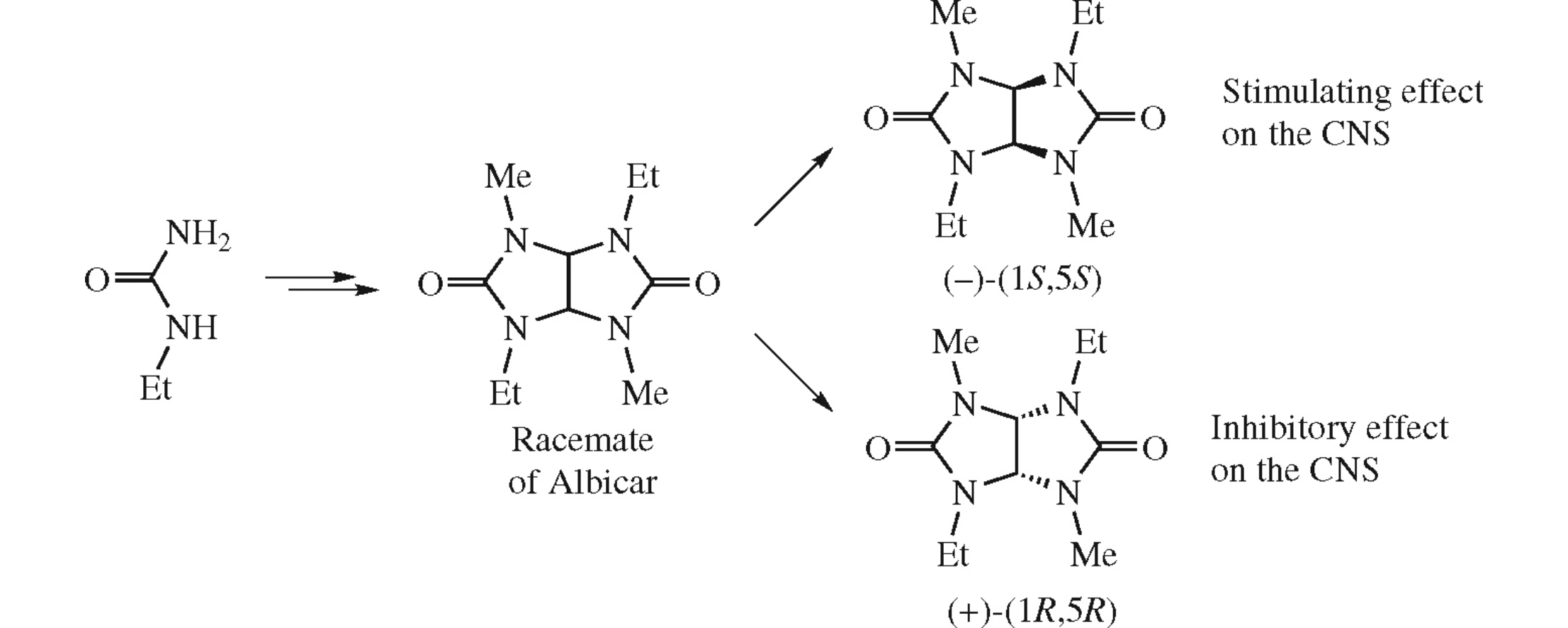
Abstract
Racemic Albicar (2,6-diethyl-4,8-dimethylglycoluril) has been synthesized and resolved into enantiomers on a preparative scale by chiral HPLC. A comparison of the pharmacological effects of these compounds has been performed for the first time. It has been demonstrated that the (−)-(1S,5S)-enantiomer exerts a stimulating effect on the central nervous system due to activation of the serotonergic system, since it potentiates the effects of 5-hydroxytryptophan (the serotonin precursor), while the antagonist of serotonin receptors blocks its activity; the (+)-(1R,5R)-enantiomer evinces an inhibitory effect.
References
1.

Kravchenko A.N., Baranov V.V., Gazieva G.A.
Russian Chemical Reviews,
2018
2.

Yazami R., Kusuzaka K., Hamwi A.
ECS Meeting Abstracts,
2008
3.
10.1016/j.mencom.2018.05.030_sbref0005c
Mashkovskii
2016
4.

Berlyand A.S., Lebedev O.V., Prokopov A.A.
Pharmaceutical Chemistry Journal,
2013
5.

Prokopov A.A., Berlyand A.S., Kazantseva O.N.
Pharmaceutical Chemistry Journal,
2003
6.

Berlyand A.S., Prokopov A.A.
Pharmaceutical Chemistry Journal,
2014
7.
![Neuroprotective activity of (+)-(S)-2-[(1S,5R)-(3,7-dioxo-2,4,6,8- tetraazabicyclo[3.3.0]oct-2-yl)]-4-methylthiobutanoic acid](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Kravchenko A.N., Baranov V.V., Anikina L.V., Vikharev Y.B., Bushmarinov I.S., Nelyubina Y.V.
Russian Journal of Bioorganic Chemistry,
2012
8.
Ryzhkina I.S., Kiseleva Y.V., Mishina O.A., Timosheva A.P., Sergeeva S.Y., Kravchenko A.N., Konovalov A.I.
Mendeleev Communications,
2013
9.
![Crystal structure, IR and 1H NMR spectra of tetranitratobis μ-(2,4,6,8-tetraethyl-2,4,6,8-tetraazabicyclo[3.3.0]octane-3,7-dione-O,O')] diethanolodicadmium](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Gazieva G.A., Golovanov D.G., Lozhkin P.V., Lysenko K.A., Kravchenko A.N.
Russian Journal of Inorganic Chemistry,
2007
10.

Ryzhkina I.S., Sergeeva S.Y., Masagutova E.M., Murtazina L.I., Mishina O.A., Timosheva A.P., Baranov V.V., Kravchenko A.N., Konovalov A.I.
Russian Chemical Bulletin,
2015
11.

Gazieva G.A., Lozhkin P.V., Baranov V.V., Kravchenko A.N., Makhova N.N.
Russian Chemical Bulletin,
2010
12.
Kostyanovsky R.G., Kadorkina G.K., Lyssenko K.A., Torbeev V.Y., Kravchenko A.N., Lebedev O.V., Grintselev-Knyazev G.V., Kostyanovsky V.R.
Mendeleev Communications,
2002
13.
10.1016/j.mencom.2018.05.030_sbref0010b
Pletnev
Russ. J. Bioorg. Chem.,
1993
14.

Chen J., Natte K., Spannenberg A., Neumann H., Beller M., Wu X.
Organic and Biomolecular Chemistry,
2014
15.
10.1016/j.mencom.2018.05.030_sbref0015b
Satyanarayana
Int. J. Chem. Tech. Res.,
2011
16.

Li Y., Coller J.K., Hutchinson M.R., Klein K., Zanger U.M., Stanley N.J., Abell A.D., Somogyi A.A.
Drug Metabolism and Disposition,
2013
17.

Muller G.W., Konnecke W.E., Smith A.M., Khetani V.D.
Organic Process Research and Development,
1999
18.

Yokoyama R., Matsumoto S., Nomura S., Higaki T., Yokoyama T., Kiyooka S.
Tetrahedron,
2009
19.

Robin S., Zhu J., Galons H., Chuong P., Claude J.R., Tomas A., Viossat B.
Tetrahedron Asymmetry,
1995
20.
10.1016/j.mencom.2018.05.030_sbref0015g
Park
J. Int. Eng. Chem.,
2002
21.
![Three-dimensional structures and spectra of 2,6- and 2,8-diethyl-2,4,6,8-tetraazabicyclo[3.3.0]octane-3,7-diones](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Shamuratov �.B., Batsanov A.S., Struchkov Y.T., Tsivadze A.Y., Tsintsadze M.G., Khmel'nitskii L.I., Simonov Y.A., Dvorkin A.A., Lebedev O.V., Markova T.B.
Chemistry of Heterocyclic Compounds,
1991
22.

Ivanov E.V., Batov D.V., Kravchenko A.N.
Journal of Chemical Thermodynamics,
2016
23.

Perals D., Griffin A.S., Bartomeus I., Sol D.
Animal Behaviour,
2017
24.

Zimcikova E., Simko J., Karesova I., Kremlacek J., Malakova J.
Seizure : the journal of the British Epilepsy Association,
2017
25.
10.1016/j.mencom.2018.05.030_sbref0025c
Chaves
Metab. Brain Dis.,
2017
26.
10.1016/j.mencom.2018.05.030_sbref0025d
Bossier
Thérapie,
1964
27.

Fernandez S.P., Nguyen M., Yow T.T., Chu C., Johnston G.A., Hanrahan J.R., Chebib M.
Neurochemical Research,
2009
28.
A. V. Val’dman and V. P. Poschivalov, Farmakologicheskaya regulyatsiya vnutrividovogo povedeniya (Pharmacological Regulation of Intraspecific Behaviour), Meditsina, Leningrad, 1984 (in Russian).
29.
https://amt.allergist.ru/mebicar_l.html.
30.

Martin P., Frances H., Simon P.
Journal of Pharmacological Methods,
1985
31.

Sperk G., Malcolm Stewart R., Campbell A., Baldessarini R.J.
Brain Research,
1978