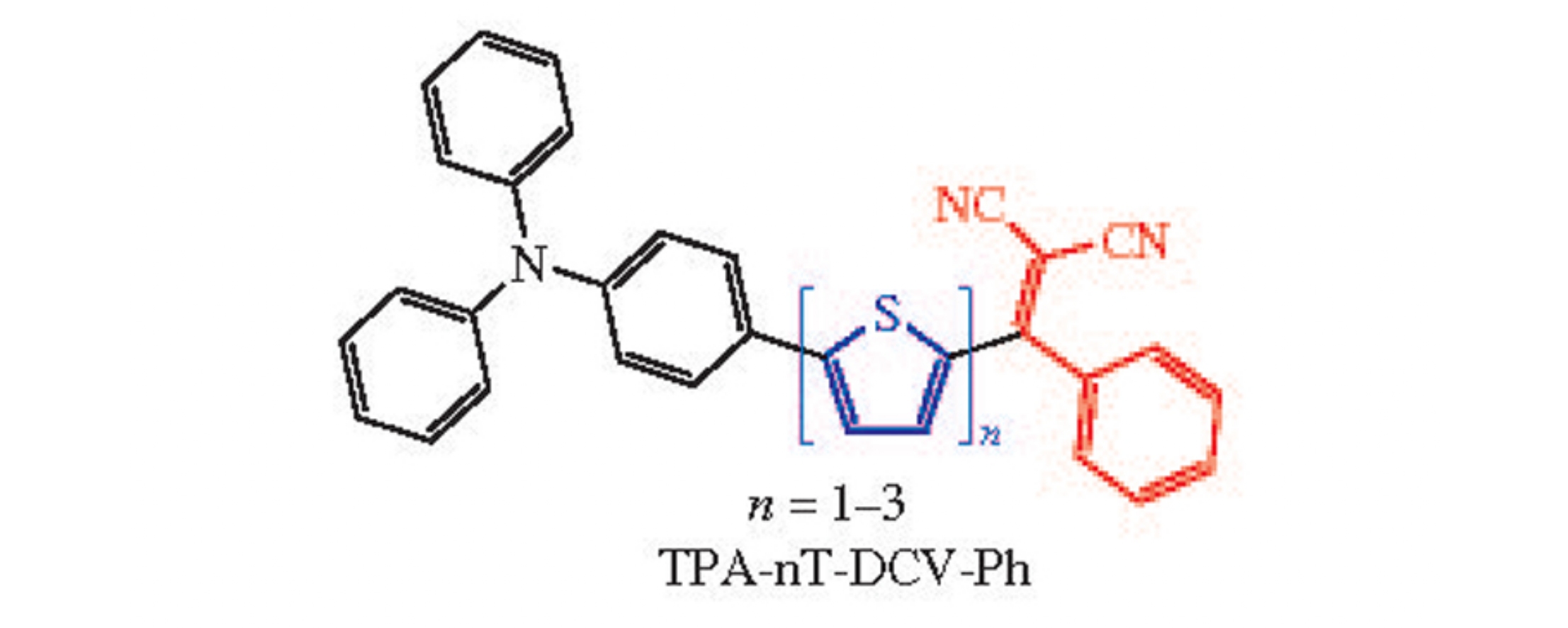
Abstract
Highly stable unsymmetrical donor–acceptor oligothiophenes equipped with terminal electron-donating triphenylamine and an electron-withdrawing phenyldicyanovinyl groups have been synthesized. An influence of the length of conjugated oligothiophene π-spacer between the donor and acceptor blocks on solubility, thermal, optical and electrochemical properties of such compounds has been revealed.
References
1.

Mishra A., Ma C., Bäuerle P.
Chemical Reviews,
2009
2.

Ouyang X., Peng R., Ai L., Zhang X., Ge Z.
Nature Photonics,
2015
3.

Hahn L., Hermannsdorfer A., Günther B., Wesp T., Bühler B., Zschieschang U., Wadepohl H., Klauk H., Gade L.H.
Journal of Organic Chemistry,
2017
4.

Tuktarov A.R., Khuzin A.A., Dzhemilev U.M.
Russian Chemical Reviews,
2017
5.

Pushkarev A.P., Bochkarev M.N.
Russian Chemical Reviews,
2016
6.

Leliège A., Grolleau J., Allain M., Blanchard P., Demeter D., Rousseau T., Roncali J.
Chemistry - A European Journal,
2013
7.

Gupta A., Ali A., Gao M., Singh T.B., Bilic A., Watkins S.E., Bach U., Evans R.A.
Dyes and Pigments,
2015
8.

Ripaud E., Rousseau T., Leriche P., Roncali J.
Advanced Energy Materials,
2011
9.

Luponosov Y.N., Solodukhin A.N., Mannanov A.L., Trukhanov V.A., Peregudova S.M., Pisarev S.A., Bakirov A.V., Shcherbina M.A., Chvalun S.N., Paraschuk D.Y., Ponomarenko S.A.
Organic Electronics,
2017
10.

Jiang Y., Cabanetos C., Allain M., Liu P., Roncali J.
Journal of Materials Chemistry C,
2015
11.

Jiang Y., Gindre D., Allain M., Liu P., Cabanetos C., Roncali J.
Advanced Materials,
2015
12.

Kozlov O.V., Luponosov Y.N., Solodukhin A.N., Flament B., Douhéret O., Viville P., Beljonne D., Lazzaroni R., Cornil J., Ponomarenko S.A., Pshenichnikov M.S.
Organic Electronics,
2018
13.

Shirota Y.
Journal of Materials Chemistry A,
2005
14.

Kanibolotsky A.L., Perepichka I.F., Skabara P.J.
Chemical Society Reviews,
2010
15.

Kagan J., Arora S.K.
Journal of Organic Chemistry,
1983
16.

Fitzner R., Reinold E., Mishra A., Mena-Osteritz E., Ziehlke H., Körner C., Leo K., Riede M., Weil M., Tsaryova O., Weiß A., Uhrich C., Pfeiffer M., Bäuerle P.
Advanced Functional Materials,
2011
17.

Min J., Luponosov Y.N., Baran D., Chvalun S.N., Shcherbina M.A., Bakirov A.V., Dmitryakov P.V., Peregudova S.M., Kausch-Busies N., Ponomarenko S.A., Ameri T., Brabec C.J.
Journal of Materials Chemistry A,
2014
18.

Meier H., Gerold J., Jacob D.
Tetrahedron Letters,
2003
19.

Meier H.
Angewandte Chemie - International Edition,
2005
20.

Bakiev A.N., Selivanova D.G., Lunegov I.V., Vasyanin A.N., Maiorova O.A., Gorbunov A.A., Shklyaeva E.V., Abashev G.G.
Chemistry of Heterocyclic Compounds,
2016