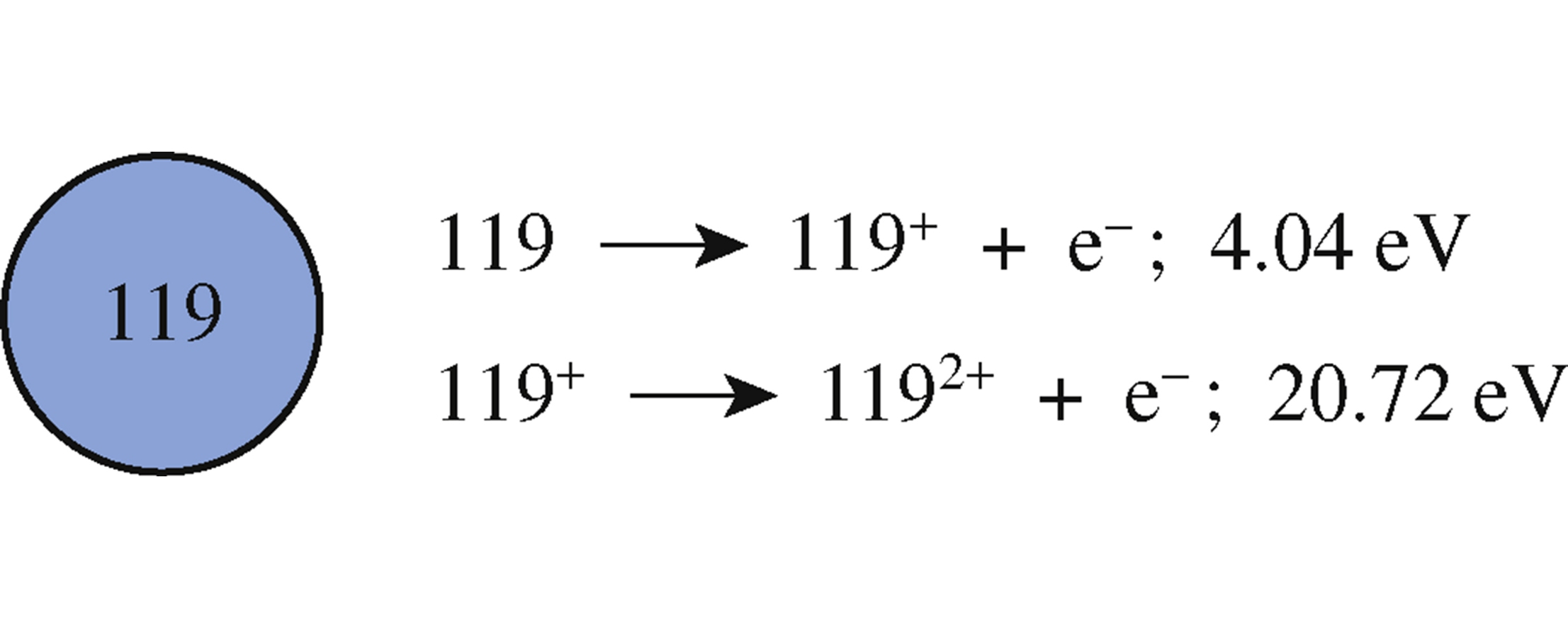Abstract
The following empirical equation was derived: UPOT=(−0.494r++122.392)(η++η−)−109.514η++1191.694kJmol−1, where r+ is the cation radius (pm), η+ is the absolute hardness of the cation, and η− is the absolute hardness of the anion. This equation in conjunction with Kapustinskii lattice energy values for element 119 halides was employed to calculate the first and second ionization energies of 4.04 and 20.72eV, respectively; for the monocation 119+, r+=202.2pm, η+=8.34 and η+=12.38. Furthermore, the second ionization energy for francium was calculated as 23.80eV.
References
1.

de Farias R.F.
Chemical Physics Letters,
2017
2.
10.1016/j.mencom.2018.05.026_bib0010
de Farias
Chem. Res. J.,
2017
3.
10.1016/j.mencom.2018.05.026_bib0015
The Chemistry of the Superheavy Elements,
2014
4.

Türler A., Pershina V.
Chemical Reviews,
2013
5.

Thierfelder C., Schwerdtfeger P., Koers A., Borschevsky A., Fricke B.
Physical Review A,
2009
6.

Kaya S., Fernandes de Farias R.
Chemical Physics Letters,
2018
7.
10.1016/j.mencom.2018.05.026_bib0035
Putz
Absolute and Chemical Electronegativity and Hardness,
2009
8.

Shannon R.D.
Acta Crystallographica Section A,
1976
9.

Kapustinskii A.F.
Quarterly Reviews Chemical Society,
1956
10.
10.1016/j.mencom.2018.05.026_bib0050
CRC Handbook of Chemistry and Physics,
2016
11.
10.1016/j.mencom.2018.05.026_bib0055
Relativistic Effects in Heavy-Element Chemistry and Physics,
2003
12.

Eliav E., Vilkas M.J., Ishikawa Y., Kaldor U.
Chemical Physics,
2005
13.

Bonchev D., Kamenska V.
The Journal of Physical Chemistry,
1981