Abstract
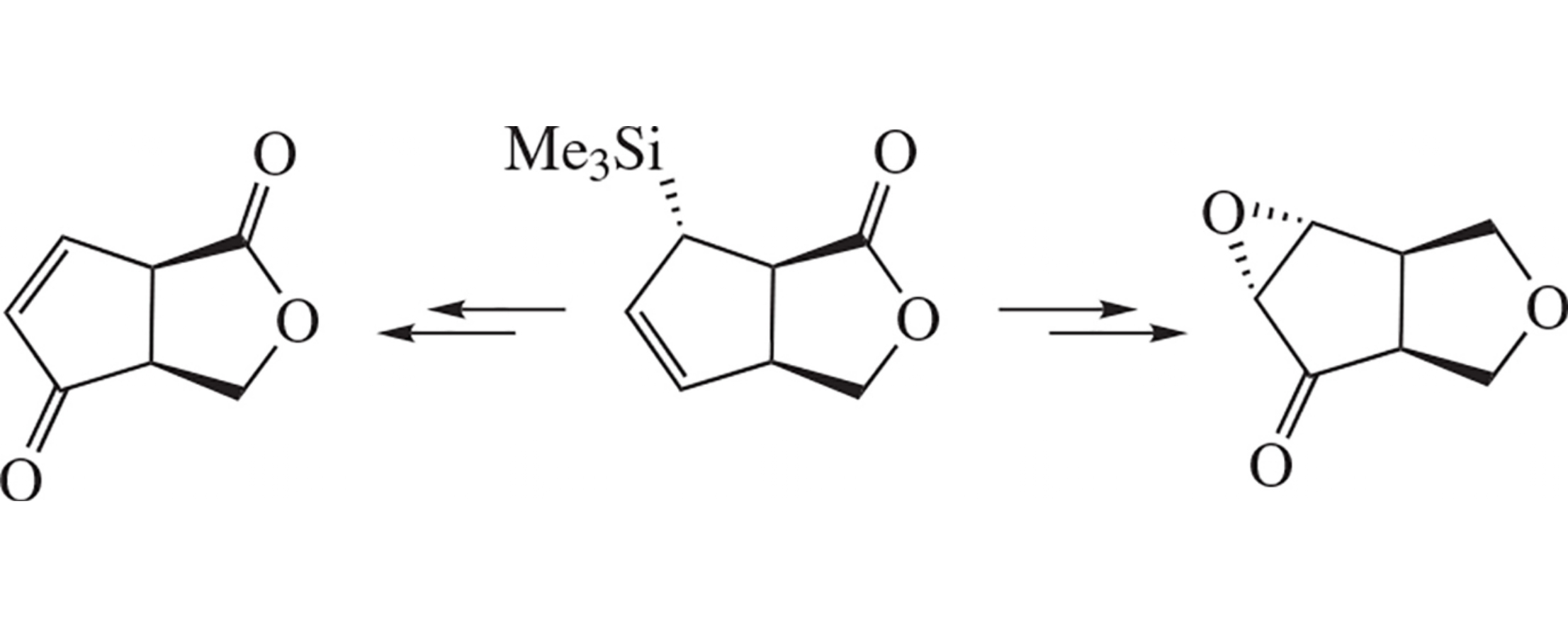
Synthetically useful chiral building blocks (3aS,6aR)-1,3,3a,6atetrahydro-4H-cyclopenta[c]furan-4-one and (3aS,6aR)-3a,6a-dihydro-1H-cyclopenta[c]furan-1,4(3H)-dione have been synthesized via a key allylsilane–allyl alcohol fragmentation pathway using (3aS,6S,6aR)-6-(trimethylsilyl)-3,3a,6,6a-tetrahydro-1H-cyclopenta[c]furan-1-one as an illustrative example.
References
1.

Scarborough R.M., Toder B.H., Smith A.B.
Journal of the American Chemical Society,
1980
2.
![A direct organocobalt mediated synthesis of substituted 3-oxabicyclo[3.3.0]oct-7-en-6-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Billington D.C.
Tetrahedron Letters,
1983
3.

González M.A.
Tetrahedron,
2008
4.

Evans D.A., Dow R.L., Shih T.L., Takacs J.M., Zahler R.
Journal of the American Chemical Society,
1990
5.

Hanessian S., Cooke N.G., DeHoff B., Sakito Y.
Journal of the American Chemical Society,
1990
6.

Ireland R.E., Anderson R.C., Badoud R., Fitzsimmons B.J., McGarvey G.J., Thaisrivongs S., Wilcox C.S.
Journal of the American Chemical Society,
1983
7.

Bartlett P.A., Meadows J.D., Ottow E.
Journal of the American Chemical Society,
1984
8.
10.1016/j.mencom.2018.07.006_bib0040
Li
Beilstein J. Org. Chem,
2008
9.

Murai A.
Pure and Applied Chemistry,
1989
10.
Gimazetdinov A.M., Gimazetdinova T.V., Miftakhov M.S.
Mendeleev Communications,
2010
11.

Straus D.S., Glass C.K.
Medicinal Research Reviews,
2001
12.

Campian M., Putala M., Sebesta R.
Current Organic Chemistry,
2014
13.
10.1016/j.mencom.2018.07.006_bib0065
Gimazetdinov
Nat. Prod. Commun,
2013
14.
![A simple and efficient synthesis of enantiomeric (3aRS,4RS,6aSR)-4-hydroxy-3,3a,4,6a-tetrahydro-1H-cyclopenta[c]furan-1-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gimazetdinov A.M., Gataullin S.S., Bushmarinov I.S., Miftakhov M.S.
Tetrahedron,
2012
15.

Gimazetdinov A.M., Al'mukhametov A.Z., Spirikhin L.V., Miftakhov M.S.
Tetrahedron Letters,
2017
16.

Gimazetdinov A.M., Almukhametov V.Z., Loza V.V., Miftakhov M.S.
Russian Journal of Organic Chemistry,
2016