Abstract
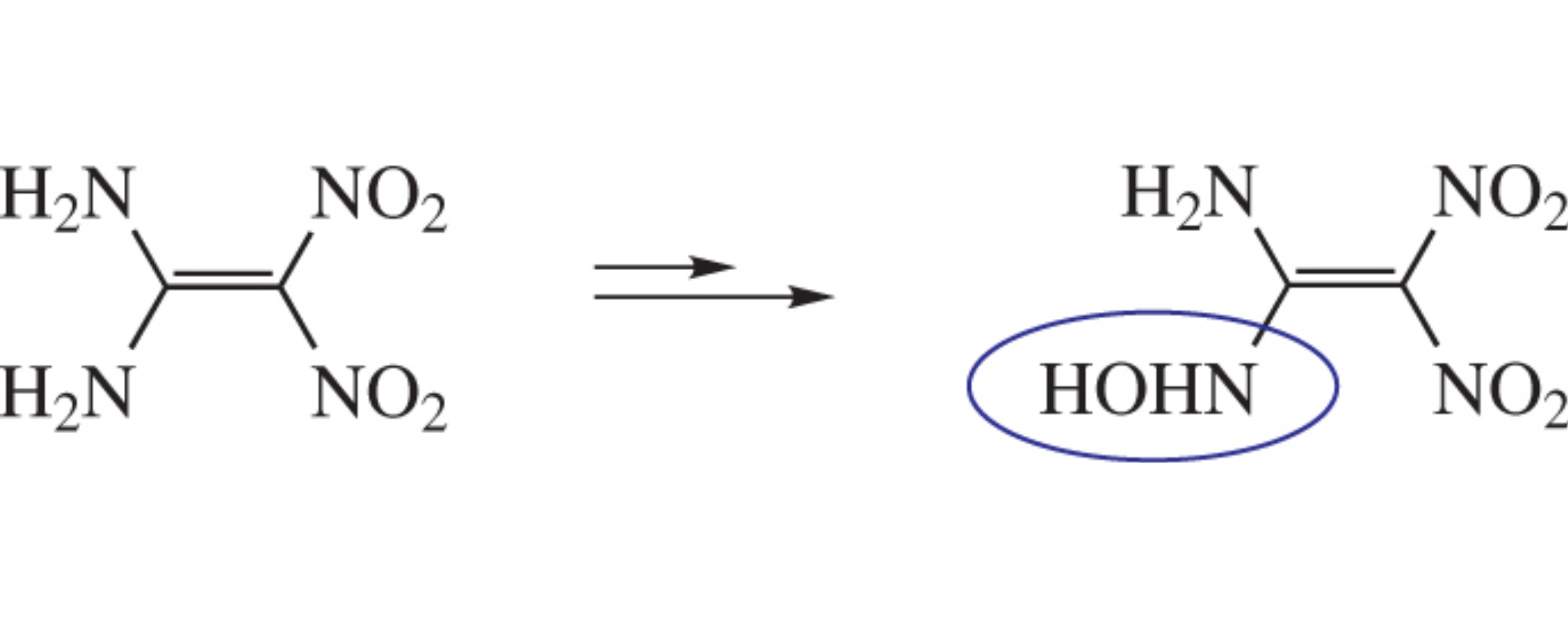
Structurally unique conjugated N-hydroxy nitro enamine, namely, 1-amino-1-hydroxyamino-2,2-dinitroethene, has been synthesized by transamination of 1,1-diamino-2,2-dinitro-ethene (FOX-7) with hydroxylamine. The compound is a highly energetic material, its structure being confirmed by X-ray crystallography.
References
1.
Green Energetic Materials, ed. T. Brinck, Wiley, 2014
2.

Kuchurov I.V., Zharkov M.N., Fershtat L.L., Makhova N.N., Zlotin S.G.
ChemSusChem,
2017
3.

Yin P., Shreeve J.M.
Advances in Heterocyclic Chemistry,
2017
4.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
5.
Ananikov V.P., Eremin D.B., Yakukhnov S.A., Dilman A.D., Levin V.V., Egorov M.P., Karlov S.S., Kustov L.M., Tarasov A.L., Greish A.A., Shesterkina A.A., Sakharov A.M., Nysenko Z.N., Sheremetev A.B., Stakheev A.Y., et. al.
Mendeleev Communications,
2017
6.
Trzciński W., Belaada A.
Central European Journal of Energetic Materials,
2016
7.

Eloy F., Lenaers R.
Chemical Reviews,
1962
8.
I. A. Poplavskaya and R. G. Kurmangalieva, Khimiya amidoksimov (Chemistry of Amidoximes), Nauka, Alma-Ata, 1988 (in Russian).
9.

Bakharev V.V., Gidaspov A.A., Parfenov V.E.
Chemistry of Heterocyclic Compounds,
2017
10.

Simkova L., Liska F., Ludvik J.
Current Organic Chemistry,
2011
11.

Zhang Y., Sun Q., Xu K., Song J., Zhao F.
Propellants, Explosives, Pyrotechnics,
2015
12.

Gao H., Shreeve J.M.
RSC Advances,
2016
13.

Bellamy A.J., Latypov N.V., Goede P.
Journal of Chemical Research,
2003
14.

Shastin A.V., Korsunskii B.L., Godovikova T.I., Lodygina V.P.
Russian Journal of Applied Chemistry,
2009
15.

Gao H., Joo Y., Parrish D.A., Vo T., Shreeve J.M.
Chemistry - A European Journal,
2011
16.
10.1016/j.mencom.2018.03.007_sbrefe0050a
Chang
Acta Chim. Sinica (Huaxue Xuebao),
2008
17.

Axthammer Q.J., Krumm B., Klapötke T.M.
Journal of Physical Chemistry A,
2017
18.

Meents A., Dittrich B., Johnas S.K., Thome V., Weckert E.F.
Acta Crystallographica Section B Structural Science,
2008
19.
Dalinger I.L., Shakhnes A.K., Monogarov K.A., Suponitsky K.Y., Sheremetev A.B.
Mendeleev Communications,
2015
20.

Dmitrienko A.O., Karnoukhova V.A., Potemkin A.A., Struchkova M.I., Kryazhevskikh I.A., Suponitsky K.Y.
Chemistry of Heterocyclic Compounds,
2017
21.

Sheremetev A.B., Lyalin B.V., Kozeev A.M., Palysaeva N.V., Struchkova M.I., Suponitsky K.Y.
RSC Advances,
2015
22.

Smirnov A.S., Smirnov S.P., Pivina T.S., Lempert D.B., Maslova L.K.
Russian Chemical Bulletin,
2016
23.

Pozharskii A.F., Kuz'menko V.V., Foces-Foces C., Llamas-Saiz A.L., Claramunt R.M., Sanz D., Elguero J.
Journal of the Chemical Society Perkin Transactions 2,
1994
24.
Sheremetev A.B., Palysaeva N.V., Struchkova M.I., Suponitsky K.Y.
Mendeleev Communications,
2012
25.

Gidaspov A.A., Zalomlenkov V.A., Bakharev V.V., Parfenov V.E., Yurtaev E.V., Struchkova M.I., Palysaeva N.V., Suponitsky K.Y., Lempert D.B., Sheremetev A.B.
RSC Advances,
2016
26.

Gidaspov A.A., Bakharev V., Suponitsky K.Y., Nikitin V.G., Sheremetev A.B.
RSC Advances,
2016