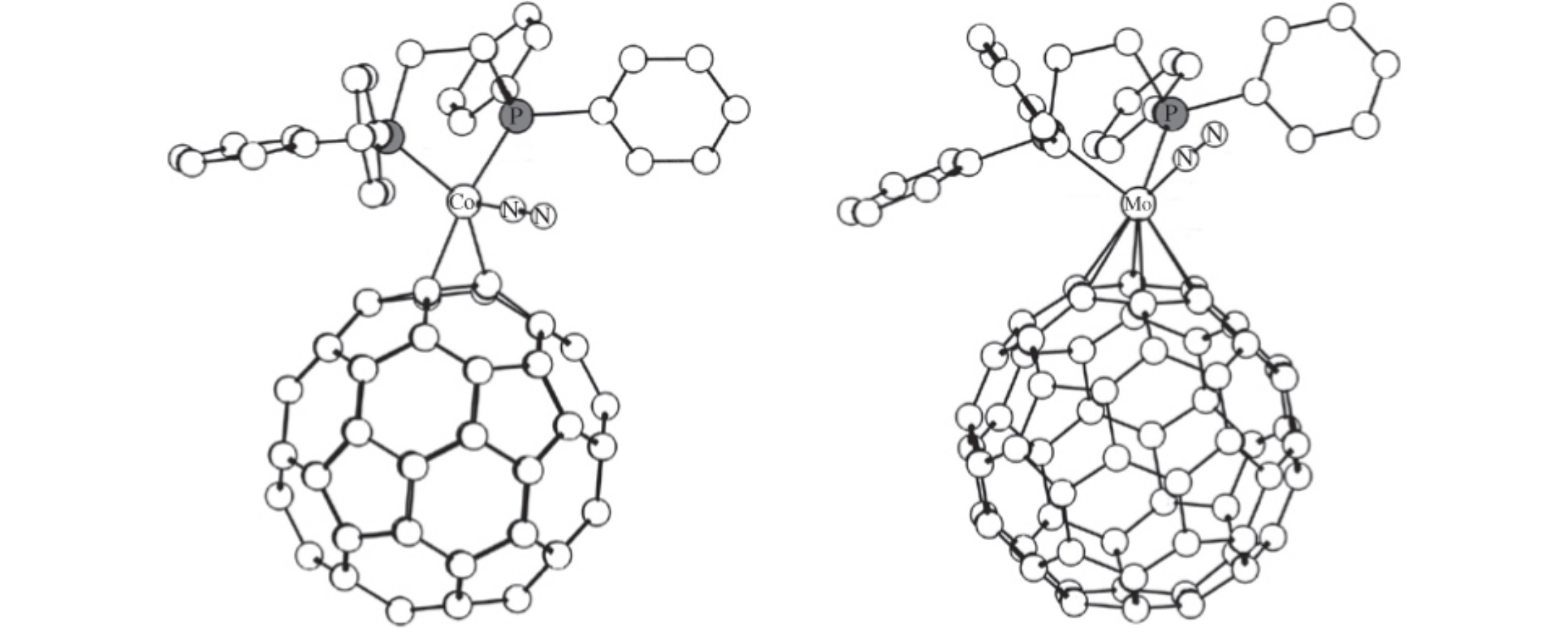
Abstract
Quantum chemical modeling demonstrates the stability of Co0 and Mo0 metal complexes with N2 and C60 ligands in the coordination sphere. These complexes may be used as convenient synthons for the synthesis of polynuclear metal complexes with bridging fullerene ligands.
References
1.

Konarev D.V., Khasanov S.S., Troyanov S.I., Nakano Y., Ustimenko K.A., Otsuka A., Yamochi H., Saito G., Lyubovskaya R.N.
Inorganic Chemistry,
2013
2.

Davis B.R., Payne N.C., Ibers J.A.
Inorganic Chemistry,
1969
3.
![Synthesis, characterization, and crystal structure of the dimeric, paramagnetic cobalt(0) complex {[MeC(CH2PPh2)3Co]2(µ-N2)}](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Cecconi F., Ghilardi C.A., Midollini S., Moneti S., Orlandini A., Bacci M.
Journal of the Chemical Society Chemical Communications,
1985
4.

Betley T.A., Peters J.C.
Journal of the American Chemical Society,
2003
5.

Hsu H., Du Y., Albrecht-Schmitt T.E., Wilson S.R., Shapley J.R.
Organometallics,
1998
6.

Henderson R.A.
Journal of the Chemical Society Dalton Transactions,
1988
7.
![Formation of {Co(dppe)}2{μ2-η2:η2-η2:η2-[(C60)2]} Dimers Bonded by Single C–C Bonds and Bridging η2-Coordinated Cobalt Atoms](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Konarev D.V., Troyanov S.I., Ustimenko K.A., Nakano Y., Shestakov A.F., Otsuka A., Yamochi H., Saito G., Lyubovskaya R.N.
Inorganic Chemistry,
2015
8.
![Linear Coordination Fullerene C60 Polymer [{Ni(Me3P)2}(μ-η2,η2-C60)]∞ Bridged by Zerovalent Nickel Atoms](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Konarev D.V., Khasanov S.S., Nakano Y., Otsuka A., Yamochi H., Saito G., Lyubovskaya R.N.
Inorganic Chemistry,
2014
9.

Konarev D.V., Troyanov S.I., Nakano Y., Otsuka A., Yamochi H., Saito G., Lyubovskaya R.N.
Dalton Transactions,
2014
10.

Konarev D.V., Troyanov S.I., Nakano Y., Ustimenko K.A., Otsuka A., Yamochi H., Saito G., Lyubovskaya R.N.
Organometallics,
2013
11.

Perdew J.P., Burke K., Ernzerhof M.
Physical Review Letters,
1996
12.

Laikov D.N.
Chemical Physics Letters,
2005
13.

Laikov D.N., Ustynyuk Y.A.
Russian Chemical Bulletin,
2005
14.

Stevens W.J., Basch H., Krauss M.
Journal of Chemical Physics,
1984
15.

Oprunenko Y.F., Gloriozov I.P.
Journal of Organometallic Chemistry,
2013
16.

Andrews L., Citra A., Chertihin G.V., Bare W.D., Neurock M.
Journal of Physical Chemistry A,
1998
17.

Hirshfeld F.L.
Theoretical Chemistry Accounts,
1977
18.
10.1016/j.mencom.2018.03.022_bib0090
Peigne
Acta Crystallogr,
2015
19.

Keogh D.W., Poli R.
Journal of the American Chemical Society,
1997
20.

Soto D., Salcedo R.
Molecules,
2012