Abstract
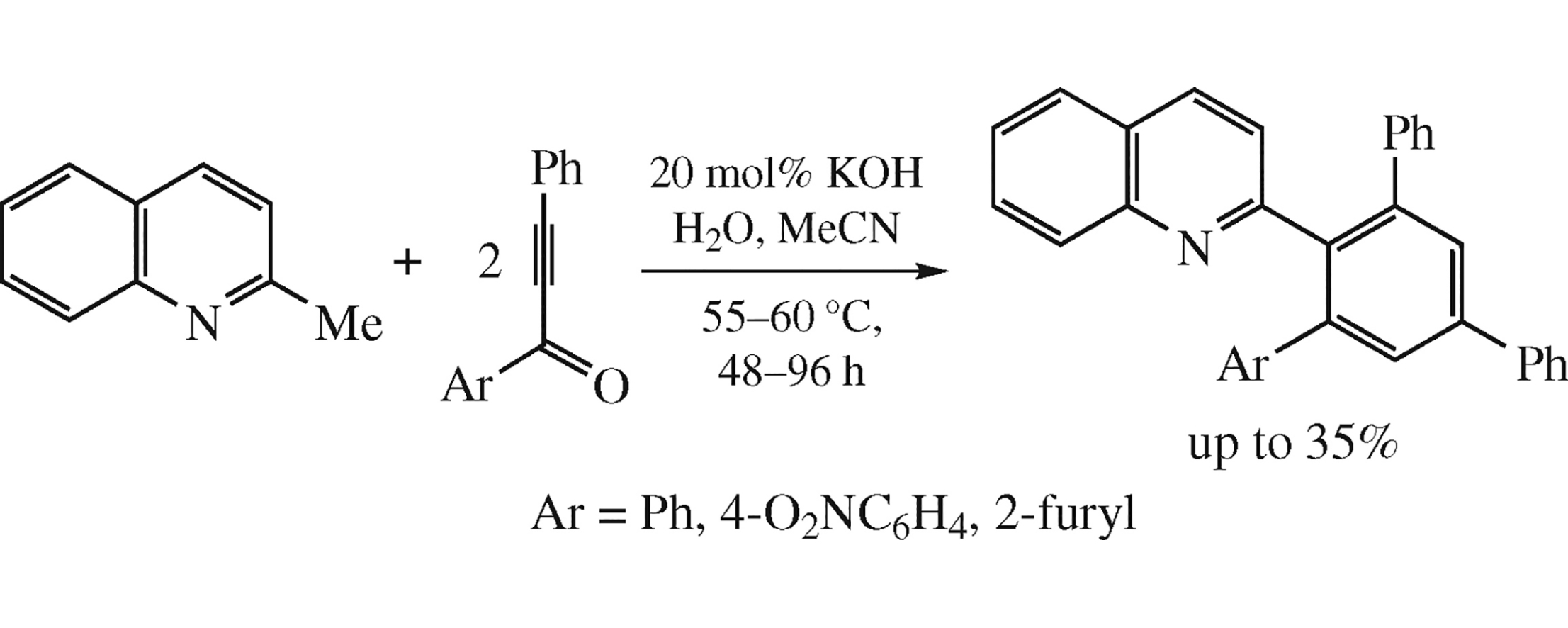
2-Methylquinoline reacts with 1-(het)aryl-3-phenylprop-2-yn-1-ones under mild transition metal-free conditions (55–60°C, 20 mol% KOH, H2O, MeCN) to afford 2-{5’-(het)aryl[1,1’ : 3’,1”]terphenyl-4’-yl}quinolines in up to 35% yield. The reaction likely proceeds via the intermediate 1,3-dipole followed by the double nucleophilic vinylation of the methyl group with two molecules of ynone and subsequent elimination of (het)arenecarboxylic acid.
References
1.
10.1016/j.mencom.2018.05.012_bib0005
Trofimov
Ser. 4,
2014
2.
Trofimov B.A., Nikitina L.P., Belyaeva K.V., Andriyankova L.V., Mal’kina A.G., Bagryanskaya I.Y., Afonin A.V., Ushakov I.A.
Mendeleev Communications,
2016
3.
Trofimov B.A., Belyaeva K.V., Andriyankova L.V., Nikitina L.P., Mal’kina A.G.
Mendeleev Communications,
2017
4.

Trofimov B.A., Andriyankova L.V., Belyaeva K.V., Mal’kina A.G., Nikitina L.P., Afonin A.V., Ushakov I.A.
Journal of Organic Chemistry,
2008
5.
Trofimov B.A., Andriyankova L.V., Belyaeva K.V., Mal’kina A.G., Nikitina L.P., Afonin A.V., Ushakov I.A.
Mendeleev Communications,
2009
6.

Acheson R.M., Wallis J.D., Woollard J.
Journal of the Chemical Society Perkin Transactions 1,
1979
7.

Yavari I., Sabbaghan M., Hossaini Z.
Synlett,
2006
8.
![Stereoselective synthesis of dialkyl 3-spiroindanedione-1,2,3,3a-tetrahydropyrrolo[1,2-a]quinoline-1,2-dicarboxylates](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Yavari I., Mirzaei A., Moradi L., Hosseini N.
Tetrahedron Letters,
2008
9.

Shaabani A., Rezayan A.H., Sarvary A., Heidary M., Ng S.W.
Tetrahedron,
2009
10.
![Diastereoselective synthesis of fused [1,3]oxazines from ethyl pyruvate, activated acetylenes and N-heterocycles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Yavari I., Mirzaei A., Hossaini Z., Souri S.
Molecular Diversity,
2009
11.
10.1016/j.mencom.2018.05.012_bib0055
Moafi
Synthesis,
2011
12.

Trofimov B.A., Andriyankova L.V., Belyaeva K.V.
Chemistry of Heterocyclic Compounds,
2012
13.

Rostami-Charati F., Hossaini Z., Gharaee E., Khalilzadeh M.A.
Journal of Heterocyclic Chemistry,
2013
14.

Ditchfield R.
Molecular Physics,
1974
15.
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, Ö. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 2009, Gaussian, Inc., Pittsburg, 2009.
16.

Galkin K.I., Ananikov V.P.
Russian Chemical Reviews,
2016
17.

Pang J., Marcotte E.J., Seward C., Brown R.S., Wang S.
Angewandte Chemie - International Edition,
2001
18.

Brunel J., Ledoux I., Zyss J., Blanchard-Desce M.
Chemical Communications,
2001
19.

Li L., Chen M., Zhang H., Nie H., Sun J.Z., Qin A., Tang B.Z.
Chemical Communications,
2015
20.

Durairaj N., Kalainathan S., Kumar R.
Optik,
2017
21.

Mongin O., Brunel J., Porrès L., Blanchard-Desce M.
Tetrahedron Letters,
2003
22.

He Q., Huang H., Yang J., Lin H., Bai F.
Journal of Materials Chemistry A,
2003
23.
![Synthesis ofC3 Benzo[1,2-e:3,4-e′:5,6-e′′]tribenzo[l]acephenanthrylenes (“Crushed Fullerene” Derivatives) by Intramolecular Palladium-Catalyzed Arylation](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gómez-Lor B., González-Cantalapiedra E., Ruiz M., de Frutos Ó., Cárdenas D.J., Santos A., Echavarren A.M.
Chemistry - A European Journal,
2004
24.

Zhang S., Xue Z., Gao Y., Mao S., Wang Y.
Tetrahedron Letters,
2012
25.

Sueki S., Okamoto C., Shimizu I., Seto K., Furukawa Y.
Bulletin of the Chemical Society of Japan,
2010
26.
S.-M. Chi, J.-H. Hah, K.-S. Kim, W.-S. Kim and M.-H. Ryoo, Patent US 20080188380 A1, 2008.(Chem. Abstr., 2008, 149, 241730).
27.
K.V. Belyaeva, L.P. Nikitina, A.V. Afonin, A.V. Vashchenko and B. A. Trofimov, Russ. Chem. Bull., Int. Ed., 2017, 67, 2258 (Izv. Akad. Nauk., Ser. Khim., 2017, 2258).