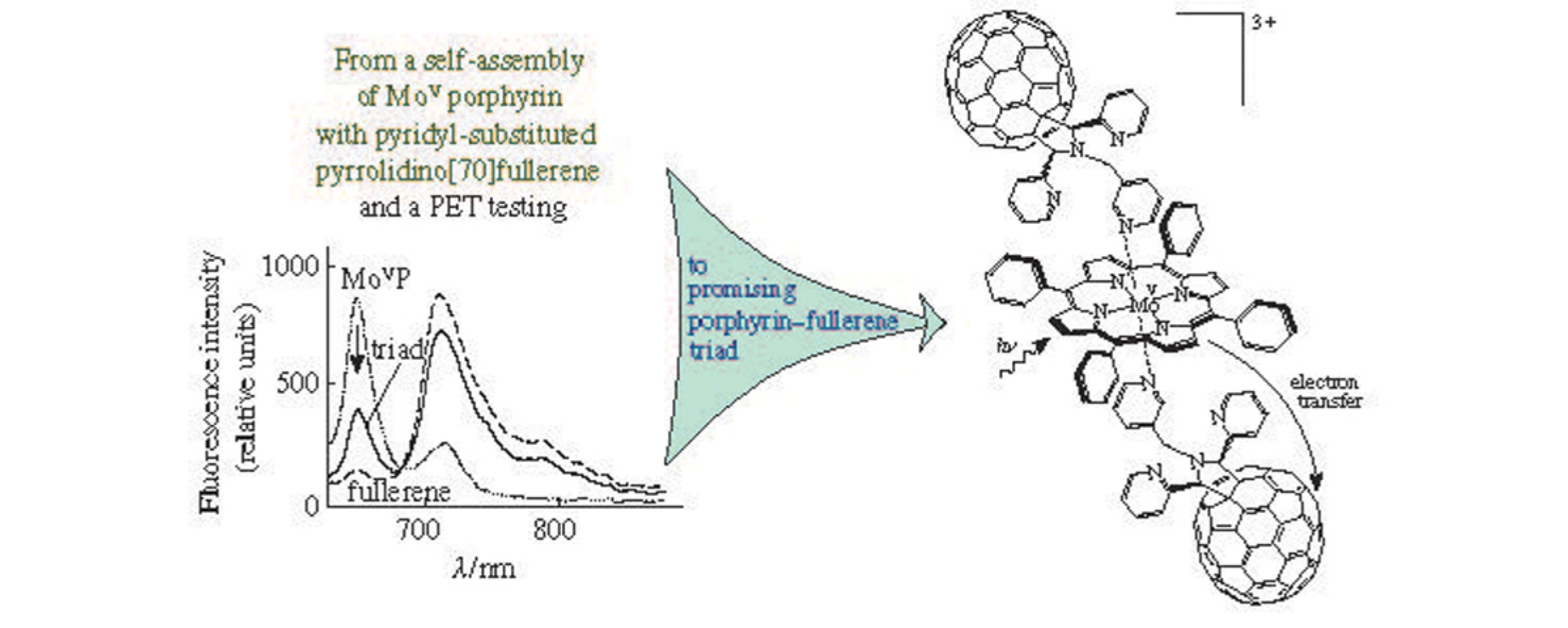
Abstract
Complexation of (hydroxy)(oxo)(5,10,15,20-tetraphenylporphyrinato) molybdenum(v) with 2’,5’-di(2-pyridyl)-1’-(3-pyridylmethyl) pyrrolidino[70]fullerene leading to a new donor–acceptor triad is characterized by quantitative description of the equilibrium and the reaction rate. The prospects of the triad as a photosynthetic antenna imitator and an active layer in solar energy conversion devices are substantiated.
References
1.
Biglova Y.N., Susarova D.K., Akbulatov A.F., Mustafin A.G., Troshin P.A., Miftakhov M.S.
Mendeleev Communications,
2015
2.
B. Grimm, A. Hausmann, A. Kahnt, W. Seitz, F. Spänig and D. M. Guldi, in Handbook of Porphyrin Science with Applications to Chemistry, Physics, Materials Science, Engineering, Biology and Medicine, eds. K. M. Kadish, K.M. Smith and R. Guilard, World Scientific, Singapore, 2012, vol. 1, pp. 133-220.
3.

Krinichnyi V.I., Yudanova E.I., Denisov N.N.
Polymer Science - Series A,
2010
4.
10.1016/j.mencom.2018.07.029_bib0020
Paraschuk
Ross. Khim. Zh.,
2008
5.

Kostyanovskiy V.A., Troshin P.A., Adam G., Sariciftci N.S., Razumov V.F.
Energy Procedia,
2012
6.

KC C.B., D'Souza F.
Coordination Chemistry Reviews,
2016
7.
P. O. Krasnov and N. S. Eliseeva, Fazovye Perekhody, Uporyadochennye Sostoyaniya i Novye Materialy, 2010, 1 (in Russian).
8.
![Synthesis and spectroscopic studies of axially bound tetra(phenothiazinyl)/tetra(bis(4′-tert-butylbiphenyl-4-yl)aniline)-zinc(II)porphyrin-fullero[C60 & C70]pyrrolidine donor–acceptor triads](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Jain K., Duvva N., Badgurjar D., Giribabu L., Chitta R.
Inorganic Chemistry Communication,
2016
9.
10.1016/j.mencom.2018.07.029_bib0045
Lomova
Russ. J. Coord. Chem.,
1993
10.

Lomova T.N., Berezin B.D.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2001
11.
10.1016/j.mencom.2018.07.029_bib0055
Tipugina
Russ. J. Phys. Chem.,
2002
12.

Ovchenkova E.N., Bichan N.G., Lomova T.N.
Tetrahedron,
2015
13.

New soluble octakis-substituted Co(II) phthalocyanines: Synthesis, spectra, supramolecular chemistry
Ovchenkova E.N., Bichan N.G., Lomova T.N.
Dyes and Pigments,
2016
14.

Gnichwitz J., Wielopolski M., Hartnagel K., Hartnagel U., Guldi D.M., Hirsch A.
Journal of the American Chemical Society,
2008
15.
10.1016/j.mencom.2018.07.029_bib0075
Lomova
Zh. Neorg. Khim.,
1985
16.

Troshin P.A., Peregudov A.S., Troyanov S.I., Lyubovskaya R.N.
Russian Chemical Bulletin,
2008
17.
10.1016/j.mencom.2018.07.029_bib0085
Lomova
Makrogeterocycles/Makrogeterotsikly,
2013
18.
![Novel 2′-(pyridin-4-yl)-5′-(pyridin-2-yl)-1′-(pyridin-2-yl)methylpyrrolidinyl[60]fullerene-hydroxyoxo(5,10,15,20-tetraphenyl-21H,23H-porphynato) molybdenum(V) dyads](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Motorina E.V., Lomova T.N., Troshin P.A., Klyuev M.V.
Russian Journal of General Chemistry,
2014
19.

Konarev D.V., Khasanov S.S., Kornev A.B., Faraonov M.A., Troshin P.A., Lyubovskaya R.N.
Dalton Transactions,
2012
20.

Sessler J.L., Jayawickramarajah J., Gouloumis A., Torres T., Guldi D.M., Maldonado S., Stevenson K.J.
Chemical Communications,
2005
21.

22.
K. Nakanishi, Infrared Absorption Spectroscopy, Holden-Day, Inc. San Francisco and Nankodo Company Ltd. Tokyo, 1962.
23.
A. J. Gordon and R. A Ford, The Chemist's Companion: A Handbook of Practical Data, Techniques, and References, Wiley, 1972.
24.

Cheremisina I.M.
Journal of Structural Chemistry,
1978
25.

Nojiri T., Watanabe A., Ito O.
Journal of Physical Chemistry A,
1998
26.

Liddell P.A., Sumida J.P., Macpherson A.N., Noss L., Seely G.R., Clark K.N., Moore A.L., Moore T.A., Gust D.
Photochemistry and Photobiology,
1994
27.
![Spectroscopic study of the reaction of cis-1,3-di(2-pyridyl)[60]fullereno[1,2-c]pyrrolidine and 2-(2-pyridylmethyl)-1,3-di(2-pyridyl)[60]fullereno[1,2-c]pyrrolidine with zinc meso-tetraphenylporphyrinate](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Lapshin A.N., Smirnov V.A., Lyubovskaya R.N., Gol’dshleger N.F.
Russian Chemical Bulletin,
2005