Abstract
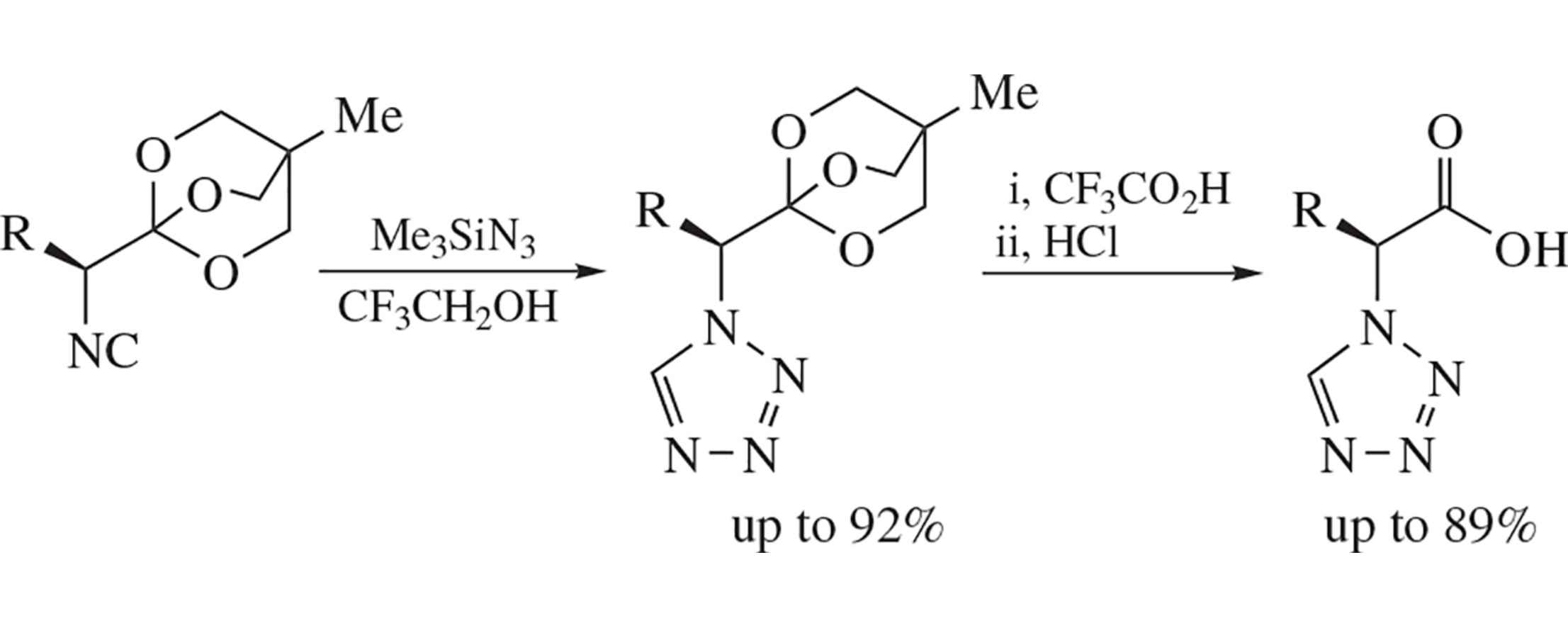
Optically active α-(tetrazol-1-yl)-substituted carboxylic acid OBO-esters were synthesized from the corresponding a-isocyano OBO-esters and trimethylsilyl azide in up to 92% yield. Subsequent acidic hydrolysis proceeds without epimerization and makes it possible to prepare enantiomerically pure α-(tetrazol-1-yl)-substituted carboxylic acids in up to 89% yield.
References
1.

Koldobskii G.I., Ostrovskii V.A.
Russian Chemical Reviews,
1994
2.

Gaponik P.N., Voitekhovich S.V., Ivashkevich O.A.
Russian Chemical Reviews,
2006
3.

Antipin I.S., Kazymova M.A., Kuznetsov M.A., Vasilyev A.V., Ishchenko M.A., Kiryushkin A.A., Kuznetsova L.M., Makarenko S.V., Ostrovskii V.A., Petrov M.L., Solod O.V., Trishin Y.G., Yakovlev I.P., Nenaidenko V.G., Beloglazkina E.K., et. al.
Russian Journal of Organic Chemistry,
2017
4.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
5.
Kormanov A.V., Shkineva T.K., Dalinger I.L.
Mendeleev Communications,
2017
6.

Myznikov L.V., Hrabalek A., Koldobskii G.I.
Chemistry of Heterocyclic Compounds,
2007
7.

Bhandari S., Mahon M.F., Molloy K.C., Palmer J.S., Sayers S.F.
Journal of the Chemical Society Dalton Transactions,
2000
8.

Brubaker C.H.
Journal of the American Chemical Society,
1960
9.

Daugherty N.A., Brubaker C.H.
Journal of Inorganic and Nuclear Chemistry,
1961
10.

Daugherty N.A., Brubaker C.H.
Journal of the American Chemical Society,
1961
11.

Gilbert G.L., Brubaker C.H.
Inorganic Chemistry,
1963
12.

Franke P.L., Groeneveld W.L.
Transition Metal Chemistry,
1981
13.

Degtyarik M.M., Gaponik P.N., Naumenko V.N., Lesnikovich A.I., Nikanovich M.V.
Spectrochimica Acta Part A Molecular Spectroscopy,
1987
14.

Poturovic S., Lu D., Heeg M.J., Winter C.H.
Polyhedron,
2008
15.

Garber L.L., Brubaker C.H.
Journal of the American Chemical Society,
1966
16.
2 and [Cu(C3tz)6](BF4)2](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Soliman A.A., Khattab M.M., Linert W.
Journal of Coordination Chemistry,
2005
17.
2](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
van Koningsbruggen P.J., Garcia Y., Bravic G., Chasseau D., Kahn O.
Inorganica Chimica Acta,
2001
18.

Popova E.A., Trifonov R.E.
Russian Chemical Reviews,
2015
19.

Habibi D., Rahmani P., Ahmadi F., Bokharaei H., Kaboudvand Z.
Letters in Organic Chemistry,
2014
20.

Maleki A., Sarvary A.
RSC Advances,
2015
21.

SMITH P.A., KALENDA N.W.
Journal of Organic Chemistry,
1958
22.

Zhdanko A.G., Nenajdenko V.G.
Journal of Organic Chemistry,
2008
23.

Konovalov A.I., Antipin I.S., Burilov V.A., Madzhidov T.I., Kurbangalieva A.R., Nemtarev A.V., Solovieva S.E., Stoikov I.I., Mamedov V.A., Zakharova L.Y., Gavrilova E.L., Sinyashin O.G., Balova I.A., Vasilyev A.V., Zenkevich I.G., et. al.
Russian Journal of Organic Chemistry,
2018
24.

Corey E.J., Raju N.
Tetrahedron Letters,
1983
25.

Gulevich A.V., Shpilevaya I.V., Nenajdenko V.G.
European Journal of Organic Chemistry,
2009
26.

Zhdanko A.G., Gulevich A.V., Nenajdenko V.G.
Tetrahedron,
2009
27.

Shmatova O.I., Nenajdenko V.G.
Journal of Organic Chemistry,
2013
28.
![Synthesis of 1-substituted tetrazoles via the acid-catalyzed [3+2] cycloaddition between isocyanides and trimethylsilyl azide](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Jin T., Kamijo S., Yamamoto Y.
Tetrahedron Letters,
2004
29.

Dömling A., Wang Y., Patil P.
Synthesis,
2016