Abstract
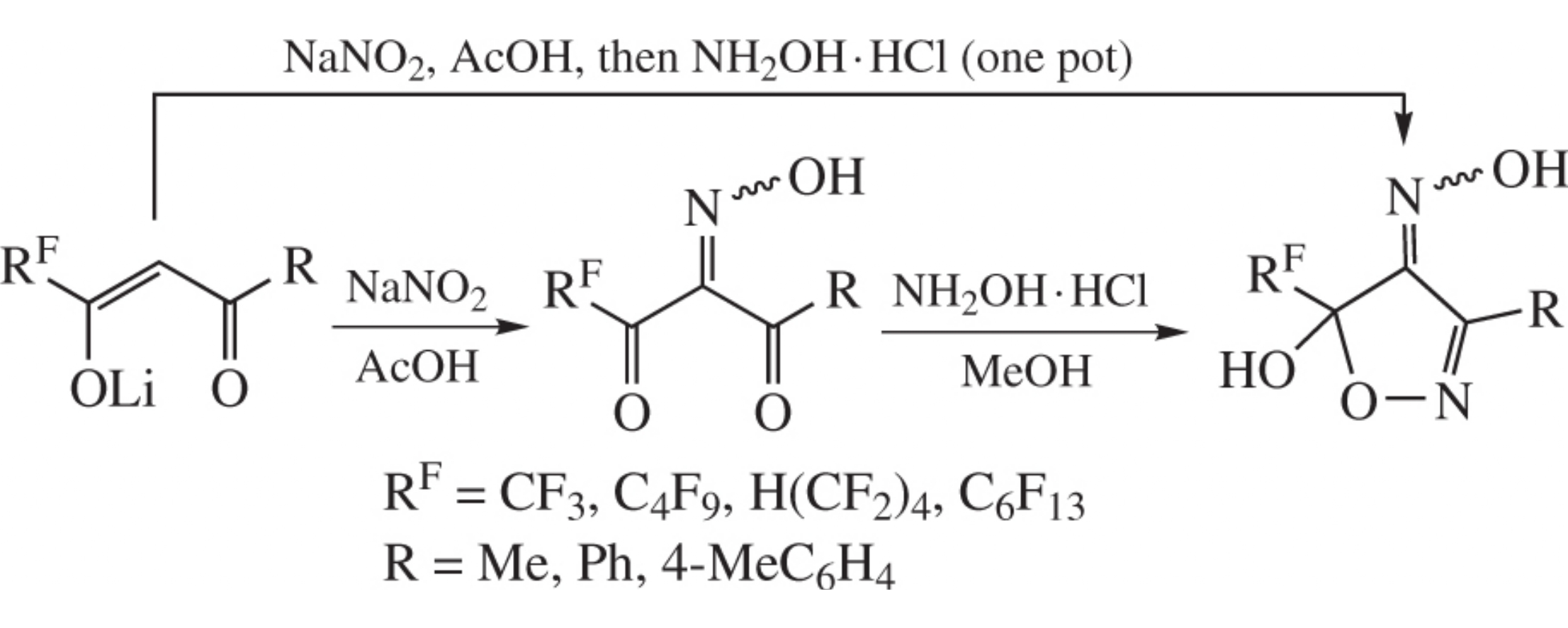
5-Hydroxy-5-(polyfluoroalkyl)isoxazol-4(5H)-one oximes have been obtained by nitrosation of lithium 3-polyfluoroalkyl-1,3-diketonates followed by treatment of intermediate 3-(poly-fluoroalkyl)propane-1,2,3-trione 2-oximes with hydroxylamine hydrochloride. The one-pot protocol comprising both stages has been elaborated.
References
1.

Akhrem A.A., Lakhvich F.A., Khripach V.A.
Chemistry of Heterocyclic Compounds,
1981
2.

Lakhvich F.A., Koroleva E.V., Akhrem A.A.
Chemistry of Heterocyclic Compounds,
1989
3.

Pinho e Melo T.
Current Organic Chemistry,
2005
4.

Shi W., Hu J., Bao N., Li D., Chen L., Sun J.
Bioorganic and Medicinal Chemistry Letters,
2017
5.

Kumar V., Kaur K.
Journal of Fluorine Chemistry,
2015
6.

Padmaja A., Payani T., Reddy G.D., Padmavathi V.
European Journal of Medicinal Chemistry,
2009
7.

Padmaja A., Rajasekhar C., Muralikrishna A., Padmavathi V.
European Journal of Medicinal Chemistry,
2011
8.
![Reaction of naphthoquinones with substituted nitromethanes. Facile synthesis and antifungal activity of naphtho[2,3-d]isoxazole-4,9-diones.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Santos M.M., Faria N., Iley J., Coles S.J., Hursthouse M.B., Martins M.L., Moreira R.
Bioorganic and Medicinal Chemistry Letters,
2010
9.
![4-[5-Methyl-3-phenylisoxazol-4-yl]- benzenesulfonamide, Valdecoxib: A Potent and Selective Inhibitor of COX-2](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Talley J.J., Brown D.L., Carter J.S., Graneto M.J., Koboldt C.M., Masferrer J.L., Perkins W.E., Rogers R.S., Shaffer A.F., Zhang Y.Y., Zweifel B.S., Seibert K.
Journal of Medicinal Chemistry,
2000
10.

Kamal A., Bharathi E.V., Reddy J.S., Ramaiah M.J., Dastagiri D., Reddy M.K., Viswanath A., Reddy T.L., Shaik T.B., Pushpavalli S.N., Bhadra M.P.
European Journal of Medicinal Chemistry,
2011
11.

Diana P., Carbone A., Barraja P., Kelter G., Fiebig H., Cirrincione G.
Bioorganic and Medicinal Chemistry,
2010
12.
Fluorine Compounds. Synthesis and Application, ed. N. Ishikawa, Kodansha, Tokyo, 1986.
13.
10.1016/j.mencom.2018.03.003_bib0065
Kirsch
Modern Fluoroorganic Chemistry: Synthesis, Reactivity Applications,
2013
14.
Filyakova V.I., Boltachova N.S., Pervova M.G., Palysaeva N.V., Slepukhin P.A., Sheremetev A.B., Charushin V.N.
Mendeleev Communications,
2017
15.

Mikhaleva A.I., Zaitsev A.B., Trofimov B.A.
Russian Chemical Reviews,
2006
16.

Fruttero R., Calvino R., Ferrarotti B., Gasco A., Aime S., Gobetto R., Chiari G., Calestani G.
Journal of the Chemical Society Perkin Transactions 2,
1987
17.

Rai G., Thomas C.J., Leister W., Maloney D.J.
Tetrahedron Letters,
2009
18.

Ibrahim S.S.
Industrial & Engineering Chemistry Research,
2000
19.

Aggarwal R., Bansal A., Mittal A.
Journal of Fluorine Chemistry,
2013
20.

Crookes M.J., Roy P., Williams D.L.
Journal of the Chemical Society Perkin Transactions 2,
1989
21.

Saloutin V.I., Burgart Y.V., Skryabina Z.E., Kuzueva O.G.
Journal of Fluorine Chemistry,
1997
22.
10.1016/j.mencom.2018.03.003_bib0110
Lin-Vien
The Handbook of Infrared and Raman Characteristic Frequencies of Organic Molecules,
1991
23.

Kumar V., Aggarwal R., Singh S.P.
Journal of Fluorine Chemistry,
2006
24.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
25.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
26.
10.1016/j.mencom.2018.03.003_bib0130
Bourhis
Acta Crystallogr.,
2015