Abstract
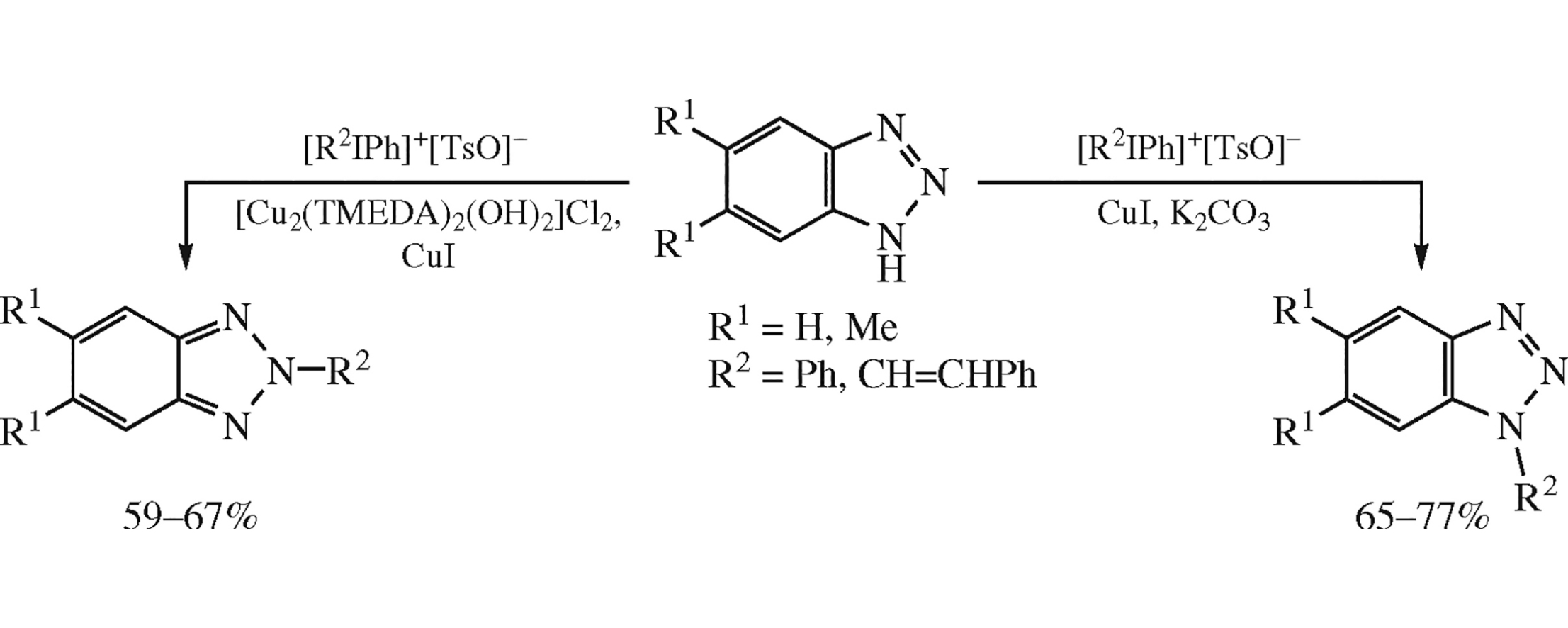
Modification of benzotriazoles with iodonium salts [diphenyl- and (E)-styrylphenyliodonium tosylates] occurs at the N1-position in the presence of K2CO3 as a base and CuI as a catalyst in CH2Cl2, whereas in the presence of stoichiometric amount of [Cu2(TMEDA)2(OH)2]Cl2 complex regioselective N2-modification proceeds. Two new CuI complexes based on benzotriazoles were synthesized and their crystal structures were determined by X-ray powder diffraction analysis.
References
1.

Katritzky A.R., Lan X., Yang J.Z., Denisko O.V.
Chemical Reviews,
1998
2.

Katritzky A.R., Rachwal S.
Chemical Reviews,
2009
3.

Katritzky A.R., Rachwal S.
Chemical Reviews,
2011
4.

Dennis Hall C., Panda S.S.
Advances in Heterocyclic Chemistry,
2016
5.

Briguglio I., Piras S., Corona P., Gavini E., Nieddu M., Boatto G., Carta A.
European Journal of Medicinal Chemistry,
2015
6.
10.1016/j.mencom.2018.05.019_sbref0010b
Mishra
Int. J. Pharm. Technol. Biotechnol.,
2015
7.

Claramunt R., María D., Pinilla E., Torres M., Elguero J.
Molecules,
2007
8.

Khatun N., Modi A., Ali W., Patel B.K.
Journal of Organic Chemistry,
2015
9.

Schöpff M.
Berichte der deutschen chemischen Gesellschaft,
1890
10.
11.

Zhang F., Moses J.E.
Organic Letters,
2009
12.

Gann A.W., Amoroso J.W., Einck V.J., Rice W.P., Chambers J.J., Schnarr N.A.
Organic Letters,
2014
13.
![[3 + 2] Cycloadditions of Azides with Arynes via Photolysis of Phthaloyl Peroxide Derivatives](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chang D., Zhu D., Shi L.
Journal of Organic Chemistry,
2015
14.

Chen Q., Yu H., Xu Z., Lin L., Jiang X., Wang R.
Journal of Organic Chemistry,
2015
15.

Alimi I., Remy R., Bochet C.G.
European Journal of Organic Chemistry,
2017
16.

Houghton P.G., Pipe D.F., Rees C.W.
Journal of the Chemical Society Perkin Transactions 1,
1985
17.

Hall J.H.
Journal of Organic Chemistry,
1968
18.
W. Fischer, K. Fritzsche, W. Wolf, L. Bore, Patent WO 2002024668 A1, 2002.
19.
![Preparation of 2-(2H-Benzotriazol-2-yl)phenols by the Reduction of 2-[(2-Nitrophenyl)azo]phenols with ThioureaS,S-Dioxide](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Tanimoto S., Kamano T.
Synthesis,
1986
20.

Kim B.H., Kim S.K., Lee Y.S., Jun Y.M., Baik W., Lee B.M.
Tetrahedron Letters,
1997
21.

Liu G., Zhao H., Yang H., Gao X., Li M., Thiemann T.
Advanced Synthesis and Catalysis,
2007
22.

Dong J., Jin B., Sun P.
Organic Letters,
2014
23.

Nykaza T.V., Harrison T.S., Ghosh A., Putnik R.A., Radosevich A.T.
Journal of the American Chemical Society,
2017
24.

Mukhopadhyay C., Tapaswi P.K., Butcher R.J.
Organic and Biomolecular Chemistry,
2010
25.

Liu Q., Wen D., Hang C., Li Q., Zhu Y.
Helvetica Chimica Acta,
2010
26.

Zimmermann V., Bräse S.
Journal of Combinatorial Chemistry,
2007
27.

Zhou J., He J., Wang B., Yang W., Ren H.
Journal of the American Chemical Society,
2011
28.

Takagi K., Al-Amin M., Hoshiya N., Wouters J., Sugimoto H., Shiro Y., Fukuda H., Shuto S., Arisawa M.
Journal of Organic Chemistry,
2014
29.

Kumar R.K., Ali M.A., Punniyamurthy T.
Organic Letters,
2011
30.

Jo J., Lee H.Y., Liu W., Olasz A., Chen C., Lee D.
Journal of the American Chemical Society,
2012
31.

Shang X., Zhao S., Chen W., Chen C., Qiu H.
Chemistry - A European Journal,
2014
32.

Ryu T., Min J., Choi W., Jeon W.H., Lee P.H.
Organic Letters,
2014
33.

Li J., Zhou H., Zhang J., Yang H., Jiang G.
Chemical Communications,
2016
34.

Nachtsheim B., Riedmüller S.
Synlett,
2015
35.

Bihari T., Babinszki B., Gonda Z., Kovács S., Novák Z., Stirling A.
Journal of Organic Chemistry,
2016
36.

Liu Z., Larock R.C.
Journal of Organic Chemistry,
2006
37.

Katritzky A.R., Rachwal S., Caster K.C., Mahni F., Law K.W., Rubio O.
Journal of the Chemical Society Perkin Transactions 1,
1987
38.

Katritzky A.R., Offerman R.J., Cabildo P., Soleiman M.
Recueil des Travaux Chimiques des Pays-Bas,
2010
39.

Qian H., Huang X.
Synthetic Communications,
2000
40.

Ley S.V., Thomas A.W.
Angewandte Chemie - International Edition,
2003
41.

Beletskaya I.P., Cheprakov A.V.
Coordination Chemistry Reviews,
2004
42.

Beletskaya I.P., Cheprakov A.V.
Organometallics,
2012
43.

Fañanás-Mastral M.
Synthesis,
2017
44.

Ueda S., Su M., Buchwald S.L.
Angewandte Chemie - International Edition,
2011
45.

Liu Y., Yan W., Chen Y., Petersen J.L., Shi X.
Organic Letters,
2008
46.

Lee H., Won J., Kim M., Park S., Jung K., Kim B.R., Lee S., Yoon Y.
Journal of Organic Chemistry,
2009
47.

Chen W., Zhang Y., Zhu L., Lan J., Xie R., You J.
Journal of the American Chemical Society,
2007
48.

Xu Z., Li H., Ren Z., Du W., Xu W., Lang J.
Tetrahedron,
2011
49.

Mukhopadhyay C., Tapaswi P.K.
Synthetic Communications,
2012
50.

Nagaradja E., Chevallier F., Roisnel T., Dorcet V., Halauko Y.S., Ivashkevich O.A., Matulis V.E., Mongin F.
Organic and Biomolecular Chemistry,
2014
51.

Taillefer M., Ouali A., Renard B., Spindler J.
Chemistry - A European Journal,
2006
52.

Kabir M.S., Lorenz M., Namjoshi O.A., Cook J.M.
Organic Letters,
2009
53.

Beletskaya I.P., Davydov D.V., Moreno-Mañas M.
Tetrahedron Letters,
1998
54.

Beletskaya I.P., Davydov D.V., Moreno-Mañas M.
Tetrahedron Letters,
1998
55.

Davydov D.V., Beletskaya I.P., Semenov B.B., Smushkevich Y.I.
Tetrahedron Letters,
2002
56.

Beletskaya I.P., Davydov D.V., Gorovoy M.S.
Tetrahedron Letters,
2002
57.

Yoshimura A., Zhdankin V.V.
Chemical Reviews,
2016
58.
10.1016/j.mencom.2018.05.019_bib0105
Albov
Acta Crystallogr.,
2004
59.

Finšgar M., Milošev I.
Corrosion Science,
2010
60.

Goreshnik E.A., Schollmeyer D., Mys'kiv M.G.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2005