Abstract
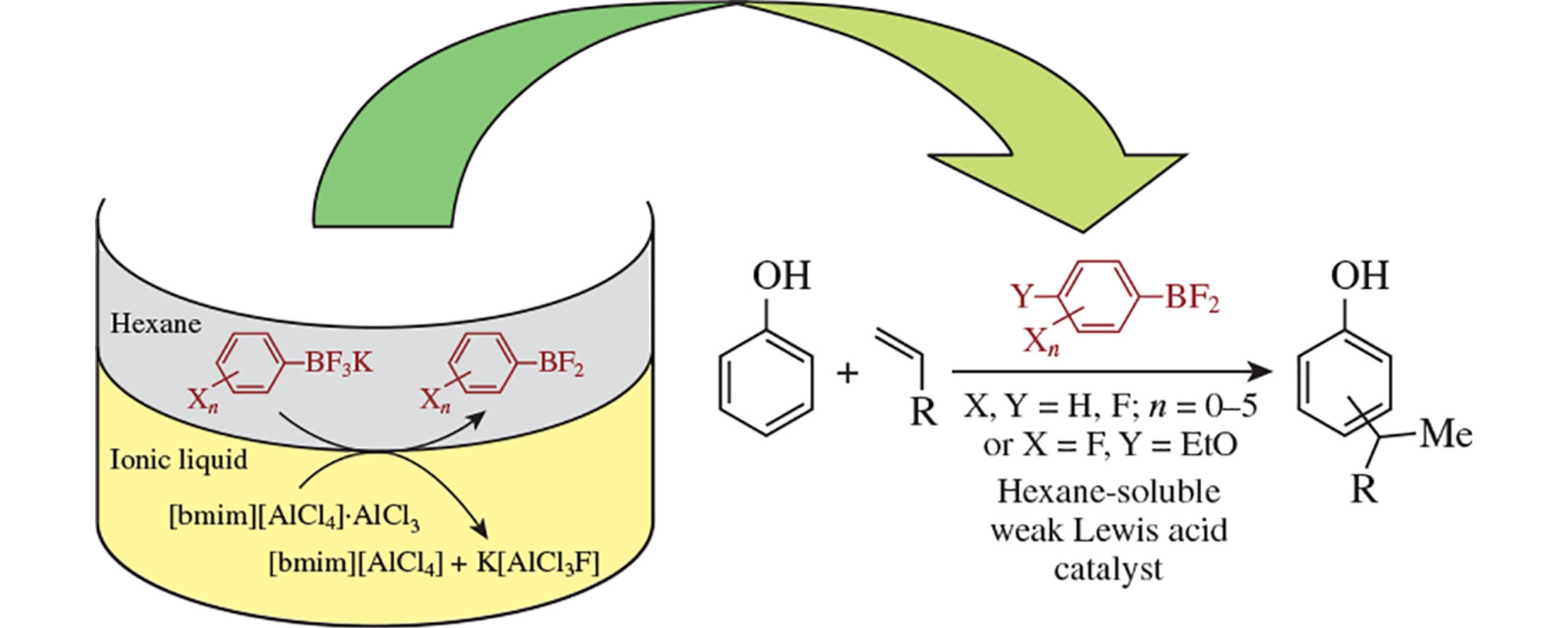
A new approach for preparation of aromatic and fluoroaromatic difluoroboranes via the interaction between corresponding aryltrifluoroborates and ionic liquids containing tetrachloroaluminate-anion and aluminum chloride has been developed. Catalytic properties of obtained aryldifluoroboranes have been investigated in model reactions of phenols alkylation. The dependence of catalytic properties on both the nature of solvent used and the type of substituents in the aromatic ring of difluoroborane has been established.
References
1.

Corma A., García H.
Chemical Reviews,
2003
2.

Boyarskiy V.P., Ryabukhin D.S., Bokach N.A., Vasilyev A.V.
Chemical Reviews,
2016
3.
4.

Chambers R.D., Chivers T.
Journal of the Chemical Society (Resumed),
1965
5.

Chambers R.D., Chivers T., Pyke D.A.
Journal of the Chemical Society (Resumed),
1965
6.

Bardin V.V., Frohn H.
Main Group Metal Chemistry,
2002
7.

Chase P.A., Henderson L.D., Piers W.E., Parvez M., Clegg W., Elsegood M.R.
Organometallics,
2005
8.

Kochnev A.I., Oleynik I.I., Oleynik I.V., Ivanchev S.S., Tolstikov G.A.
Russian Chemical Bulletin,
2007
9.

Cai X., Cui S., Qu L., Yuan D., Lu B., Cai Q.
Catalysis Communications,
2008
10.

Gutmann V.
Coordination Chemistry Reviews,
1976
11.

Sivaev I.B., Bregadze V.I.
Coordination Chemistry Reviews,
2014