Abstract
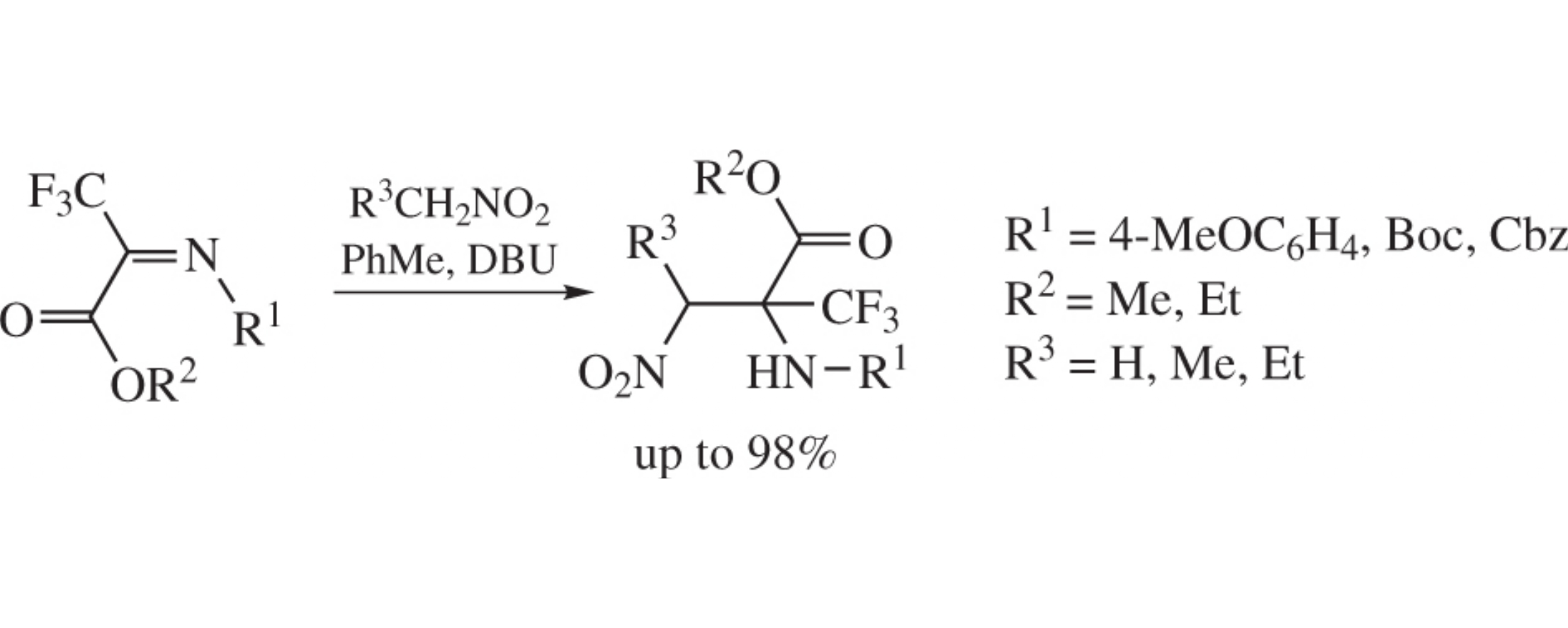
The aza-Henry addition of nitroalkanes at ketimines derived from trifluoropiruvate affords the corresponding α-nitro amines in up to 98% yields.
References
1.

Salwiczek M., Nyakatura E.K., Gerling U.I., Ye S., Koksch B.
Chemical Society Reviews,
2012
2.

Ahrens T., Kohlmann J., Ahrens M., Braun T.
Chemical Reviews,
2014
3.
Handbook of Reagents for Organic Synthesis. Fluorine-Containing Reagents, ed. L. A. Paquette, Wiley, New York, 2007
4.
10.1016/j.mencom.2018.03.006_sbref0005d
Uneyama
Organofluorine Chemistry,
2006
5.
Flourine-Containing Synthons, ed. V. A. Soloshonok, ACS Symp. Ser., vol. 911, American Chemical Society, Washington, DC, 2005
6.
10.1016/j.mencom.2018.03.006_sbref0005f
Kirsch
Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications,
2004
7.
10.1016/j.mencom.2018.03.006_sbref0005g
Hiyama
Organofluorine Compounds,
2000
8.

O’Hagan D.
Journal of Fluorine Chemistry,
2010
9.

Purser S., Moore P.R., Swallow S., Gouverneur V.
Chemical Society Reviews,
2008
10.

Hagmann W.K.
Journal of Medicinal Chemistry,
2008
11.

Müller K., Faeh C., Diederich F.
Science,
2007
12.
10.1016/j.mencom.2018.03.006_sbref0005l
Bégué
Bioorganic and Medicinal Chemistry of Fluorine,
2007
13.

Jeschke P.
ChemBioChem,
2004
14.
K Andra K.
Biochemistry & Analytical Biochemistry,
2015
15.

Sutherland A., Willis C.L.
Natural Product Reports,
2000
16.

Yoder N.C., Kumar K.
Chemical Society Reviews,
2002
17.

Meng H., Kumar K.
Journal of the American Chemical Society,
2007
18.

Tossi A., Sandri L., Giangaspero A.
Biopolymers,
2000
19.

Coates A., Hu Y., Bax R., Page C.
Nature Reviews Drug Discovery,
2002
20.

21.

Hancock R.E., Sahl H.
Nature Biotechnology,
2006
22.

Kutovaya I.V., Shmatova O.I., Tkachuk V.M., Melnichenko N.V., Vovk M.V., Nenajdenko V.G.
European Journal of Organic Chemistry,
2015
23.
Kutovaya I.V., Shmatova O.I., Tkachuk V.M., Sukach V.A., Vovk M.V., Nenajdenko V.G.
Mendeleev Communications,
2016