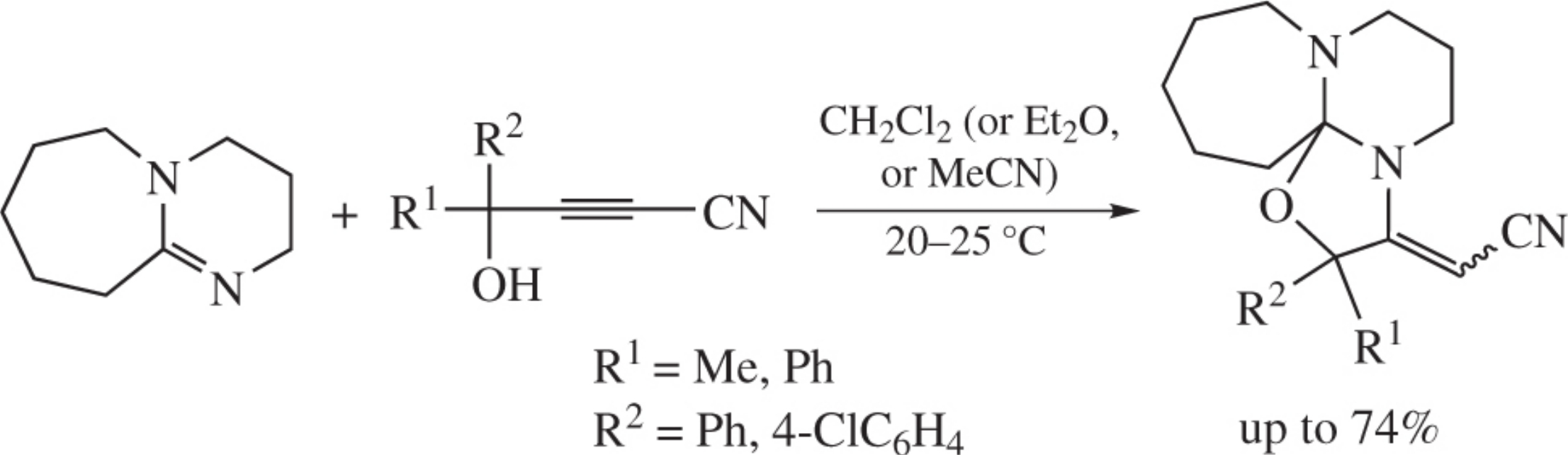
Abstract
Organic superbase DBU, 1,8-diazabicyclo[5.4.0]undec-7-ene, readily annulated with aromatic cyanopropargylic alcohols nder mild conditions (CH2Cl2, 20–25 °C, 30 min) to afford new cyanomethylideneperhydro[1,3]oxazolo[2’,3’:2,3]pyrimido-,2-a]azepines.
References
1.
2.
Superbases for Organic Synthesis: Guanidines, Amidines, Phosphazenes and Related Organocatalysts, ed. T. Ishikawa, John Wiley & Sons, Chichester, 2009
3.

Taylor J.E., Bull S.D., Williams J.M.
Chemical Society Reviews,
2012
4.
![1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU): A Versatile Reagent in Organic Synthesis](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
Nand B., Khanna G., Chaudhary A., Lumb A., M. Khurana J.
Current Organic Chemistry,
2015
5.
![Three-membered rings. 8. Reaction of 1-halocyclopropane 1,2-diesters with 1,8-diazabicyclo[5.4.0]undec-7-ene. Unexpected products](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
McCoy L.L., Mal D.
Journal of Organic Chemistry,
1981
6.

Juneja T.R., Garg D.K., Schäfer W.
Tetrahedron,
1982
7.
![Unusual reaction of 1,8‐diazabicyelo‐[5.4.0]undec‐7‐ene with diethyl maleate](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Perbost M., Lucas M., Chavis C., Imbach J.
Journal of Heterocyclic Chemistry,
1993
8.

Lammers H., Cohen-Fernandes P., Habraken C.L.
Tetrahedron,
1994
9.
![1,8-Diazabicyclo[5.4.0]undec-7-ene as a difunctional nucleophile](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Chambers R.D., Roche A.J., Batsanov A.S., Howard J.A.
Journal of the Chemical Society Chemical Communications,
1994
10.
![Nucleophilic reaction of 1,8-diazabicyclo[5.4.0]undec-7-ene and 1,5-diazabicyclo[4.3.0]non-5-ene with methyl pheophorbide a. Unexpected products](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
11.

Johnson M.G., Foglesong R.J.
Tetrahedron Letters,
1997
12.
![Reaction of 1,8-diazabicyclo[5.4.0]undec-8-ene with methyl 3,5-dinitrobenzoate and 1,3,5-trinitrobenzene](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Sutherland J.K.
Chemical Communications,
1997
13.

Kraft A.
Journal of the Chemical Society Perkin Transactions 1,
1999
14.

Lönnqvist J.S., Jalander L.F.
Journal of Chemical Research,
2000
15.
10.1016/j.mencom.2018.03.004_sbref0010k
Im
Bull. Korean Chem. Soc.,
2001
16.

Bulman Page P.C., Vahedi H., Bethell D., Barkley J.V.
Synthetic Communications,
2003
17.

Gierczyk B., Schroeder G., Brzezinski B.
Journal of Organic Chemistry,
2003
18.
![The reaction of 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) with carbon dioxide.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Heldebrant D.J., Jessop P.G., Thomas C.A., Eckert C.A., Liotta C.L.
Journal of Organic Chemistry,
2005
19.

Gryko D.T., Piechowska J., Tasior M., Waluk J., Orzanowska G.
Organic Letters,
2006
20.

Nirmala R., Ponpandian T., Venkatraman B.R., Rajagopal S.
Tetrahedron Letters,
2013
21.

Mishra R., Panini P., Sankar J.
Organic Letters,
2014
22.

Baravkar S.B., Roy A., Gawade R.L., Puranik V.G., Sanjayan G.J.
Synthetic Communications,
2014
23.

Poronik Y.M., Gryko D.T.
Chemical Communications,
2014
24.

Chen J., Natte K., Wu X.
Tetrahedron Letters,
2015
25.
![Michael-type addition of 1,8-diazabicyclo[5.4.0]undec-7-ene to dimethyl acetylenedicarboxylate](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Ma L., Dolphin D.
Journal of the Chemical Society Chemical Communications,
1995
26.
L.-Q. Nie, Z.-J. Shi and W.-Q. Cao, Bopuxue Zazhi, 2002, 19, 377 (in Chinese)
27.
Shi Y.
Jixie Gongcheng Xuebao/Chinese Journal of Mechanical Engineering,
2003
28.
Z.-J. Shi, L.-Q. Nie and W.-Q. Cao, Fenxi Kexue Xuebao, 2004, 20, 586 (in Chinese).
29.

Trofimov B.A., Shemyakina O.A., Mal'kina A.G., Stepanov A.V., Volostnykh O.G., Ushakov I.A., Vashchenko A.V.
European Journal of Organic Chemistry,
2016
30.
T. P. Cripe, M. A. Currier and I. Orienti, Patent WO 2015/103005 A1, 2015
31.

Orienti I., Falconi M., Teti G., Currier M.A., Wang J., Phelps M., Cripe T.P.
Pharmaceutical Research,
2016
32.

Parhizkar G., Khosropour A.R., Mohammadpoor-Baltork I., Parhizkar E., Rudbari H.A.
Tetrahedron,
2017
33.

Drandarov K., Guggisberg A., Hesse M.
Helvetica Chimica Acta,
1999
34.
10.1016/j.mencom.2018.03.004_sbref0035b
Suleiman
Der Pharma Chem.,
2015
35.
K. J. Schmalzi, S. C. Sharma and R. I. Christopherson, Patent US 4873228 A, 1989
36.

Liu S., Jin J., Chen C., Liu J., Li J., Wang F., Jiang Z., Hu J., Gao Z., Yao F., You X., Si S., Sun C.
Journal of Antibiotics,
2013
37.
10.1016/j.mencom.2018.03.004_sbref0040c
Ghorab
Biomed. Res.,
2015
38.
10.1016/j.mencom.2018.03.004_sbref0045a
Khan
Pharmazie,
2002
39.

Sondhi S.M., Singh N., Johar M., Kumar A.
Bioorganic and Medicinal Chemistry,
2005
40.
10.1016/j.mencom.2018.03.004_sbref0045c
El-Sayed
Bull. Pharm. Sci.,
2006
41.
J. Kokosi, I. Hermecz, Z. Meszaros, G. Szasz, A. Horvath, T. Breining and L. Vasvari, Patent CA 1143729 A, 1983
42.
J. Kokosi, I. Hermecz, Z. Meszaros, G. Szasz, L. Vasvari, A. Horvath and T. Breining, Patent US 4404205 A, 1983.
43.
A. Nagaoka and T. Naka, Patent EP 0383465 A3, 1991
44.
10.1016/j.mencom.2018.03.004_sbref0055b
Herold
Acta Pol. Pharm.,
2004
45.
10.1016/j.mencom.2018.03.004_sbref0060a
Miller
in Selective Organic Transformation,
1970
46.
10.1016/j.mencom.2018.03.004_sbref0060b
Dickstein
in The Carbon–Carbon Triple Bond,
1978
47.

Landor S.R., Demetriou B., Grzeskowiak R., Pavey D.F.
Journal of Organometallic Chemistry,
1975
48.

Mal’kina A.G., Sokolyanskaya L.V., Kudyakova R.N., Sinegovskaya L.M., Albanov A.I., Shemyakina O.A., Trofimov B.A.
Russian Journal of Organic Chemistry,
2005