Abstract
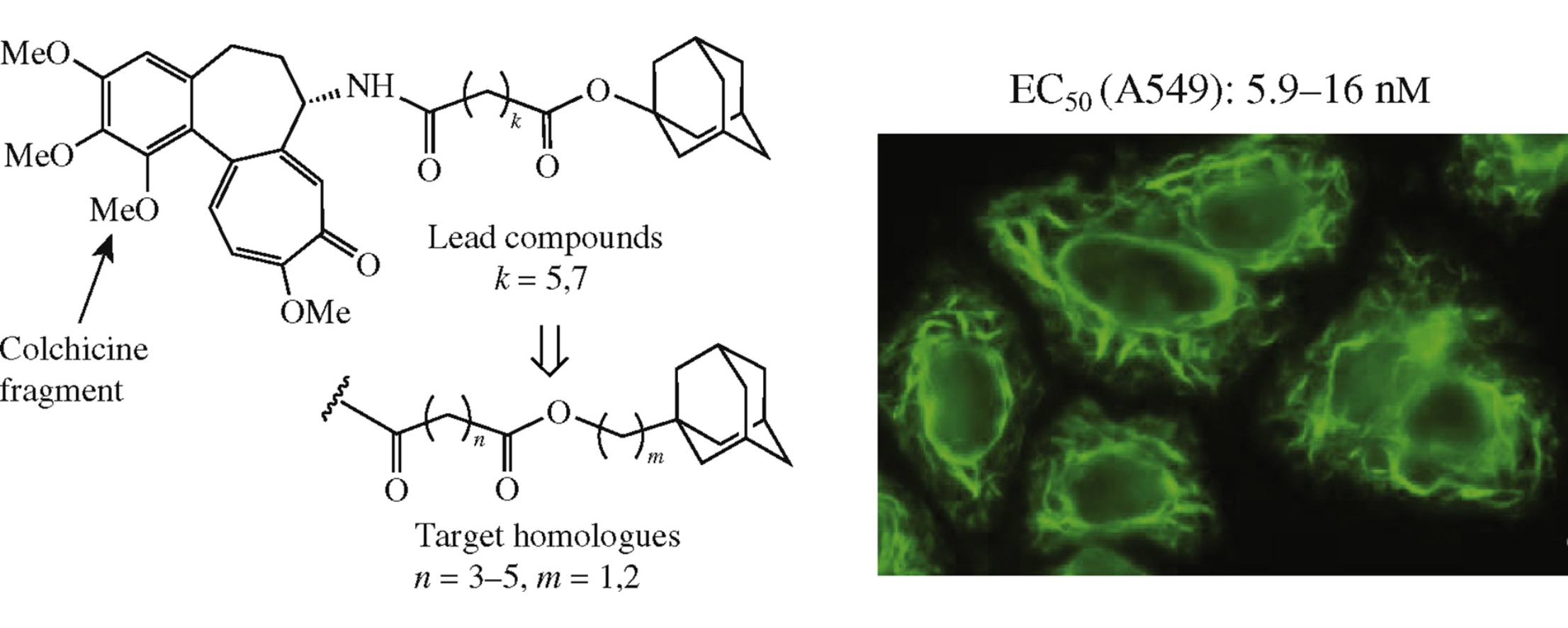
Homologues of N-[7-(adamantan-1-yloxy)-7-oxoheptanoyl]-N-deacetylcolchicine with sequential shift of the ester group in the chain connecting colchicine and adamantane moieties were synthesized to find an optimal position of this group. All homologues possessed very high cytotoxicity to human lung carcinoma cell line A549 demonstrating a weak dependence of toxic activity on the ester group position. The cytotoxicity (EC50=5.9nm) of the most active compound was close to that of clinically used anti-tubulin anticancer drug taxol.
References
1.
10.1016/j.mencom.2018.05.027_bib0005
The Practice of Medicinal Chemistry,
2003
2.
Marrazzo A., Pappalardo A., Prezzavento O., Vittorio F.
Arkivoc,
2004
3.

Neumeyer J.L., Zhang A., Xiong W., Gu X., Hilbert J.E., Knapp B.I., Negus S.S., Mello N.K., Bidlack J.M.
Journal of Medicinal Chemistry,
2003
4.

Zefirova O.N., Nurieva E.V., Shishov D.V., Baskin I.I., Fuchs F., Lemcke H., Schröder F., Weiss D.G., Zefirov N.S., Kuznetsov S.A.
Bioorganic and Medicinal Chemistry,
2011
5.

Zefirova O.N., Nurieva E.V., Wobith B., Gogol V.V., Zefirov N.A., Ogonkov A.V., Shishov D.V., Zefirov N.S., Kuznetsov S.A.
Molecular Diversity,
2017
6.

Bagnato J.D., Eilers A.L., Horton R.A., Grissom C.B.
Journal of Organic Chemistry,
2004
7.

Mosmann T.
Journal of Immunological Methods,
1983
8.
Vasilenko D.A., Averina E.B., Zefirov N.A., Wobith B., Grishin Y.K., Rybakov V.B., Zefirova O.N., Kuznetsova T.S., Kuznetsov S.A., Zefirov N.S.
Mendeleev Communications,
2017
9.
Vostrikov N.S., Lobko I.F., Spirikhin L.V., Vakhitova Y.V., Pivnitsky K.K., Miftakhov M.S.
Mendeleev Communications,
2017
10.

Balaev A.N., Osipov V.N., Okhmanovich K.A., Ruchko E.A., Baryshnikova M.A., Khachatryan D.S.
Russian Chemical Bulletin,
2016
11.

Zefirov N.A., Nurieva E.V., Pikulina Y.A., Ogon´kov A.V., Wobith B., Kuznetsov S.A., Zefirova O.N.
Russian Chemical Bulletin,
2017
12.

Potten C.
British Journal of Cancer,
1996