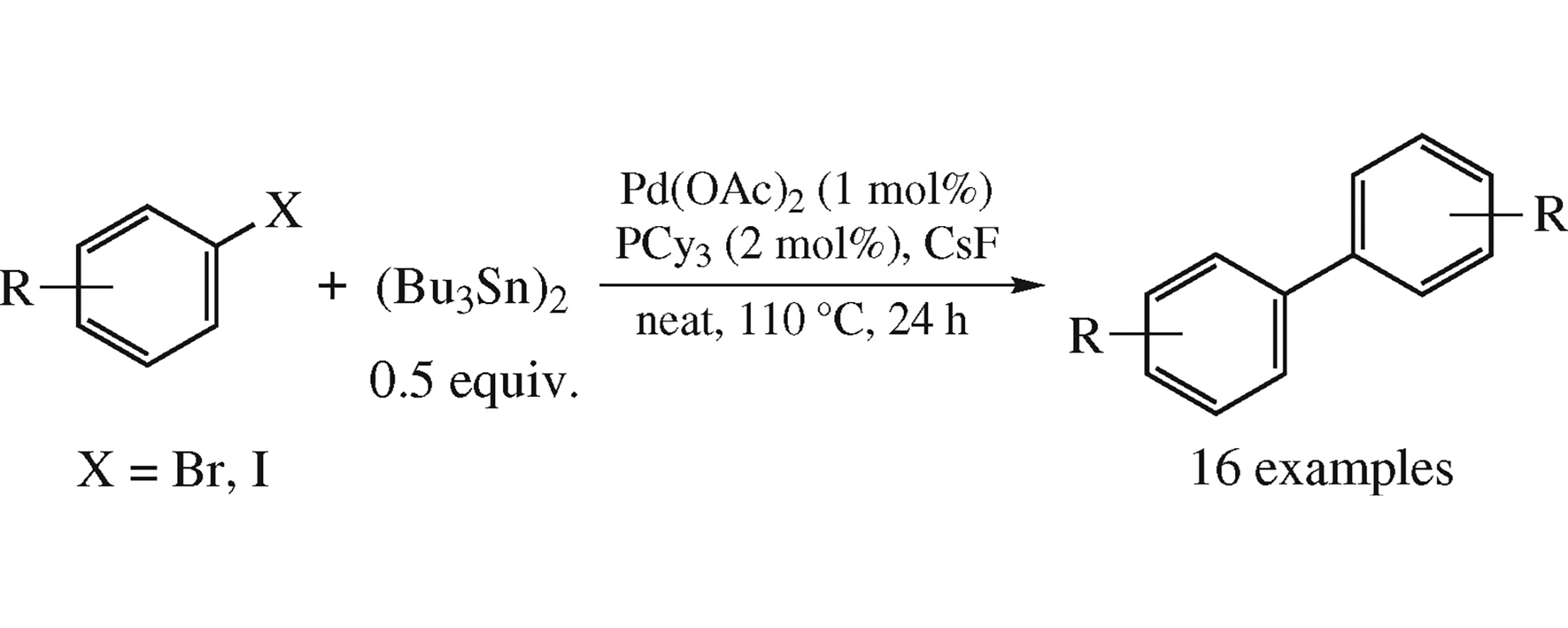
Abstract
A new highly efficient solvent-free method for aryl bromide (iodide) homocoupling comprising the use of Pd(OAc)2/PCy3 system in the presence of CsF is suitable for substrates bearing functional groups not tolerant to lithium-, magnesium-, zincorganic reagents and strong bases.
References
1.

Huang Y., Liu L., Feng W.
ChemistrySelect,
2016
2.

Zhu S.S., Swager T.M.
Advanced Materials,
1996
3.
Klimovich I.V., Prudnov F.A., Inasaridze L.N., Kuznetsov I.E., Peregudov A.S., Troshin P.A.
Mendeleev Communications,
2017
4.

Mei X., Wolf C.
Journal of the American Chemical Society,
2006
5.

Buter J., Heijnen D., Vila C., Hornillos V., Otten E., Giannerini M., Minnaard A.J., Feringa B.L.
Angewandte Chemie - International Edition,
2016
6.

Bringmann G., Gulder T., Gulder T.A., Breuning M.
Chemical Reviews,
2010
7.

8.

Berthod M., Mignani G., Woodward G., Lemaire M.
Chemical Reviews,
2005
9.
10.

Campeau L., Fagnou K.
Chemical Society Reviews,
2007
11.
10.1016/j.mencom.2018.05.032_bib0045
Palladium in Heterocyclic Chemistry,
2006
12.
![Pentamidine analogs as inhibitors of [3H]MK-801 and [3H]ifenprodil binding to rat brain NMDA receptors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Berger M.L., Maciejewska D., Vanden Eynde J.J., Mottamal M., Żabiński J., Kaźmierczak P., Rezler M., Jarak I., Piantanida I., Karminski-Zamola G., Mayence A., Rebernik P., Kumar A., Ismail M.A., Boykin D.W., et. al.
Bioorganic and Medicinal Chemistry,
2015
13.

Yamaguchi Y., Nishizono N., Kobayashi D., Yoshimura T., Wada K., Oda K.
Bioorganic and Medicinal Chemistry Letters,
2017
14.

He Q., Li T., Yan C., Liu Y., Wang J., Wang M., Lin Y., Zhan X.
Dyes and Pigments,
2016
15.

Henssler J.T., Matzger A.J.
Journal of Organic Chemistry,
2012
16.

Bruce J.I., Chambron J., Kölle P., Sauvage J.
Journal of the Chemical Society Perkin Transactions 1,
2002
17.

Mendiola J., Castellote I., Alvarez-Builla J., Fernández-Gadea J., Gómez A., Vaquero J.J.
Journal of Organic Chemistry,
2006
18.

Morgan B.J., Xie X., Phuan P., Kozlowski M.C.
Journal of Organic Chemistry,
2007
19.

Khanasa T., Prachumrak N., Rattanawan R., Jungsuttiwong S., Keawin T., Sudyoadsuk T., Tuntulani T., Promarak V.
Journal of Organic Chemistry,
2013
20.

Hancock J.M., Gifford A.P., Tonzola C.J., Jenekhe S.A.
Journal of Physical Chemistry C,
2007
21.

Tonzola C.J., Alam M.M., Jenekhe S.A.
Macromolecules,
2005
22.

Polander L.E., Romanov A.S., Barlow S., Hwang D.K., Kippelen B., Timofeeva T.V., Marder S.R.
Organic Letters,
2012
23.

Keawin T., Sooksai C., Prachumrak N., Kaewpuang T., Muenmart D., Namuangruk S., Jungsuttiwong S., Sudyoadsuk T., Promarak V.
RSC Advances,
2015
24.

Bocknack B.M., Wang L., Hughes F.W., Krische M.J.
Tetrahedron,
2005
25.

Khunchalee J., Tarsaeng R., Jungsuttiwong S., Keawin T., Sudyoadsuk T., Promarak V.
Tetrahedron Letters,
2012
26.

García-Cuadrado D., Cuadro A.M., Alvarez-Builla J., Sancho U., Castaño O., Vaquero J.J.
Organic Letters,
2006
27.
10.1016/j.mencom.2018.05.032_bib0060
Zhao
Chemistry Bulletin/Huaxue Tongbao,
2008
28.

Pan C., Liu M., Duan X.
Chinese Journal of Organic Chemistry,
2015
29.

Chesnokov G.A., Topchiy M.A., Dzhevakov P.B., Gribanov P.S., Tukov A.A., Khrustalev V.N., Asachenko A.F., Nechaev M.S.
Dalton Transactions,
2017
30.

Topchiy M.A., Dzhevakov P.B., Rubina M.S., Morozov O.S., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2016
31.

Dzhevakov P.B., Topchiy M.A., Zharkova D.A., Morozov O.S., Asachenko A.F., Nechaev M.S.
Advanced Synthesis and Catalysis,
2016
32.

Asachenko A.F., Sorochkina K.R., Dzhevakov P.B., Topchiy M.A., Nechaev M.S.
Advanced Synthesis and Catalysis,
2013
33.

Topchiy M.A., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2014
34.

Gribanov P.S., Golenko Y.D., Topchiy M.A., Minaeva L.I., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2018
35.

Bedford R.B., Cazin C.S., Hazelwood S.L.
Chemical Communications,
2002
36.

Okitsu T., Iwatsuka K., Wada A.
Chemical Communications,
2008
37.

Ariafard A., Yates B.F.
Journal of the American Chemical Society,
2009
38.

Shibata I., Kato H., Ishida T., Yasuda M., Baba A.
Angewandte Chemie - International Edition,
2004
39.

Mee S.P., Lee V., Baldwin J.E.
Angewandte Chemie - International Edition,
2004
40.

Lou S., Fu G.
Advanced Synthesis and Catalysis,
2010
41.

Littke A.F., Schwarz L., Fu G.C.
Journal of the American Chemical Society,
2002
42.

Martin R., Buchwald S.L.
Accounts of Chemical Research,
2008
43.

Melvin P.R., Nova A., Balcells D., Dai W., Hazari N., Hruszkewycz D.P., Shah H.P., Tudge M.T.
ACS Catalysis,
2015
44.

45.

Murahashi S., Yamamura M., Yanagisawa K., Mita N., Kondo K.
Journal of Organic Chemistry,
1979
46.

Tamao K., Sumitani K., Kumada M.
Journal of the American Chemical Society,
1972
47.

King A.O., Okukado N., Negishi E.
Journal of the Chemical Society Chemical Communications,
1977
48.

Türkmen H., Can R., Çetinkaya B.
Dalton Transactions,
2009
49.

Alacid E., Nájera C.
Journal of Organic Chemistry,
2008