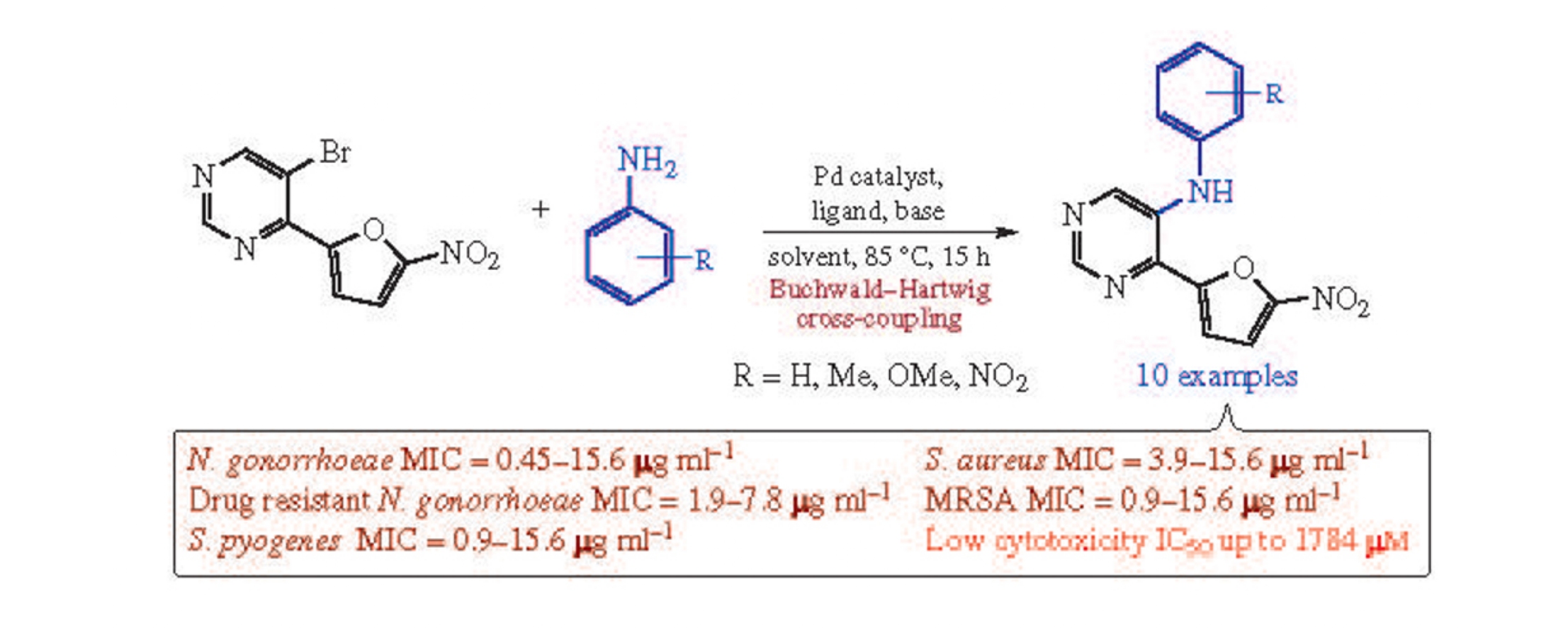
Abstract
A facile synthetic approach to 5-arylamino-4-(5-nitrofuran- 2-yl)pyrimidines by the Buchwald–Hartwig cross-coupling with various anilines has been developed. All synthesized compounds demonstrated a significant level of in vitro antibacterial activity against Neisseria gonorrhoeae, Streptococcus pyogene and Staphylococcus aureus, including their drug-resistant strains, which is much higher than that of the commercial drug Spectinomycin.
References
1.

Walsh C.T., Wencewicz T.A.
Journal of Antibiotics,
2013
2.

Silver L.L.
Clinical Microbiology Reviews,
2011
3.

McDougal L.K., Steward C.D., Killgore G.E., Chaitram J.M., McAllister S.K., Tenover F.C.
Journal of Clinical Microbiology,
2003
4.

DeLeo F.R., Chambers H.F.
Journal of Clinical Investigation,
2009
5.

Picconi P., Prabaharan P., Auer J.L., Sandiford S., Cascio F., Chowdhury M., Hind C., Wand M.E., Sutton J.M., Rahman K.M.
Bioorganic and Medicinal Chemistry,
2017
6.
Rise in Antibiotic-Resistant Gonorrhea, WHO Report Reveals, WHO Media Centre, 2017, http://www.who.int/mediacentre/news/releases/2017/Antibiotic-resistant-gonorrhoea/en.
7.

Criss A.K., Tang C.
Pathogens and Disease,
2017
8.

Dann O., Möller E.F.
Chemische Berichte,
1947
9.

Chamberlain R.E.
Journal of Antimicrobial Chemotherapy,
1976
10.

Viodé C., Bettache N., Cenas N., Krauth-Siegel R.L., Chauvière G., Bakalara N., Périé J.
Biochemical Pharmacology,
1999
11.

Purohit V., Basu A.K.
Chemical Research in Toxicology,
2000
12.
![Genotoxicity of 2-nitro-7-methoxy-naphtho[2,1-b]furan (R7000): A case study with some considerations on nitrofurantoin and nifuroxazide](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Hofnung M., Quillardet P., Michel V., Touati E.
Research in Microbiology,
2002
13.

Ginsberg A.M., Laurenzi M.W., Rouse D.J., Whitney K.D., Spigelman M.K.
Antimicrobial Agents and Chemotherapy,
2009
14.

Matsumoto M., Hashizume H., Tomishige T., Kawasaki M., Tsubouchi H., Sasaki H., Shimokawa Y., Komatsu M.
PLoS Medicine,
2006
15.

Kim P., Zhang L., Manjunatha U.H., Singh R., Patel S., Jiricek J., Keller T.H., Boshoff H.I., Barry C.E., Dowd C.S.
Journal of Medicinal Chemistry,
2009
16.
J.C. Howard, Patent US 3121083, 1964 (Chem. Abstr. 1964, 60, 12027).
17.
10.1016/j.mencom.2018.07.017_bib0085
Gronowitz
Acta Pharm. Suec,
1968
18.
H. Berger, R. Gall, H. Merdes, K. Stach, W. Sauer and W. Voemel, Patent DE 1909346, 1970 (Chem. Abstr. 1970, 73, 98974).
19.
H. Berger, R. Gall, H. Merdes, K. Stach, W. Voemel and W. Sauer, Patent ZA 6904822, 1972.
20.
H. Berger, R. Gall, H. Merdes, K. Stach, W. Voemel and W. Sauer, Patent US 3704301 A, 1972.
21.
K. Gutsche and F. W. Kohlmann, Patent DE 2240242, 1974.
22.

Verbitskiy E.V., Baskakova S.A., Gerasimova N.A., Evstigneeva N.P., Zil'berberg N.V., Kungurov N.V., Kravchenko M.A., Skornyakov S.N., Pervova M.G., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Bioorganic and Medicinal Chemistry Letters,
2017
23.

Verbitskiy E.V., Cheprakova E.M., Slepukhin P.A., Kodess M.I., Ezhikova M.A., Pervova M.G., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Tetrahedron,
2012
24.

Verbitskiy E.V., Rusinov G.L., Charushin V.N., Chupakhin O.N., Cheprakova E.M., Slepukhin P.A., Pervova M.G., Ezhikova M.A., Kodess M.I.
European Journal of Organic Chemistry,
2012
25.

Chupakhin O.N., Charushin V.N.
Tetrahedron Letters,
2016
26.

Ruiz-Castillo P., Buchwald S.L.
Chemical Reviews,
2016
27.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
28.

Mosmann T.
Journal of Immunological Methods,
1983
29.

Sun H., Tawa G., Wallqvist A.
Drug Discovery Today,
2012