Abstract
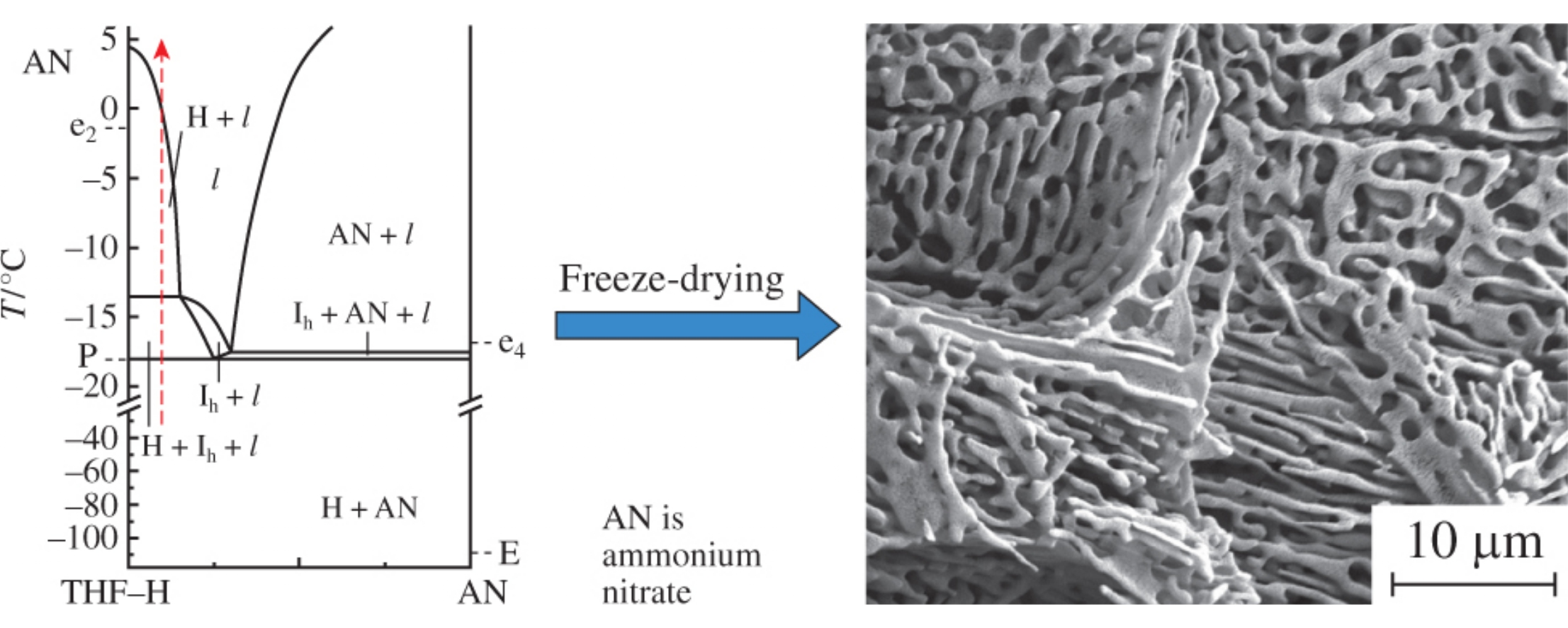
Fine powders of ammonium nitrate were prepared by the freeze-drying of a solution in a THF–water mixture. Freezing gives a THF hydrate and dispersed ammonium nitrate particles; the THF hydrate can be then removed by sublimation to leave fine ammonium nitrate powder. To optimize the process, the stability of cubic structure II THF clathrate hydrate in the presence of ammonium nitrate was studied by X-ray diffraction and thermal analysis.
References
1.

Oommen C.
Journal of Hazardous Materials,
1999
2.

Yunoshev A.S., Rafeichik S.I., Plastinin A.V., Sil’vestrov V.V.
Combustion, Explosion and Shock Waves,
2013
3.

Shlyakhtin O.A., Tretyakov Y.D.
Journal of Materials Chemistry A,
1999
4.

Generalov M.B., Trutnev N.S.
Theoretical Foundations of Chemical Engineering,
2007
5.
Yu. A. Dyadin, I.V. Bondaryuk and F. V. Zhurko, in Inclusion Compounds, eds. J. L. Atwood, J.E. D. Davies and D. D. MacNicol, Oxford University Press, Oxford, 1991, vol. 5, pp. 214-275.
6.

Ogienko A.G., Drebushchak V.A., Bogdanova E.G., Yunoshev A.S., Ogienko A.A., Boldyreva E.V., Manakov A.Y.
Journal of Thermal Analysis and Calorimetry,
2016
7.

Hendricks S.B., Posnjak E., Kracek F.C.
Journal of the American Chemical Society,
1932
8.

Rodebush W.H.
Journal of the American Chemical Society,
1918
9.

Millican I.L., Joseph A.F., Lowry T.M.
Journal of the Chemical Society Transactions,
1922
10.

Stoporev A.S., Manakov A.Y., Altunina L.K., Strelets L.A., Kosyakov V.I.
Canadian Journal of Chemistry,
2015
11.

Bhatnagar B.S., Martin S.M., Teagarden D.L., Shalaev E.Y., Suryanarayanan R.
Journal of Pharmaceutical Sciences,
2010
12.

Lebedev B.V., Rabinovich I.B., Milov V.I., Lityagov V.Y.
Journal of Chemical Thermodynamics,
1978
13.

Overhoff K.A., Johnston K.P., Tam J., Engstrom J., Williams R.O.
Journal of Drug Delivery Science and Technology,
2009
14.
Zelenov V.P., Bukalov S.S., Leites L.A., Aysin R.R., Subbotin A.N., Struchkova M.I., Fedyanin I.V.
Mendeleev Communications,
2017