Abstract
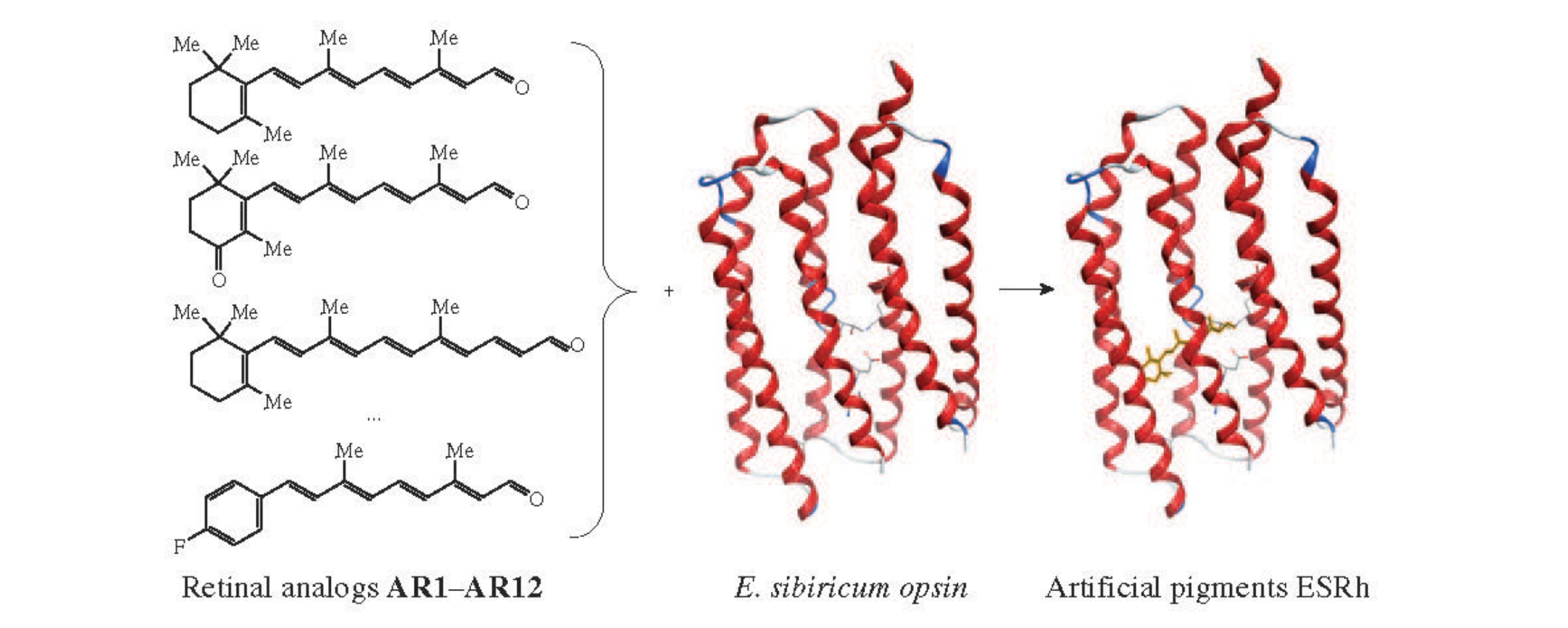
The effects of chromophoric group structures on the functional properties of bacteriorhodopsin (BR) and proteorhodopsin from E. sibiricum (ESRh) were compared. ESRh retinal binding site was found as preserving the similar stereo- and spatial restrictions on the chromophore structure during the retinal protein reconstitution process (except for C25-analog AR8). It was revealed that the structure peculiarities of the chromophore analog molecules affect the optical parameters of ESRh and BR pigment families in similar ways.
References
1.

OESTERHELT D., STOECKENIUS W.
Nature New Biology,
1971
2.
A. A. Khodonov, N.E. Belikov and O. V. Demina, Properties of Artificial Bacteriorhodopsin Analogs, ed. V. V. Chupin, IBCP/MIPT, Moscow, 2017, version 1, https://cmm-mipt.ru/bacteriorhodopsin-analogs.html.
3.

Petrovskaya L.E., Lukashev E.P., Chupin V.V., Sychev S.V., Lyukmanova E.N., Kryukova E.A., Ziganshin R.H., Spirina E.V., Rivkina E.M., Khatypov R.A., Erokhina L.G., Gilichinsky D.A., Shuvalov V.A., Kirpichnikov M.P.
FEBS Letters,
2010
4.

Petrovskaya L.E., Shulga A.A., Bocharova O.V., Ermolyuk Y.S., Kryukova E.A., Chupin V.V., Blommers M.J., Arseniev A.S., Kirpichnikov M.P.
Biochemistry (Moscow),
2010
5.

Balashov S.P., Petrovskaya L.E., Lukashev E.P., Imasheva E.S., Dioumaev A.K., Wang J.M., Sychev S.V., Dolgikh D.A., Rubin A.B., Kirpichnikov M.P., Lanyi J.K.
Biochemistry,
2012
6.

Gushchin I., Chervakov P., Kuzmichev P., Popov A.N., Round E., Borshchevskiy V., Ishchenko A., Petrovskaya L., Chupin V., Dolgikh D.A., Arseniev A.S., Kirpichnikov M., Gordeliy V.
Proceedings of the National Academy of Sciences of the United States of America,
2013
7.

Balashov S.P., Petrovskaya L.E., Imasheva E.S., Lukashev E.P., Dioumaev A.K., Wang J.M., Sychev S.V., Dolgikh D.A., Rubin A.B., Kirpichnikov M.P., Lanyi J.K.
Journal of Biological Chemistry,
2013
8.

Dioumaev A.K., Petrovskaya L.E., Wang J.M., Balashov S.P., Dolgikh D.A., Kirpichnikov M.P., Lanyi J.K.
Journal of Physical Chemistry B,
2013
9.

Petrovskaya L.E., Balashov S.P., Lukashev E.P., Imasheva E.S., Gushchin I.Y., Dioumaev A.K., Rubin A.B., Dolgikh D.A., Gordeliy V.I., Lanyi J.K., Kirpichnikov M.P.
Biochemistry (Moscow),
2015
10.
10.1016/j.mencom.2018.07.022_bib0050
Khodonov
Bioorg. Khim.,
1996
11.

Nakanishi K., Balogh-Nair V., Arnaboldi M., Tsujimoto K., Honig B.
Journal of the American Chemical Society,
1980
12.

Motto M.G., Sheves M., Tsujimoto K., Balogh-Nair V., Nakanishi K.
Journal of the American Chemical Society,
1980
13.

Muthyala R., Watanabe D., Asato A.E., Liu R.S.
Photochemistry and Photobiology,
2001
14.

López S., Rodríguez V., Montenegro J., Saá C., Alvarez R., Silva López C., de Lera A.R., Simón R., Lazarova T., Padrós E.
ChemBioChem,
2005
15.

Klapper S.D., Swiersy A., Bamberg E., Busskamp V.
Frontiers in Systems Neuroscience,
2016
16.

Wang P., Chang A.Y., Novosad V., Chupin V.V., Schaller R.D., Rozhkova E.A.
ACS Nano,
2017
17.

Ganapathy S., Venselaar H., Chen Q., de Groot H.J., Hellingwerf K.J., de Grip W.J.
Journal of the American Chemical Society,
2017