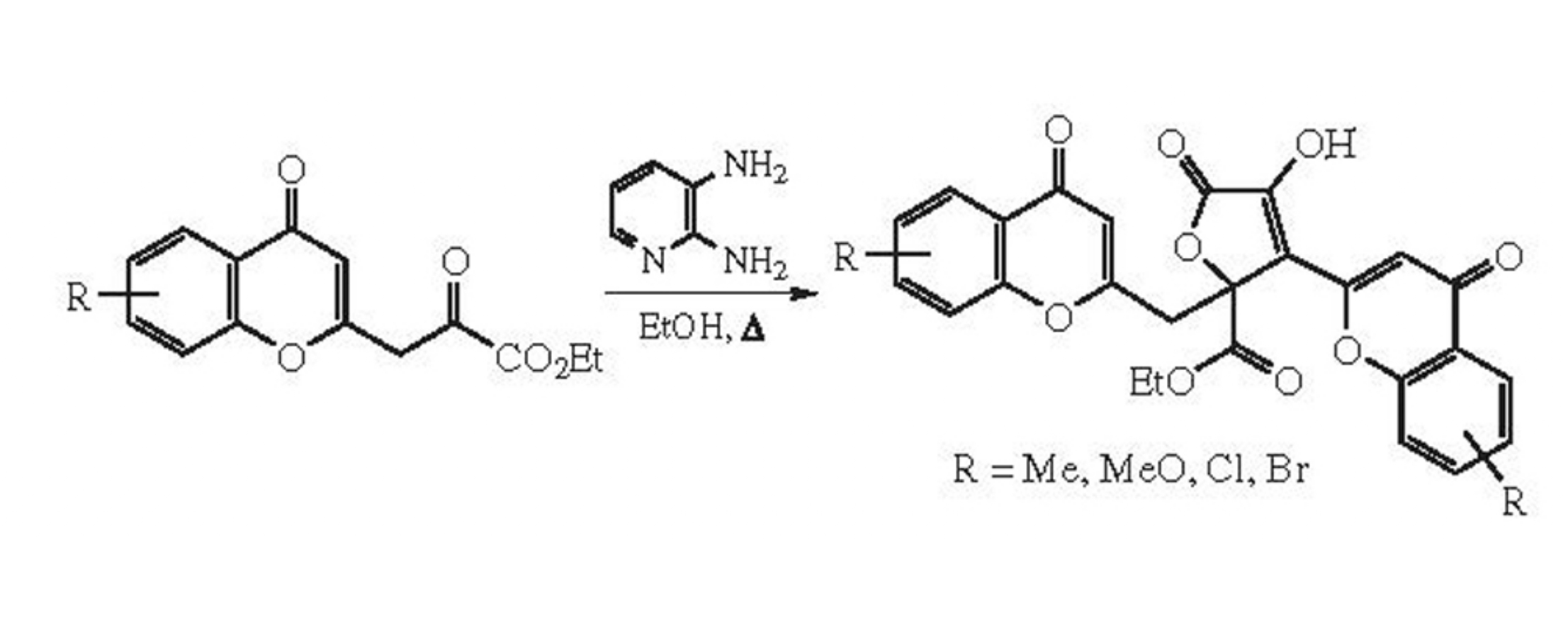
Abstract
2-(Ethoxalylmethyl)chromones undergo dimerization to dichromonyl derivatives of isotetronic acid in 62–89% yields on refluxing with 2,3-diaminopyridine in ethanol for 1–2h.
References
1.

Monnin J.
Helvetica Chimica Acta,
1957
2.

Sakan T., Kato M., Miwa T., Nakamura N.
Bulletin of the Chemical Society of Japan,
1964
3.

Lohrisch H., Kopanski L., Herrmann R., Schmidt H., Steglich W.
Liebigs Annalen der Chemie,
1986
4.

Tejedor D., Santos-Expósito A., García-Tellado F.
Chemical Communications,
2006
5.

Tejedor D., Sántos-Expósito A., García-Tellado F.
Chemistry - A European Journal,
2007
6.

Janin Y.L.
Journal of Heterocyclic Chemistry,
2013
7.
10.1016/j.mencom.2018.07.032_sbref0005g
Sun
Synthesis,
2008
8.

Dambruoso P., Massi A., Dondoni A.
Organic Letters,
2005
9.

SULSER H., DePIZZOL J., BÜHI W.
Journal of Food Science,
1967
10.
10.1016/j.mencom.2018.07.032_sbref0010b
Takahashi
Agric. Biol. Chem,
1976
11.

Zymonic Acid, a New Metabolic Product of Some Yeasts Grown in Aerated Culture. I. Structure Studies2
Stodola F.H., Shotwell O.L., Lockwood L.B.
Journal of the American Chemical Society,
1952
12.

Bloomer J.L., Gross M.A.
Journal of the Chemical Society D Chemical Communications,
1970
13.

Bloomer J.L., Gross M.A., Kappler F.E., Pandey G.N.
Journal of the Chemical Society D Chemical Communications,
1970
14.

Anderson J.R., Edwards R.L., Whalley A.J.
Journal of the Chemical Society Perkin Transactions 1,
1982
15.
Chen H., Ma X., Li Z., Wang Q., Tao F.
Arkivoc,
2009
16.

Düwel S., Hundshammer C., Gersch M., Feuerecker B., Steiger K., Buck A., Walch A., Haase A., Glaser S.J., Schwaiger M., Schilling F.
Nature Communications,
2017
17.

NITTA K., FUJITA N., YOSHIMURA T., ARAI K., YAMAMOTO Y.
Chemical and Pharmaceutical Bulletin,
2011
18.

Suzuki M., Hosaka Y., Matsushima H., Goto T., Kitamura T., Kawabe K.
Cancer Letters,
1999
19.
10.1016/j.mencom.2018.07.032_sbref0030b
Kitagawa
Oncogene,
1993
20.
10.1016/j.mencom.2018.07.032_sbref0030c
Kitagawa
Oncogene,
1994
21.

Huwe A., Mazitschek R., Giannis A.
Angewandte Chemie - International Edition,
2003
22.

Horton D.A., Bourne G.T., Smythe M.L.
Chemical Reviews,
2003
23.

Shcherbakov K.V., Burgart Y.V., Saloutin V.I., Chupakhin O.N.
Russian Chemical Bulletin,
2016
24.

Safrygin A.V., Sosnovskikh V.Y.
Russian Chemical Reviews,
2017
25.
Kornev M.Y., Moshkin V.S., Eltsov O.S., Sosnovskikh V.Y.
Mendeleev Communications,
2016
26.

Jones W.D.
Journal of the Chemical Society Perkin Transactions 1,
1981
27.
10.1016/j.mencom.2018.07.032_sbref0045a
Ibrahim
Elem.,
2002
28.
![Novel method for the synthesis of 3-[(3-arylpyrazol-5-yl)methylidene]-quinoxalin-2-ones on the basis of 2-methylchromones](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Safrygin A.V., Vetyugova D.A., Sosnovskikh V.Y.
Chemistry of Heterocyclic Compounds,
2016