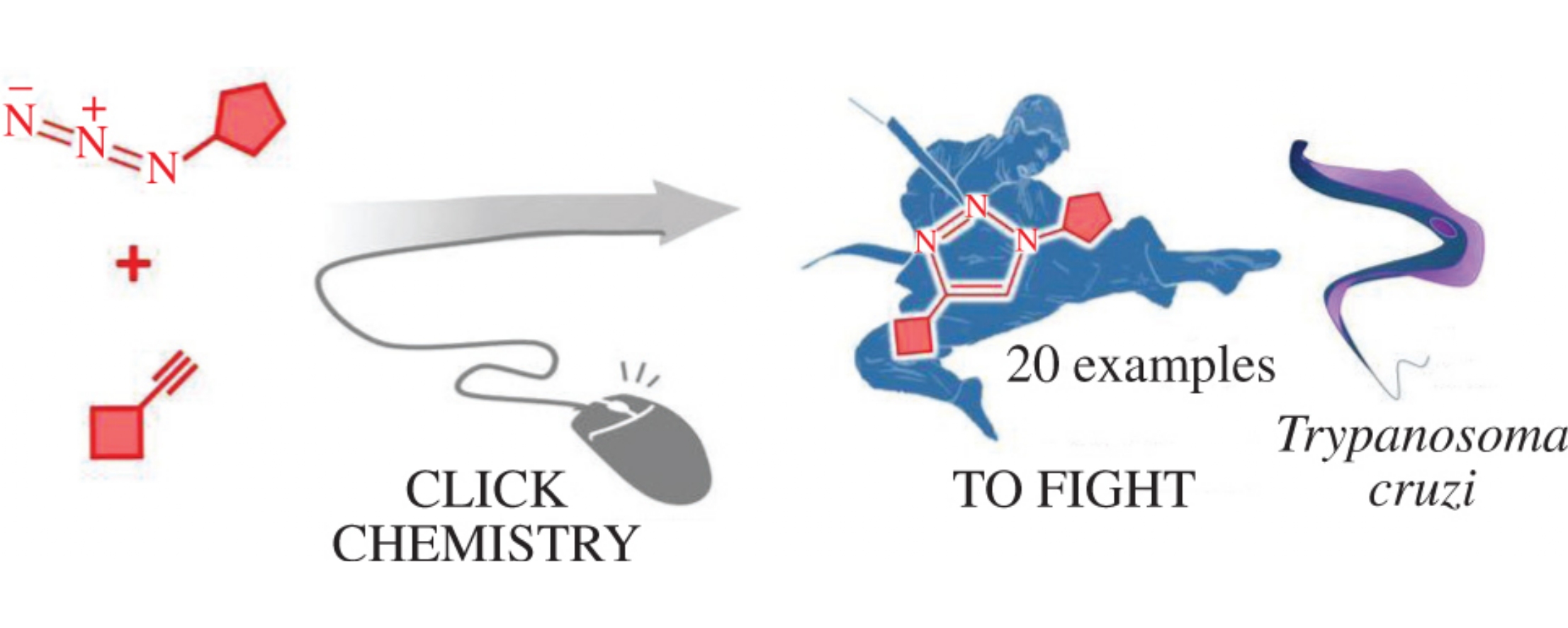
Abstract
Twenty novel azole-naftifine analogues were obtained using azide–alkyne click reaction. Five of them were more potent than the positive control naftifine revealing an unprecedented antiproliferative effects against Trypanosoma cruzi.
References
1.

de Fátima Â., Marquissolo C., de Albuquerque S., Carraro-Abrahão A.A., Pilli R.A.
European Journal of Medicinal Chemistry,
2006
2.

3.

Coura J.R., Borges-Pereira J.
Acta Tropica,
2010
4.

5.
Coura J.R., Castro S.L.
Memorias do Instituto Oswaldo Cruz,
2002
6.

Salomao K., Figueiredo Sadok Menna-Barreto R., Lisboa de Castro S.
Current Topics in Medicinal Chemistry,
2016
7.

G. Ferreira L., T. de Oliveira M., D. Andricopulo A.
Current Topics in Medicinal Chemistry,
2016
8.

Buckner F., Yokoyama K., Lockman J., Aikenhead K., Ohkanda J., Sadilek M., Sebti S., Van Voorhis W., Hamilton A., Gelb M.H.
Proceedings of the National Academy of Sciences of the United States of America,
2003
9.

Trypanocidal activity of 2-propen-1-amine derivatives on trypomastigotes culture and in animal model
Oliveira D.A., Pereira D.G., Fernandes A.M., De Castro S.L., Souza Brito A.R., De Souza A.O., Durán N.
Parasitology Research,
2004
10.

Gerpe A., Odreman-Nuñez I., Draper P., Boiani L., Urbina J.A., González M., Cerecetto H.
Bioorganic and Medicinal Chemistry,
2008
11.

Gerpe A., Boiani L., Hernández P., Sortino M., Zacchino S., González M., Cerecetto H.
European Journal of Medicinal Chemistry,
2010
12.

de Macedo-Silva S., Souza W., Rodrigues J.
Current Medicinal Chemistry,
2015
13.

Petranyi G., Ryder N.S., Stütz A.
Science,
1984
14.

15.

Ryder N.S., Dupont M.C.
Biochemical Journal,
1985
16.

Sud I.J., Feingold D.S.
Journal of Investigative Dermatology,
1981
17.

Van den Bossche H., Willemsens G., Cools W., Cornelissen F., Lauwers W.F., van Cutsem J.M.
Antimicrobial Agents and Chemotherapy,
1980
18.

Vanden Bossche H., Bellens D., Cools W., Gorrens J., Marichal P., Verhoeven H., Willemsens G., De Coster R., Beerens D., Haelterman C., Coene M., Lauwers W., le Jeune L.
Drug Development Research,
1986
19.

Goad L.J., Berens R.L., Marr J.J., Beach D.H., Holz G.G.
Molecular and Biochemical Parasitology,
1989
20.

McCabe R.E., Remington J.S., Araujo F.G.
Journal of Infectious Diseases,
1984
21.

McCabe R.E., Araujo F.G., Remington J.S.
American Journal of Tropical Medicine and Hygiene,
1986
22.

23.

Totobenazara J., Burke A.J.
Tetrahedron Letters,
2015
24.
10.1016/j.mencom.2018.03.029_bib0120
Borgati
J. Braz. Chem. Soc.,
2013
25.
10.1016/j.mencom.2018.03.029_bib0125
Zauli-Nascimento
Trop. Med. Int. Health,
2010