Abstract
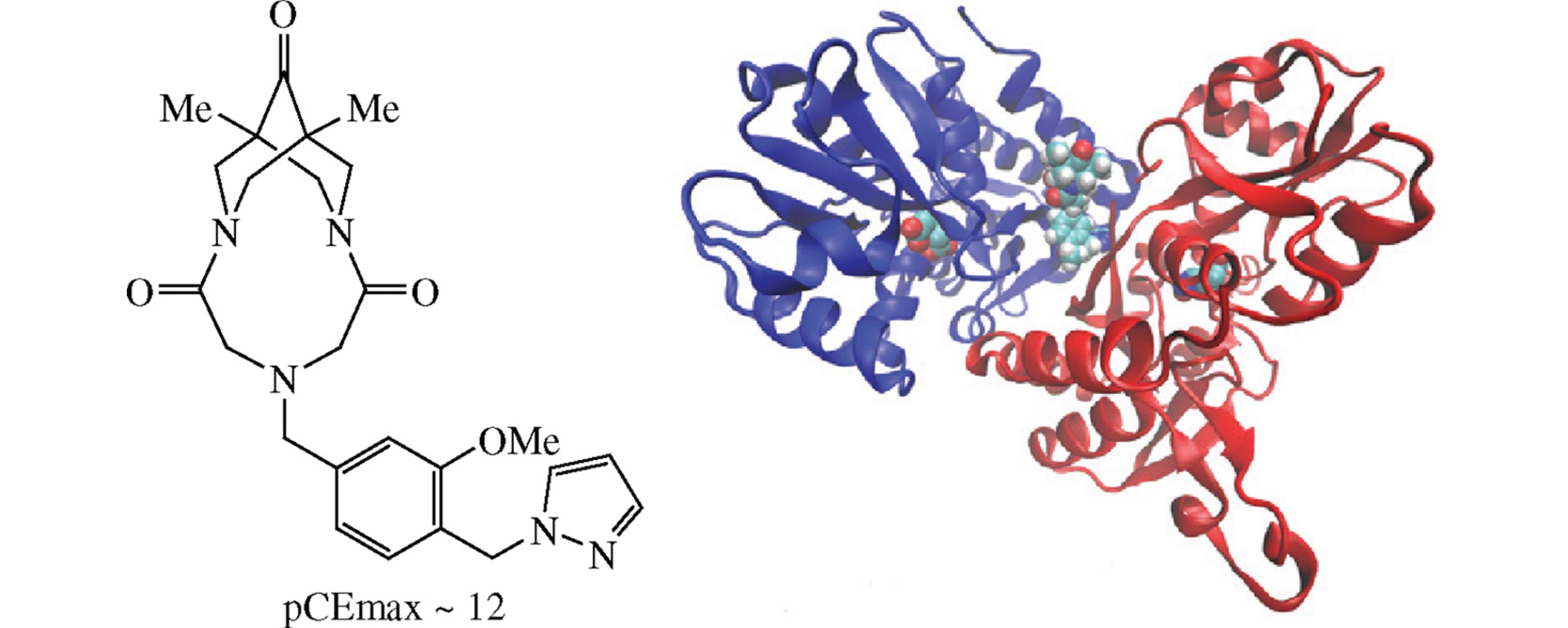
Molecular modeling, synthesis, and activity evaluation of a novel positive allosteric modulator of the AMPA receptor, 6-[4-methoxy-3-(1H-pyrazol-1-ylmethyl)benzyl]-1,11-dimethyl-3,6,9-triazatricyclo[7.3.1.13,11]tetradecane-4,8,12-trione, are presented. The main point of the design was to fill the interdomain cavity of GluA2 ligand binding domains based on the cyclothiazide binding mode. Electrophysiological studies showed high potentiation of the kainate-induced currents (pECmax∼12).
References
1.

Francotte P., de Tullio P., Fraikin P., Counerotte S., Goffin E., Pirotte B.
Recent Patents on CNS Drug Discovery,
2006
2.

Greger I.H., Esteban J.A.
Current Opinion in Neurobiology,
2007
3.

Palmer C.L., Cotton L., Henley J.M.
Pharmacological Reviews,
2005
4.
10.1016/j.mencom.2018.05.028_bib0020
O’Neill
IDrugs,
2007
5.

Ring-fused thiadiazines as core structures for the development of potent AMPA receptor potentiators.
Pirotte B., Francotte P., Goffin E., Fraikin P., Danober L., Lesur B., Botez I., Caignard D.-., Lestage P., de Tullio P.
Current Medicinal Chemistry,
2010
6.

E. Ward S., Harries M.
Current Medicinal Chemistry,
2010
7.

Dicou E., Rangon C., Guimiot F., Spedding M., Gressens P.
Brain Research,
2003
8.

Zarate C.A., Manji H.K.
Experimental Neurology,
2008
9.

Alt A., Nisenbaum E.S., Bleakman D., Witkin J.M.
Biochemical Pharmacology,
2006
10.

Goff D.C., Leahy L., Berman I., Posever T., Herz L., Leon A.C., Johnson S.A., Lynch G.
Journal of Clinical Psychopharmacology,
2003
11.

O'Neill M., Bleakman D., Zimmerman D., Nisenbaum E.
Current Drug Targets - CNS & Neurological Disorders,
2004
12.

Lynch G.
Current Opinion in Pharmacology,
2004
13.

Adler L.A., Kroon R.A., Stein M., Shahid M., Tarazi F.I., Szegedi A., Schipper J., Cazorla P.
Biological Psychiatry,
2012
14.

Lauterborn J.C., Pineda E., Chen L.Y., Ramirez E.A., Lynch G., Gall C.M.
Neuroscience,
2009
15.

Tikhonova I.G., Lavrov M.I., Palyulin V.A., Zefirov N.S.
Doklady Biochemistry and Biophysics,
2004
16.

Lavrov M.I., Lapteva V.L., Grigor’ev V.V., Palyulin V.A., Bachurin S.O., Zefirov N.S.
Pharmaceutical Chemistry Journal,
2012
17.

Lavrov M.I., Grigor’ev V.V., Bachurin S.O., Palyulin V.A., Zefirov N.S.
Doklady Biochemistry and Biophysics,
2015
18.

Karlov D.S., Lavrov M.I., Palyulin V.A., Zefirov N.S.
Russian Chemical Bulletin,
2016
19.
Radchenko E.V., Karlov D.S., Lavrov M.I., Palyulin V.A.
Mendeleev Communications,
2017
20.

Davis I.W., Raha K., Head M.S., Baker D.
Protein Science,
2009
21.
VIDA 4.3.0: OpenEye Scientific Software, Santa Fe, NM, http://www.eyesopen.com.
22.
Grapheme Toolkit 2017.Feb.1 OpenEye Scientific Software, Santa Fe, NM, http://www.eyesopen.com.
23.

McGann M.R., Almond H.R., Nicholls A., Grant J.A., Brown F.K.
Biopolymers,
2002
24.

Yim E.S., Park M.K., Han B.H.
Ultrasonics Sonochemistry,
1997
25.

Thompson M.J., Chen B.
Journal of Organic Chemistry,
2009
26.

Kuznetsov A.I., Basargin E.B., Moskovkin A.S., Ba M.K., Miroshnichenko I.V., Botnikov M.Y., Unkovskii B.V.
Chemistry of Heterocyclic Compounds,
1985
27.

Perlovich G.L., Proshin A.N., Volkova T.V., Kurkov S.V., Grigoriev V.V., Petrova L.N., Bachurin S.O.
Journal of Medicinal Chemistry,
2009