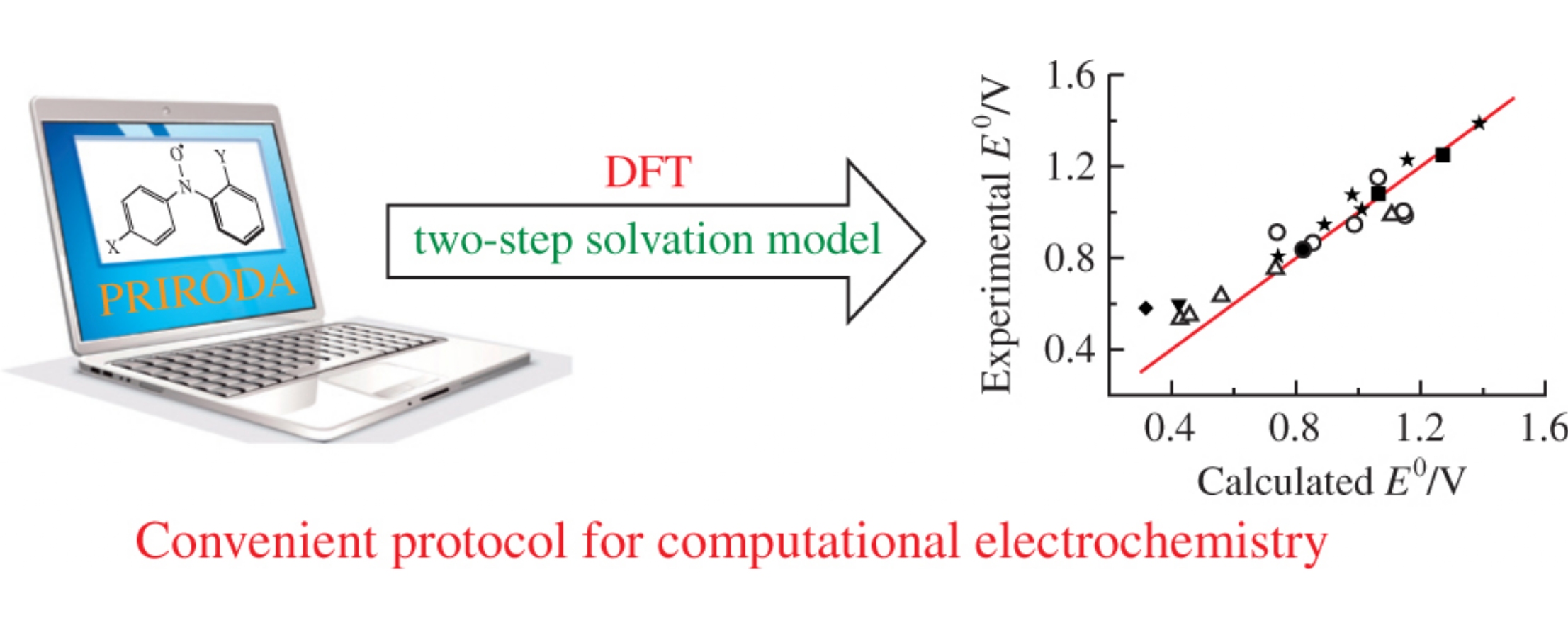
Abstract
Based on a combination of a gas-phase DFT electronic structure calculation at PBE/L2 level implemented in the highly efficient ‘Priroda’ program package with consecutive fast solvation energy evaluation by continuum solvation model, a liable methodology has been developed for accurate calculation of the standard redox potentials for diarylnitroxides in acetonitrile solution.
References
1.

Poizot P., Dolhem F.
Energy and Environmental Science,
2011
2.

Liang Y., Tao Z., Chen J.
Advanced Energy Materials,
2012
3.

Brushett F.R., Vaughey J.T., Jansen A.N.
Advanced Energy Materials,
2012
4.
10.1016/j.mencom.2018.03.026_bib0020
Suga
Stable Radicals: Fundamentals and Applied Aspects of Odd-Electron Compounds,
2010
5.

Nakahara K., Iwasa S., Satoh M., Morioka Y., Iriyama J., Suguro M., Hasegawa E.
Chemical Physics Letters,
2002
6.

Nishide H., Iwasa S., Pu Y., Suga T., Nakahara K., Satoh M.
Electrochimica Acta,
2004
7.

8.

Yoshikawa H., Kazama C., Awaga K., Satoh M., Wada J.
Chemical Communications,
2007
9.

Qu J., Morita R., Satoh M., Wada J., Terakura F., Mizoguchi K., Ogata N., Masuda T.
Chemistry - A European Journal,
2008
10.

Levitskiy O.A., Sentyurin V.V., Magdesieva T.V.
Electrochimica Acta,
2018
11.

Marenich A.V., Ho J., Coote M.L., Cramer C.J., Truhlar D.G.
Physical Chemistry Chemical Physics,
2014
12.

Fish J.R., Swarts S.G., Sevilla M.D., Malinski T.
The Journal of Physical Chemistry,
1988
13.

Rychnovsky S.D., Vaidyanathan R., Beauchamp T., Lin R., Farmer P.J.
Journal of Organic Chemistry,
1999
14.

Gryn'ova G., Barakat J.M., Blinco J.P., Bottle S.E., Coote M.L.
Chemistry - A European Journal,
2012
15.

Hodgson J.L., Namazian M., Bottle S.E., Coote M.L.
Journal of Physical Chemistry A,
2007
16.

Shibuya M., Pichierri F., Tomizawa M., Nagasawa S., Suzuki I., Iwabuchi Y.
Tetrahedron Letters,
2012
17.

Blinco J.P., Hodgson J.L., Morrow B.J., Walker J.R., Will G.D., Coote M.L., Bottle S.E.
Journal of Organic Chemistry,
2008
18.

Laikov D.N., Ustynyuk Y.A.
Russian Chemical Bulletin,
2005
19.

Laikov D.N.
Chemical Physics Letters,
1997
20.
![Generalized Gradient Approximation Made Simple [Phys. Rev. Lett. 77, 3865 (1996)]](/storage/images/resized/nrK64iXHTzj43wMrfN1ZoUQ0vanswGzWPN45K3jA_small_thumb.webp)
Perdew J.P., Burke K., Ernzerhof M.
Physical Review Letters,
1997
21.

Laikov D.N.
Chemical Physics Letters,
2005
22.

Schmidt am Busch M., Knapp E.
Journal of the American Chemical Society,
2005
23.

Nakahara K., Iwasa S., Iriyama J., Morioka Y., Suguro M., Satoh M., Cairns E.J.
Electrochimica Acta,
2006
24.

25.

Marx L., Schöllhorn B.
New Journal of Chemistry,
2006
26.

Bogart J.A., Lee H.B., Boreen M.A., Jun M., Schelter E.J.
Journal of Organic Chemistry,
2013
27.

Levitskiy O.A., Eremin D.B., Bogdanov A.V., Magdesieva T.V.
European Journal of Organic Chemistry,
2017
28.
Levitskiy O.A., Sentyurin V.V., Bogdanov A.V., Magdesieva T.V.
Mendeleev Communications,
2016
29.

Levitskiy O.A., Grishin Y.K., Sentyurin V.V., Magdesieva T.V.
Chemistry - A European Journal,
2017
30.

Calder A., Forrester A.R.
Journal of the Chemical Society C Organic,
1969