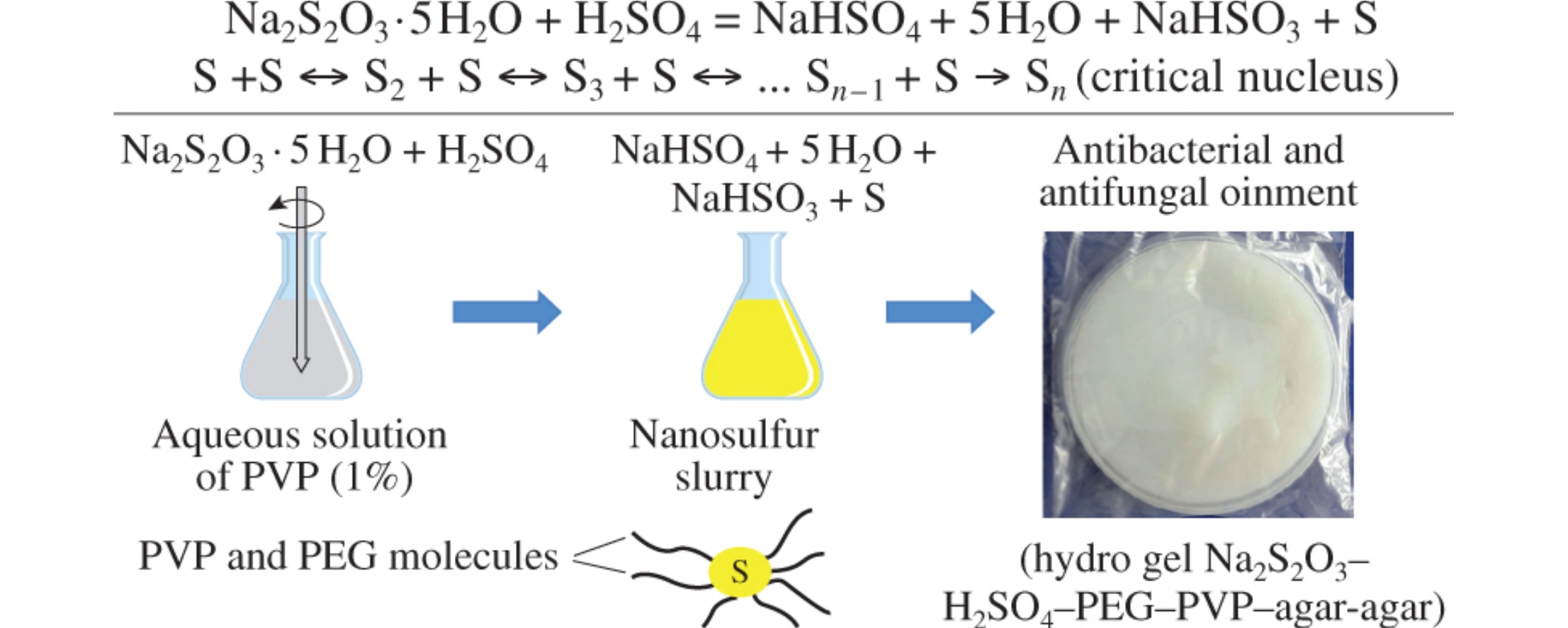
Abstract
A method was developed to prepare sulfur nanoparticles (nanosulfur) via reaction between sodium thiosulfate and sulfuric acid in the presence of water-soluble poly-N-vinylpyrrolidone (PVP) with different molecular weights. Polymeric hydrogel ointments were prepared from agar-agar, PVP, poly(ethylene glycol) solutions with virable concentrations of sodium thiosulfate and sulfuric acid. The ointment samples showed antibacterial and antifungal activities against Staphylococcus aureus, Escherichia coli and Candida guilliermondii.
References
1.
10.1016/j.mencom.2018.03.017_bib0005
Kutney
Sulfur: History, Technology Applications and Industry,
2013
2.
10.1016/j.mencom.2018.03.017_bib0010
Kumar
Curr. Sci. India,
2011
3.
F. Kh. Urakaev, A.I. Bulavchenko, B.M. Uralbekov, I.A. Massalimov, B.B. Tatykaev, A.K. Bolatov, D.N. Zharlykasimova and M. M. Burkitbayev, Colloid J., 2016, 78, 210 (Kolloid. Zh., 2016, 78, 193).
4.
10.1016/j.mencom.2018.03.017_bib0020
Suleiman
J. Mater. Environ. Sci.,
2015
5.

Roy Choudhury S., Mandal A., Ghosh M., Basu S., Chakravorty D., Goswami A.
Applied Microbiology and Biotechnology,
2013
6.

Rao K.J., Paria S.
RSC Advances,
2013
7.
10.1016/j.mencom.2018.03.017_bib0035
Massalimov
Am. Eurasian J. Agric. Environ. Sci.,
2016
8.
10.1016/j.mencom.2018.03.017_bib0040
Massalimov
Architecture,
2013
9.

Chen T., Cheng B., Zhu G., Chen R., Hu Y., Ma L., Lv H., Wang Y., Liang J., Tie Z., Jin Z., Liu J.
Nano Letters,
2016
10.

Jeong T., Chun J., Cho B., Lee J., Kim Y.
Scientific Reports,
2017
11.

Li F., Liu W., Lai Y., Qin F., Zou L., Zhang K., Li J.
Journal of Alloys and Compounds,
2017
12.

Li J., Guo J., Deng J., Huang Y.
Materials Letters,
2017
13.

Nie L., Wang H., Ma J., Liu S., Yuan R.
Journal of Materials Science,
2016
14.

Shao D., Smolianova I., Tang D., Zhang L.
RSC Advances,
2017
15.

Wang H., Huang K., Wang P., Zhong J.
Journal of Alloys and Compounds,
2017
16.

Zhang Y., Li K., Huang J., Wang Y., Peng Y., Li H., Wang J., Zhao J.
Carbon,
2017
17.

Lan Y., Deng B., Kim C., Thornton E.C., Xu H.
Environmental Science & Technology,
2005
18.

Cao M., Ding Y., Ren W., Wang Q., Ragulskis M., Ding Z.
Applied Sciences (Switzerland),
2017
19.

Khang N.C., Van D.Q., Thuy N.M., Minh N.V., Minh P.N.
Journal of Physics and Chemistry of Solids,
2016
20.

Qian H., Tang J., Wang Z., Kim J., Kim J.H., Alshehri S.M., Yanmaz E., Wang X., Yamauchi Y.
Chemistry - A European Journal,
2016
21.

22.

Zhang K., Li J., Liu W., Liu J., Yan C.
International Journal of Hydrogen Energy,
2016
23.

Ghanemi K., Nikpour Y., Omidvar O., Maryamabadi A.
Talanta,
2011
24.

Bura-Nakić E., Marguš M., Jurašin D., Milanović I., Ciglenečki-Jušić I.
Geochemical Transactions,
2015
25.

Li R., Yang T., Li Z., Gu Z., Wang G., Liu J.
Analytica Chimica Acta,
2017
26.
Urakaev F.K.
Mendeleev Communications,
2005
27.

Urakaev F.K.
Combustion Science and Technology,
2013
28.

Mer V.K.
Industrial & Engineering Chemistry,
1952
29.

Urakaev F.K., Bazarov L.S., Meshcheryakov I.N., Feklistov V.V., Drebushchak T.N., Savintsev Y.P., Gordeeva V.I., Shevchenko V.S.
Journal of Crystal Growth,
1999
30.
Urakaev F.K., Drebushchak T.N., Savintsev Y.P., Drebushchak V.A.
Mendeleev Communications,
2003
31.
10.1016/j.mencom.2018.03.017_bib0155
Urakaev
Int. J. Comput. Mater. Sci. Surf. Eng.,
2011
32.
10.1016/j.mencom.2018.03.017_bib0160
Buhler
Polyvinylpyrrolidone: Excipients for Pharmaceuticals (Povidone, Crosspovidone and Copovidone),
2005
33.
B. V. Robinson, F.M. Sullivan, J.F. Borzelleca and S. L. Schwartz, PVP: A Critical Review of the Kinetics and Toxicology of Polyvinylpyrrolidone (Povidone), Lewis Publisher, Chelsea, MI, 1990.
34.
Poly(Ethylene Glycol) Chemistry. Biotechnical and Biomedical Applications, ed. J. M. Harris, Springer, Boston, MA, 1992.
35.

Xie X., Li L., Zheng P., Zheng W., Bai Y., Cheng T., Liu J.
Materials Research Bulletin,
2012
36.

Sepassi S., Goodwin D.J., Drake A.F., Holland S., Leonard G., Martini L., Lawrence M.J.
Journal of Pharmaceutical Sciences,
2007
37.
10.1016/j.mencom.2018.03.017_bib0185
Russ. Chem. Bull., Int.,
2015
38.

Rac O., Suchorska-Woźniak P., Fiedot M., Teterycz H.
Beilstein Journal of Nanotechnology,
2014
39.
Kurmaz S.V., Obraztsova N.A.
Mendeleev Communications,
2015
40.

Shah S., Noor I.M., Pitawala J., Albinson I., Bandara T.M., Mellander B.-., Arof A.K.
Optical Materials Express,
2017
41.
10.1016/j.mencom.2018.03.017_bib0205
Torchilin
Advances in Blood Substitutes: Industrial Opportunities and Medical Challenges,
2012
42.
J. Rosiak, A. Rucinska-Rybus and W. Pekala, Patent US 4871490 A, 1989.