Abstract
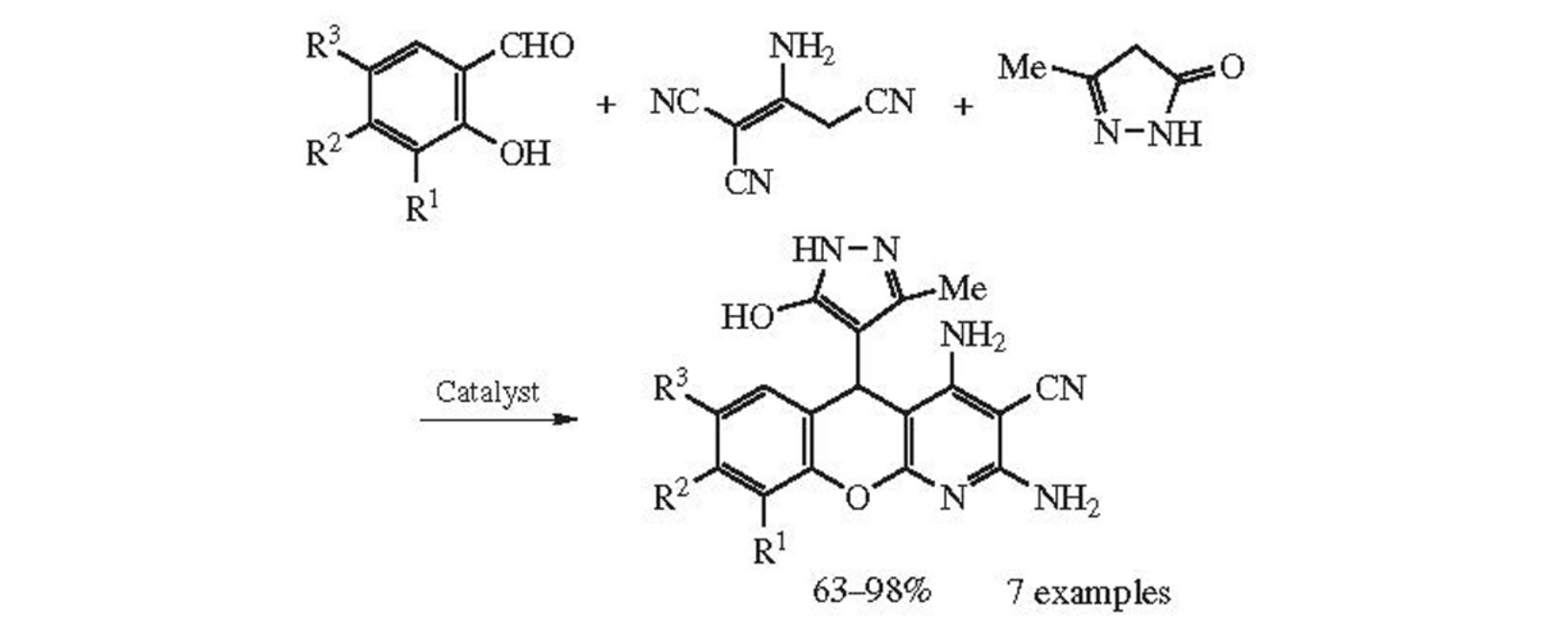
The new multicomponent reaction comprises the triethylamine catalyzed assembling of salicylaldehydes, 2-aminoprop- 1-ene-1,1,3-tricarbonitrile and 3-methyl-2-pyrazolin-5-one in small amount of propanol, which affords substituted 2,4-diamino-5-(5-hydroxy-3-methyl-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitriles in 63–98% yields. This process is in agreement with PASE principles and opens the way to new chromeno[2,3-b]pyridine scaffold containing 3-methyl-2-pyrazolin-5-one fragment, which is promising for various biomedical applications.
References
1.

Clarke P.A., Santos S., Martin W.H.
Green Chemistry,
2007
2.

Cioc R.C., Ruijter E., Orru R.V.
Green Chemistry,
2014
3.

Kolokythas G., Pouli N., Marakos P., Pratsinis H., Kletsas D.
European Journal of Medicinal Chemistry,
2006
4.

Azuine M.A., Tokuda H., Takayasu J., Enjyo F., Mukainaka T., Konoshima T., Nishino H., Kapadia G.J.
Pharmacological Research,
2004
5.
Y. Maruyama, K. Goto and M. Terasawa, Ger. Offen. DE 3010751, 1981.
6.

Anderson D.R., Hegde S., Reinhard E., Gomez L., Vernier W.F., Lee L., Liu S., Sambandam A., Snider P.A., Masih L.
Bioorganic and Medicinal Chemistry Letters,
2005
7.
J. Elguero, P. Goya, N. Jagerovic and A. M. S. Silva, in Targets in Heterocyclic Systems. Chemistry and Properties, eds. O. A. Attanasi and D. Spinelli, Italian Society of Chemistry, Rome, 2002, vol. 6, pp. 53-99.
8.
Elinson M.N., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2012
9.

Elinson M.N., Ilovaisky A.I., Merkulova V.M., Belyakov P.A., Barba F., Batanero B.
Tetrahedron,
2012
10.

Elinson M.N., Ilovaisky A.I., Merkulova V.M., Barba F., Batanero B.
Tetrahedron,
2013
11.

Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Egorov M.P.
Comptes Rendus Chimie,
2013
12.
![Pot, atom and step-economic (PASE) synthesis of medicinally relevant spiro[oxindole-3,4′-pyrano[4,3-b]pyran] scaffold](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Elinson M.N., Ryzhkov F.V., Korolev V.A., Egorov M.P.
Heterocyclic Communications,
2016
13.
Elinson M.N., Ryzhkov F.V., Vereshchagin A.N., Korshunov A.D., Novikov R.A., Egorov M.P.
Mendeleev Communications,
2017
14.
Vereshchagin A.N., Elinson M.N., Anisina Y.E., Ryzhkov F.V., Goloveshkin A.S., Bushmarinov I.S., Zlotin S.G., Egorov M.P.
Mendeleev Communications,
2015
15.
![Synthesis, structural, spectroscopic and docking studies of new 5C-substituted 2,4-diamino-5H-chromeno[2,3-b]pyridine-3-carbonitriles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Vereshchagin A.N., Elinson M.N., Anisina Y.E., Ryzhkov F.V., Goloveshkin A.S., Novikov R.A., Egorov M.P.
Journal of Molecular Structure,
2017
16.
17.

18.
Elinson M.N., Medvedev M.G., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2013
19.

Butler R.N., Coyne A.G.
Organic and Biomolecular Chemistry,
2016
20.
Demchuk D.V., Elinson M.N., Nikishin G.I.
Mendeleev Communications,
2011
21.

22.

Sarkar A., Santra S., Kundu S.K., Hajra A., Zyryanov G.V., Chupakhin O.N., Charushin V.N., Majee A.
Green Chemistry,
2016
23.
Elinson M.N., Ryzhkov F.V., Nasybullin R.F., Zaimovskaya T.A., Egorov M.P.
Mendeleev Communications,
2014
24.
![One-Pot ‘On-solvent’ Multicomponent Protocol for the Synthesis of Medicinally Relevant 4H-Pyrano[3,2-c]quinoline Scaffold](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Vereshchagin A.N., Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Bobrovsky S.I., Bushmarinov I.S., Egorov M.P.
Helvetica Chimica Acta,
2015
25.
![Chemical and electrocatalytic cascade cyclization of salicylaldehyde with three molecules of malononitrile: ‘one-pot’ simple and efficient way to the chromeno[2,3-b]pyridine scaffold](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Elinson M.N., Gorbunov S.V., Vereshchagin A.N., Nasybullin R.F., Goloveshkin A.S., Bushmarinov I.S., Egorov M.P.
Tetrahedron,
2014
26.
Elinson M.N., Ryzhkov F.V., Vereshchagin A.N., Zaimovskaya T.A., Korolev V.A., Egorov M.P.
Mendeleev Communications,
2016