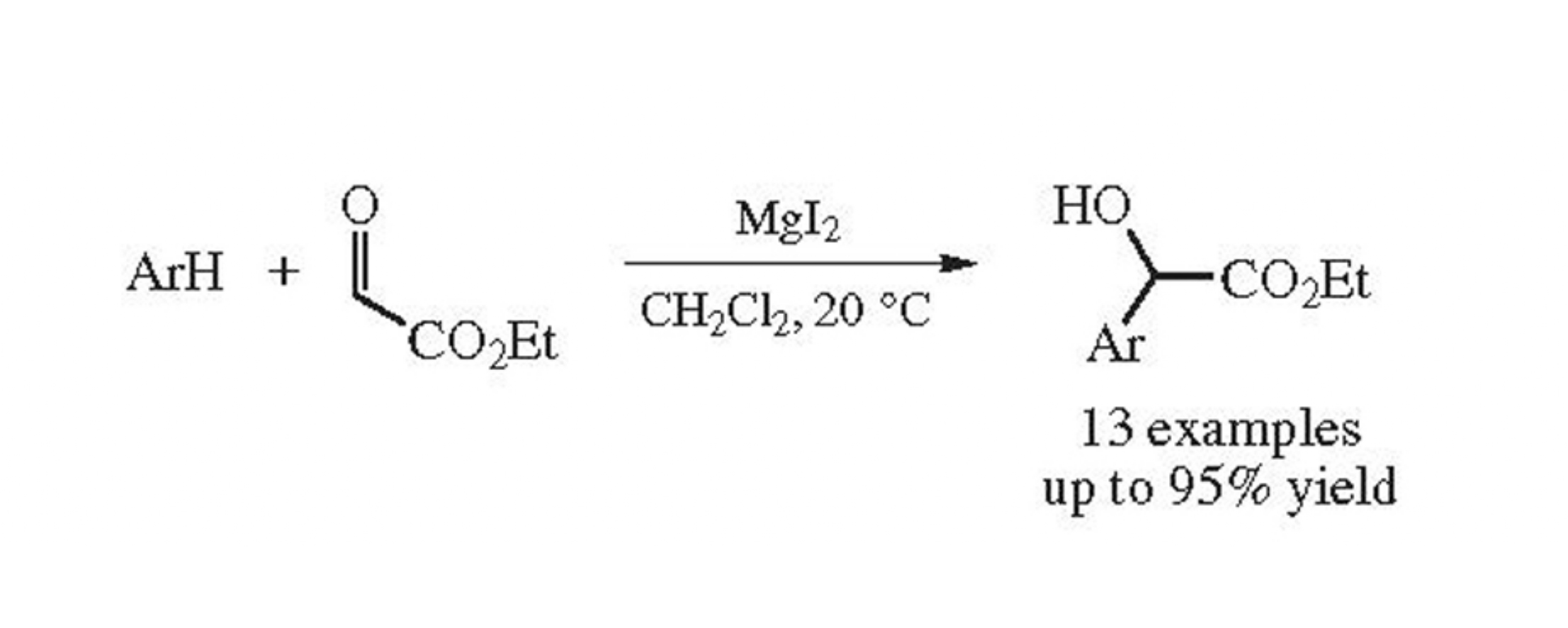
Abstract
Magnesium iodide-catalyzed addition of electron-rich (het)arenes to ethyl glyoxylate proceeds at room temperature with high chemoselectivity to afford ethyl 2-(het)aryl- 2-hydroxyacetates in yields up to 95%.
References
1.
L. H. Baekeland, Patent US 942699, 1909.
2.

Fuson R.C., Weinstock H.H., Ullyot G.E.
Journal of the American Chemical Society,
1935
3.

Menegheli P., Rezende M.C., Zucco C.
Synthetic Communications,
1987
4.
![Synthesis of 1,3-disubstituted-pyrrolo[2,1-a]isoquinoline-2-carboxylic acids, esters and amides](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Bridge A.W., Fenton G., Halley F., Hursthouse M.B., Lehmann C.W., Lythgoe D.J.
Journal of the Chemical Society Perkin Transactions 1,
1993
5.

Adams S.R., Kao J.P., Grynkiewicz G., Minta A., Tsien R.Y.
Journal of the American Chemical Society,
1988
6.

Lyle M.P., Draper N.D., Wilson P.D.
Organic Letters,
2005
7.

Li C., Guo F., Xu K., Zhang S., Hu Y., Zha Z., Wang Z.
Organic Letters,
2014
8.

Yuan Y., Wang X., Li X., Ding K.
Journal of Organic Chemistry,
2003
9.

Huang Z., Zhang J., Zhou Y., Wang N.
European Journal of Organic Chemistry,
2010
10.

Blay G., Fernández I., Monleón A., Pedro J.R., Vila C.
Organic Letters,
2008
11.

Ishii A., Soloshonok V.A., Mikami K.
Journal of Organic Chemistry,
2000
12.

Majer J., Kwiatkowski P., Jurczak J.
Organic Letters,
2009
13.

Blay G., Fernández I., Muñoz M.C., Pedro J.R., Recuenco A., Vila C.
Journal of Organic Chemistry,
2011
14.

Zhuang W., Gathergood N., Hazell R.G., Jørgensen K.A.
Journal of Organic Chemistry,
2001
15.

Ren H., Wang P., Wang L., Tang Y.
Organic Letters,
2015
16.

Zhao J., Liu L., Sui Y., Liu Y., Wang D., Chen Y.
Organic Letters,
2006
17.

Aikawa K., Asai Y., Hioki Y., Mikami K.
Tetrahedron Asymmetry,
2014
18.

Kochanowska-Karamyan A.J., Hamann M.T.
Chemical Reviews,
2010
19.

Soueidan M., Collin J., Gil R.
Tetrahedron Letters,
2006
20.

Dong H., Lu H., Lu L., Chen C., Xiao W.
Advanced Synthesis and Catalysis,
2007
21.
10.1016/j.mencom.2018.07.030_bib0105
Willot
Synlett,
2009
22.

Hui Y., Zhang Q., Jiang J., Lin L., Liu X., Feng X.
Journal of Organic Chemistry,
2009
23.

Zhuang W., Jørgensen K.A.
Chemical Communications,
2002
24.

Beletskaya I., Feofanov M., Anokhin M., Averin A.
Synthesis,
2017
25.

Beletskaya I., Desyatkin V.
Synthesis,
2017
26.
10.1016/j.mencom.2018.07.030_bib0130
Tarasenko
Synthesis,
2017
27.

Desyatkin V.G., Anokhin M.V., Rodionov V.O., Beletskaya I.P.
Russian Journal of Organic Chemistry,
2016
28.

P. Beletskaya I., D. Averin A.
Current Organocatalysis,
2015
29.

Anokhin M.V., Feofanov M.N., Averin A.D., Beletskaya I.P.
ChemistrySelect,
2018
30.
31.
Lozhkin B.A., Shlyakhtin A.V., Bagrov V.V., Ivchenko P.V., Nifant’ev I.E.
Mendeleev Communications,
2018