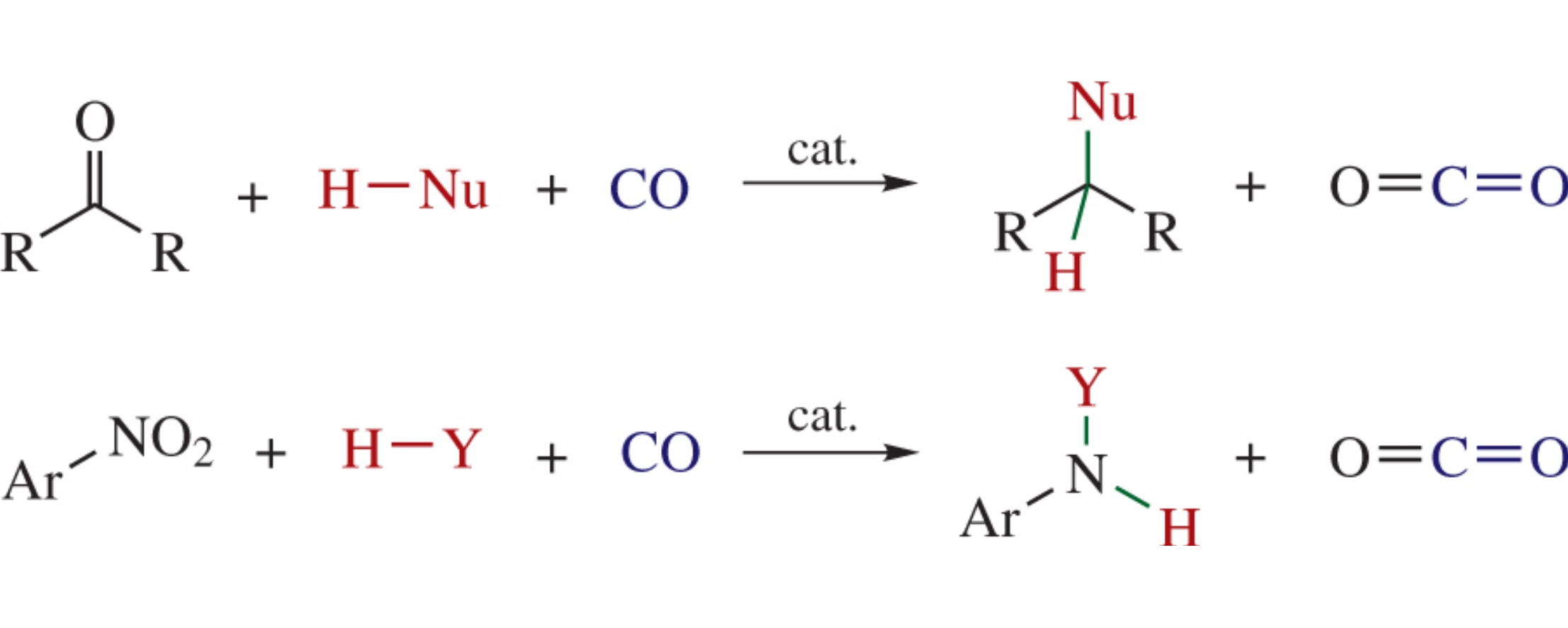
Abstract
Carbon monoxide as an example of reducing agent, in contrast to classical reducing agents (hydrogen and metal hydrides), can provide very high atom precision for reductive addition of substrates with various functional groups. This enables synthesis of new compounds with unique structures and properties.
References
1.

Aavula S.K., Chikkulapalli A., Hanumanthappa N., Jyothi I., Vinod Kumar C.H., Manjunatha S.G.
Tetrahedron Letters,
2013
2.

Ananikov V.P., Khemchyan L.L., Ivanova Y.V., Bukhtiyarov V.I., Sorokin A.M., Prosvirin I.P., Vatsadze S.Z., Medved'ko A.V., Nuriev V.N., Dilman A.D., Levin V.V., Koptyug I.V., Kovtunov K.V., Zhivonitko V.V., Likholobov V.A., et. al.
Russian Chemical Reviews,
2014
3.

4.

Tafesh A.M., Weiguny J.
Chemical Reviews,
1996
5.

Ambrosi A., Denmark S.E.
Angewandte Chemie - International Edition,
2016
6.

Chusov D., List B.
Angewandte Chemie - International Edition,
2014
7.

Kolesnikov P.N., Yagafarov N.Z., Usanov D.L., Maleev V.I., Chusov D.
Organic Letters,
2014
8.

Yagafarov N.Z., Usanov D.L., Moskovets A.P., Kagramanov N.D., Maleev V.I., Chusov D.
ChemCatChem,
2015
9.

Afanasyev O.I., Tsygankov A.A., Usanov D.L., Perekalin D.S., Shvydkiy N.V., Maleev V.I., Kudinov A.R., Chusov D.
ACS Catalysis,
2016
10.

Moskovets A.P., Usanov D.L., Afanasyev O.I., Fastovskiy V.A., Molotkov A.P., Muratov K.M., Denisov G.L., Zlotskii S.S., Smol'yakov A.F., Loginov D.A., Chusov D.
Organic and Biomolecular Chemistry,
2017
11.
10.1016/j.mencom.2018.03.001_bib0055
Runikhina
J. Organomet. Chem.,
2017
12.
![Iridium halide complexes [1,1-X2-8-SMe2-1,2,8-IrC2B9H10]2 (X = Cl, Br, I): Synthesis, reactivity and catalytic activity](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Molotkov A.P., Vinogradov M.M., Moskovets A.P., Chusova O., Timofeev S.V., Fastovskiy V.A., Nelyubina Y.V., Pavlov A.A., Chusov D.A., Loginov D.A.
European Journal of Inorganic Chemistry,
2017
13.

Abdel-Magid A.F., Carson K.G., Harris B.D., Maryanoff C.A., Shah R.D.
Journal of Organic Chemistry,
1996
14.

Yagafarov N.Z., Kolesnikov P.N., Usanov D.L., Novikov V.V., Nelyubina Y.V., Chusov D.
Chemical Communications,
2016
15.

Shvydkiy N.V., Trifonova E.A., Shved A.M., Nelyubina Y.V., Chusov D., Perekalin D.S., Kudinov A.R.
Organometallics,
2016
16.
10.1016/j.mencom.2018.03.001_bib0080
Tsygankov
Synlett,
2017
17.

Pototskiy R.A., Afanasyev O.I., Nelyubina Y.V., Chusov D., Kudinov A.R., Perekalin D.S.
Journal of Organometallic Chemistry,
2017
18.

Kolesnikov P.N., Usanov D.L., Barablina E.A., Maleev V.I., Chusov D.
Organic Letters,
2014
19.

Afanasyev O.I., Tsygankov A.A., Usanov D.L., Chusov D.
Organic Letters,
2016
20.

Afanasyev O., Tsygankov A., Usanov D., Perekalin D., Samoylova A., Chusov D.
Synthesis,
2017
21.

Kolesnikov P.N., Usanov D.L., Muratov K.M., Chusov D.
Organic Letters,
2017
22.

Yagafarov N.Z., Muratov K.M., Biriukov K., Usanov D.L., Chusova O., Perekalin D.S., Chusov D.
European Journal of Organic Chemistry,
2018
23.

Cenini S., Crotti C., Pizzotti M., Porta F.
Journal of Organic Chemistry,
1988
24.

Cenini S., Pizzotti M., Crotti C., Porta F., Monica G.L.
Journal of the Chemical Society Chemical Communications,
1984
25.

Ragaini F., Cenini S.
Journal of Molecular Catalysis A Chemical,
2000
26.

Ragaini F., Ghitti A., Cenini S.
Organometallics,
1999
27.

Cenini S., Pizzotti M., Crotti C., Ragaini F., Porta F.
Journal of Molecular Catalysis,
1988
28.

Liu C., Cheng C.
Journal of Organometallic Chemistry,
1991
29.

Mukherjee D.K., Palit B.K., Saha C.R.
Journal of Molecular Catalysis,
1994
30.

Reductive carbonylation of nitrobenzene to phenylurethane catalyzed by Ru(III)-schiff base complexes
Khan M.M., Halligudi S.B., Shukla S., Shaikh Z.A.
Journal of Molecular Catalysis,
1990
31.

Khan M.M., Halligudi S.B., Shukla S., Shaikh Z.A.
Journal of Molecular Catalysis,
1990
32.

Halligudi S.B., Bhatt K.N., Khan N.H., Khan M.M.
Journal of Molecular Catalysis,
1992
33.

Gorbunova L.V., Knyazeva I.L., Nefedov B.K., Khoshdurdyev K.O., Manov-Yuvenskii V.I.
Russian Chemical Bulletin,
1981
34.

Cenini S., Ragaini F., Pizzotti M., Porta F., Mestroni G., Alessio E.
Journal of Molecular Catalysis,
1991
35.

Islam S.M., Mal D., Palit B.K., Saha C.R.
Journal of Molecular Catalysis A Chemical,
1999
36.
![[Rh(CO)4]−, [Rh5(CO)15]−, and bimetallic clusters as catalysts for the carbonylation of nitrobenzene to methyl phenylcarbamate](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ragaini F., Cenini S., Fumagalli A., Crotti C.
Journal of Organometallic Chemistry,
1992
37.
![Promotion of the [PPN][Rh(CO)4]-catalysed carbonylation of nitrobenzene by 2-hydroxypyridine and related molecules: an apparent bifunctional activation](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ragaini F., Gallo E., Cenini S.
Journal of Organometallic Chemistry,
2000
38.

Kim J., Kim D., Cheong M., Kim H., Mukherjee D.K.
Bulletin of the Korean Chemical Society,
2010
39.

Alessio E., Mestroni G.
Journal of Molecular Catalysis,
1984
40.

Alessio E., Mestroni G.
Journal of Organometallic Chemistry,
1985
41.

Drent E.
Pure and Applied Chemistry,
1990
42.
![Reductive carbonylation of nitroaromatic compounds to urethanes catalyzed by [Pd(1,10-phenanthroline)2][PF6]2 and related complexes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bontempi A., Alessio E., Chanos G., Mestroni G.
Journal of Molecular Catalysis,
1987
43.

Wehman P., Borst L., Kamer P.C., Leeuwen P.W.
Chemische Berichte,
1997
44.

Wehman P., Dol G.C., Moorman E.R., Kamer P.C., van Leeuwen P.W., Fraanje J., Goubitz K.
Organometallics,
1994
45.

Wehman P., Kaasjager V.E., Hartl F., Kamer P.C., van Leeuwen P.W., Fraanje J., Goubitz K.
Organometallics,
1995
46.

Wehman P., Borst L., Kamer P.C., van Leeuwen P.W.
Journal of Molecular Catalysis A Chemical,
1996
47.

Wehman P., Kamer P.C., van Leeuwen P.W.
Chemical Communications,
1996
48.

Santi R., Romano A.M., Panella F., Santini C.
Journal of Molecular Catalysis A Chemical,
1997
49.

Gasperini M.
Journal of Molecular Catalysis A Chemical,
2003
50.

Santi R., Romano A.M., Panella F., Mestroni G., Sessanti O. Santi A.
Journal of Molecular Catalysis A Chemical,
1999
51.

Shi F., He Y., Li D., Ma Y., Zhang Q., Deng Y.
Journal of Molecular Catalysis A Chemical,
2006
52.

Yang Q., Robertson A., Alper H.
Organic Letters,
2008
53.

Ragaini F., Cognolato C., Gasperini M., Cenini S.
Angewandte Chemie - International Edition,
2003
54.

Ragaini F., Gasperini M., Cenini S.
Advanced Synthesis and Catalysis,
2004
55.

Fluoride effect on the palladium–phenanthroline catalyzed carbonylation of nitroarenes to carbamates
Gasperini M., Ragaini F., Cenini S., Gallo E., Fantauzzi S.
Applied Organometallic Chemistry,
2007
56.

Ferretti F., Ragaini F., Lariccia R., Gallo E., Cenini S.
Organometallics,
2010
57.

Ferretti F., Gallo E., Ragaini F.
Journal of Organometallic Chemistry,
2014
58.

Gasperini M., Ragaini F., Cazzaniga C., Cenini S.
Advanced Synthesis and Catalysis,
2005
59.

Valli V.L., Alper H.
Journal of the American Chemical Society,
1993
60.

Choudary B.M., Rao K.K., Pirozhkov S.D., Lapidus A.L.
Journal of Molecular Catalysis,
1994
61.

Reddy N.P., Masdeu A.M., El Ali B., Alper H.
Journal of the Chemical Society Chemical Communications,
1994
62.

Wehman P., van Donge H.M., Hagos A., Kamer P.C., van Leeuwen P.W.
Journal of Organometallic Chemistry,
1997
63.

Lee J.S., Woo J.W., Lee C.W., Hong K.S., Yeo J.K.
Korean Journal of Chemical Engineering,
1990
64.

Izumi Y., Satoh Y., Urabe K.
Chemistry Letters,
1990
65.

Izumi Y., Satoh Y., Kondoh H., Urabe K.
Journal of Molecular Catalysis,
1992
66.

Lapidus A.L., Pirozhkov S.D., Tumanova A.R., Dolldze A.V., Yukhimenko A.M.
Russian Chemical Bulletin,
1991
67.

Skupińska J., Karpińska M.
Journal of Molecular Catalysis A Chemical,
2000
68.

Skupińska J., Karpińska M.
Applied Catalysis A: General,
2004
69.

Skupińska J., Karpińska M., Wachowski L., Hofman A.M.
Reaction Kinetics and Catalysis Letters,
2004
70.
10.1016/j.mencom.2018.03.001_bib0350
Skupińska
Cent. Eur. J. Chem.,
2005
71.

Krogul A., Skupińska J., Litwinienko G.
Journal of Molecular Catalysis A Chemical,
2011
72.

Krogul A., Skupińska J., Litwinienko G.
Journal of Molecular Catalysis A Chemical,
2014
73.

Watanabe Y., Tsuji Y., Suzuki N.
Chemistry Letters,
1982
74.

Watanabe Y., Tsuji Y., Takeuchi R., Suzuki N.
Bulletin of the Chemical Society of Japan,
1983
75.

Watanabe Y., Tsuji Y., Takeuchi R.
Bulletin of the Chemical Society of Japan,
1984
76.

Zhang X., Jing H.
Journal of Molecular Catalysis A Chemical,
2009
77.

Chen L., Mei F., Li G., Xiang Y.
Kinetics and Catalysis,
2010
78.

Zhang J., Li Y., Zhao B.
Acta Chimica Sinica,
2012
79.

Dieck H.A., Laine R.M., Heck R.F.
Journal of Organic Chemistry,
1975
80.

Lee J.S., Lee C.W., Lee S.M., Park K.H.
Journal of Molecular Catalysis,
1990
81.

Oh J.S., Lee S.M., Yeo J.K., Lee C.W., Lee J.S.
Industrial & Engineering Chemistry Research,
1991
82.

Kim K.D., Lee S.M., Cho N.S., Oh J.S., Lee C.W., Lee J.S.
Journal of Molecular Catalysis,
1992
83.

Sang M.L., Nag S.C., Kye D.K., Jae S.O., Chul W.L., Jae S.L.
Journal of Molecular Catalysis,
1992
84.

Lee C.W., Lee S.M., Oh J.S., Lee J.S.
Catalysis Letters,
1993
85.

Lee C.W., Lee J.S., Lee S.M., Kim K.D., Cho N.S., Oh J.S.
Journal of Molecular Catalysis,
1993
86.

Vavasori A., Ronchin L.
Pure and Applied Chemistry,
2012
87.

Tsuji Y., Takeuchi R., Watanabe Y.
Journal of Organometallic Chemistry,
1985
88.

Bassoli A., Rindone B., Cenini S.
Journal of Molecular Catalysis,
1991
89.

Bassoli A., Rindone B., Tollari S., Cenini S., Crotti C.
Journal of Molecular Catalysis,
1990
90.

Islam S.M., Ghosh K., Roy A.S., Molla R.A., Salam N., Chatterjee T., Iqubal M.A.
Journal of Organometallic Chemistry,
2014
91.

Mukherjee D.
Journal of Catalysis,
2002
92.

Yang Y., Lu S.
Tetrahedron Letters,
1999
93.

Ling G., Chen J., Lu S.
Journal of Molecular Catalysis A Chemical,
2003
94.

Chen J., Ling G., Lu S.
European Journal of Organic Chemistry,
2003
95.

Ling G., Chen J., Lu S.
Journal of Chemical Research,
2003
96.

97.

Chen J., Ling G., Yu Z., Wu S., Zhao X., Wu X., Lu S.
Advanced Synthesis and Catalysis,
2004
98.

Cenini S., Ragaini F., Tollari S., Paone D.
Journal of the American Chemical Society,
1996
99.

Ragaini F., Cenini S., Tollari S., Tummolillo G., Beltrami R.
Organometallics,
1999
100.

Ragaini F., Cenini S., Turra F., Caselli A.
Tetrahedron,
2004
101.

Srivastava R.S., Nicholas K.M.
Chemical Communications,
1998
102.
![A Cyclic Carbamoyl Complex Is a Resting State in Allylic Aminations Catalyzed by [Cp*Fe(CO)2]2](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Kolel-Veetil M.K., Khan M.A., Nicholas K.M.
Organometallics,
2000
103.

Srivastava R.S., Kolel-Veetil M., Nicholas K.M.
Tetrahedron Letters,
2002
104.

Iqbal A.F.
Journal of Organic Chemistry,
1972
105.

Akazome M., Kondo T., Watanabe Y.
Chemistry Letters,
1992
106.

Kmiecik J.E.
Journal of Organic Chemistry,
1965
107.

Pizzotti M., Cenini S., Quici S., Tollari S.
Journal of the Chemical Society Perkin Transactions 2,
1994
108.

Smitrovich J.H., Davies I.W.
Organic Letters,
2004
109.

Crotti C., Cenini S., Rindone B., Tollari S., Demartin F.
Journal of the Chemical Society Chemical Communications,
1986
110.

Crotti C., Cenini S., Todeschini R., Tollari S.
Journal of the Chemical Society Faraday Transactions,
1991
111.

Bassoli A., Cenini S., Farina F., Orlandi M., Rindone B.
Journal of Molecular Catalysis,
1994
112.

Akazome M., Kondo T., Watanabe Y.
Journal of Organic Chemistry,
1994
113.

Ragaini F., Sportiello P., Cenini S.
Journal of Organometallic Chemistry,
1999
114.

Tollari S., Cenini S., Crotti C., Gianella E.
Journal of Molecular Catalysis,
1994
115.
![Transition metal-mediated N-heterocyclisation reactions. Synthesis of 2-phenylindole by reduction by CO of 2-nitrostilbene catalysed by [Rh(CO)4]−](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ragaini F., Tollari S., Cenini S., Bettetini E.
Journal of Molecular Catalysis A Chemical,
1996
116.

Söderberg B.C., Shriver J.A.
Journal of Organic Chemistry,
1997
117.

Nishiyama Y., Maema R., Ohno K., Hirose M., Sonoda N.
Tetrahedron Letters,
1999
118.

Davies I.W., Smitrovich J.H., Sidler R., Qu C., Gresham V., Bazaral C.
Tetrahedron,
2005
119.

Söderberg B.C., Chisnell A.C., O'Nei S.N., Shriver J.A.
Journal of Organic Chemistry,
1999
120.

Hsieh T.H., Dong V.M.
Tetrahedron,
2009
121.

Tollari S., Cenini S., Rossi A., Palmisano G.
Journal of Molecular Catalysis A Chemical,
1998
122.

Söderberg B.C., Rector S.R., O'Neil S.N.
Tetrahedron Letters,
1999
123.

Söderberg B.C., Hubbard J.W., Rector S.R., O'Neil S.N.
Tetrahedron,
2005
124.

Clawson R.W., Deavers R.E., Akhmedov N.G., Söderberg B.C.
Tetrahedron,
2006
125.

Söderberg B., Banini S., Turner M., Minter A., Arrington A.
Synthesis,
2008
126.

Kuethe J.T., Davies I.W.
Tetrahedron,
2006
127.

Dacko C.A., Akhmedov N.G., Söderberg B.C.
Tetrahedron Asymmetry,
2008
128.
![Double Palladium Catalyzed Reductive Cyclizations. Synthesis of 2,2′-, 2,3′-, and 3,3′-Bi-1H-indoles, Indolo[3,2-b]indoles, and Indolo[2,3-b]indoles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ansari N.H., Dacko C.A., Akhmedov N.G., Söderberg B.C.
Journal of Organic Chemistry,
2016
129.

Ansari N.H., Taylor M.C., Söderberg B.C.
Tetrahedron Letters,
2017
130.

Zhang Y., Hubbard J.W., Akhmedov N.G., Petersen J.L., Söderberg B.C.
Journal of Organic Chemistry,
2015
131.

Gorugantula S.P., Carrero-Martínez G.M., Dantale S.W., Söderberg B.C.
Tetrahedron,
2010
132.
10.1016/j.mencom.2018.03.001_bib0660
Okuro
J. Org. Chem.,
2011
133.

El-Atawy M.A., Ferretti F., Ragaini F.
European Journal of Organic Chemistry,
2017
134.

Ferretti F., EL-Atawy M.A., Muto S., Hagar M., Gallo E., Ragaini F.
European Journal of Organic Chemistry,
2015
135.

Banini S.R., Turner M.R., Cummings M.M., Söderberg B.C.
Tetrahedron,
2011
136.

Penoni A., Nicholas K.M.
Chemical Communications,
2002
137.

Penoni A., Volkmann J., Nicholas K.M.
Organic Letters,
2002
138.

Ragaini F., Ventriglia F., Hagar M., Fantauzzi S., Cenini S.
European Journal of Organic Chemistry,
2009
139.

Ragaini F., Rapetti A., Visentin E., Monzani M., Caselli A., Cenini S.
Journal of Organic Chemistry,
2006
140.

Pizzotti M., Cenini S., Psaro R., Costanzi S.
Journal of Molecular Catalysis,
1990
141.

Crotti C., Cenini S., Ragaini F., Porta F., Tollari S.
Journal of Molecular Catalysis,
1992
142.

Hubbard J.W., Piegols A.M., Söderberg B.C.
Tetrahedron,
2007
143.

Akazome M., Kondo T., Watanabe Y.
Journal of the Chemical Society Chemical Communications,
1991
144.

Okuro K., Gurnham J., Alper H.
Tetrahedron Letters,
2012
145.

Watanabe Y., Yamamoto J., Akazome M., Kondo T., Mitsudo T.
Journal of Organic Chemistry,
1995
146.

Ragaini F., Cenini S., Borsani E., Dompé M., Gallo E., Moret M.
Organometallics,
2001
147.

Ragaini F., Cenini S., Brignoli D., Gasperini M., Gallo E.
Journal of Organic Chemistry,
2002
148.

O'Dell D.K., Nicholas K.M.
Journal of Organic Chemistry,
2003
149.

O'Dell D.K., Nicholas K.M.
Tetrahedron,
2003
150.

Tollari S., Penoni A., Cenini S.
Journal of Molecular Catalysis A Chemical,
2000
151.

Söderberg B.C., Wallace J.M., Tamariz J.
Organic Letters,
2002
152.

Zhou F., Wang D., Driver T.G.
Advanced Synthesis and Catalysis,
2015
153.

Afanasyev O.I., Usanov D.L., Chusov D.
Organic and Biomolecular Chemistry,
2017
154.

Formenti D., Ferretti F., Ragaini F.
ChemCatChem,
2017