Abstract
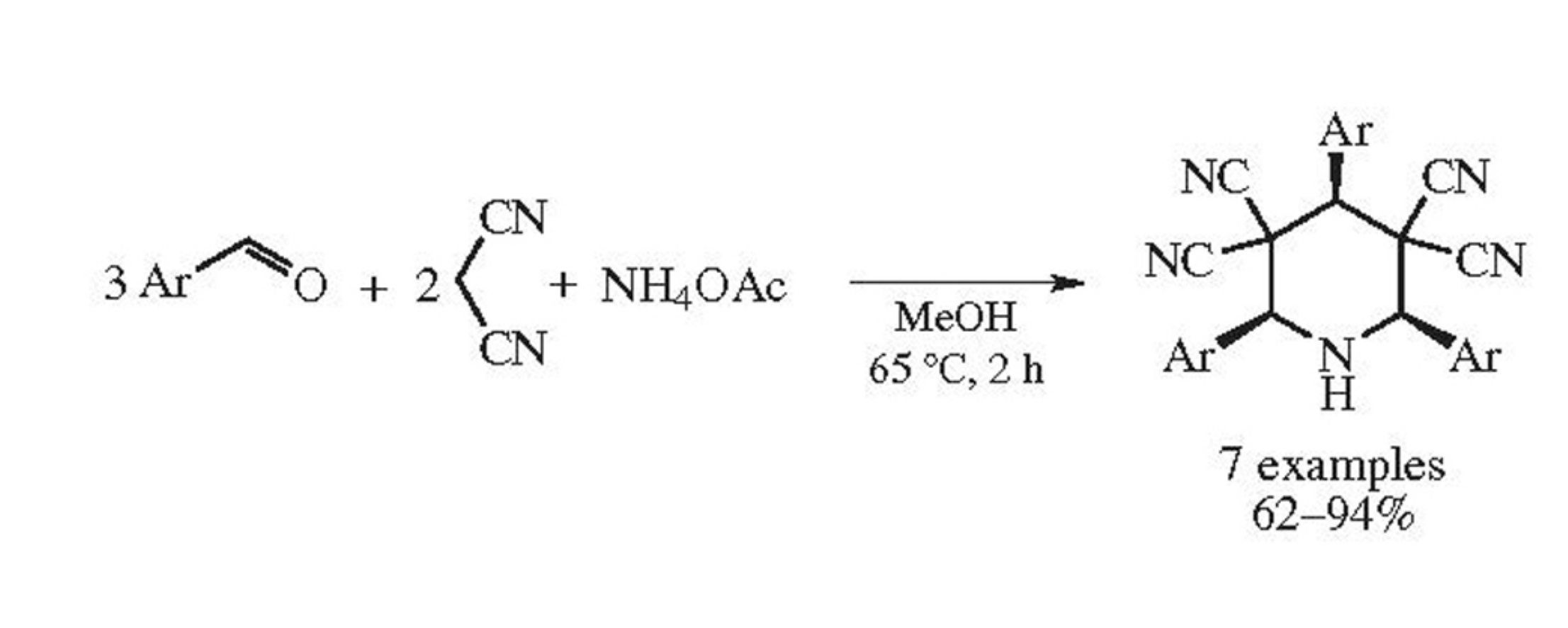
The Knövenagel–Michael–Mannich cascade reaction of aromatic aldehyde (3 equiv.), malononitrile (2 equiv.) and ammonium acetate or aqueous ammonia provides convenient stereoselective access to cis,cis-2,4,6-triaryl-3,3,5,5-tetracyanopiperidines in 62–94% yields. Six new bonds form as a result of the domino process, ammonium acetate serving as a nitrogen source.
References
2.

Catalysis of Cascade and Multicomponent Reactions of Carbonyl Compounds and CH Acids by Electricity
Elinson M.N., Vereshchagin A.N., Ryzhkov F.V.
Chemical Record,
2016
3.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
4.

Vereshchagin A.N.
Russian Chemical Bulletin,
2017
5.

Slobbe P., Ruijter E., Orru R.V.
MedChemComm,
2012
6.

Dömling A.
Chemical Reviews,
2005
7.
D. A. Elbein and R. Molyneux, in Alkaloids: Chemical and Biological Perspectives, ed. S. W. Palletier, Wiley, New York, 1987, vol. 5, pp. 1-54.
8.

Weis R., Schweiger K., Faist J., Rajkovic E., Kungl A.J., Fabian W.M., Schunack W., Seebacher W.
Bioorganic and Medicinal Chemistry,
2008
9.

Wang W., Hu Y.
Medicinal Research Reviews,
2011
10.

Liu H., Zhou Z., Sun Q., Li Y., Li Y., Liu J., Yan P., Wang D., Wang C.
ACS Combinatorial Science,
2012
11.

Elinson M.N., Feducovich S.K., Dorofeev A.S., Vereshchagin A.N., Nikishin G.I.
Russian Chemical Bulletin,
2003
12.
Vereshchagin A.N., Elinson M.N., Stepanov N.O., Nikishin G.I.
Mendeleev Communications,
2009
13.

Vereshchagin A.N., Elinson M.N., Egorov M.P.
RSC Advances,
2015
14.

Elinson M.N., Vereshchagin A.N., Feducovich S.K., Zaimovskaya T.A., Starikova Z.A., Belyakov P.A., Nikishin G.I.
Tetrahedron Letters,
2007
15.

Elinson M.N., Feducovich S.K., Zaimovskaya T.A., Dorofeev A.S., Vereshchagin A.N., Nikishin G.I.
Russian Chemical Bulletin,
2003
16.

Vereshchagin A.N., Elinson M.N., Dorofeeva E.O., Stepanov N.O., Zaimovskaya T.A., Nikishin G.I.
Tetrahedron,
2013
17.

Vereshchagin A.N., Elinson M.N., Ryzhkov F.V., Nasybullin R.F., Bobrovsky S.I., Goloveshkin A.S., Egorov M.P.
Comptes Rendus Chimie,
2015
18.
Elinson M.N., Ryzhkov F.V., Vereshchagin A.N., Korshunov A.D., Novikov R.A., Egorov M.P.
Mendeleev Communications,
2017
19.
![PASE Pseudo-Four-Component Synthesis and Docking Studies of New 5-C-Substituted 2,4-Diamino-5H-Chromeno[2,3-b]pyridine-3-Carbonitriles](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Vereshchagin A.N., Elinson M.N., Anisina Y.E., Ryzhkov F.V., Novikov R.A., Egorov M.P.
ChemistrySelect,
2017
20.
![One-Pot ‘On-solvent’ Multicomponent Protocol for the Synthesis of Medicinally Relevant 4H-Pyrano[3,2-c]quinoline Scaffold](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Vereshchagin A.N., Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Bobrovsky S.I., Bushmarinov I.S., Egorov M.P.
Helvetica Chimica Acta,
2015
21.
Elinson M.N., Ryzhkov F.V., Vereshchagin A.N., Zaimovskaya T.A., Korolev V.A., Egorov M.P.
Mendeleev Communications,
2016
22.

Lemek T., Mayr H.
Journal of Organic Chemistry,
2003
23.

24.

Elinson M.N., Vereshchagin A.N., Stepanov N.O., Ilovaisky A.I., Vorontsov A.Y., Nikishin G.I.
Tetrahedron,
2009