Abstract
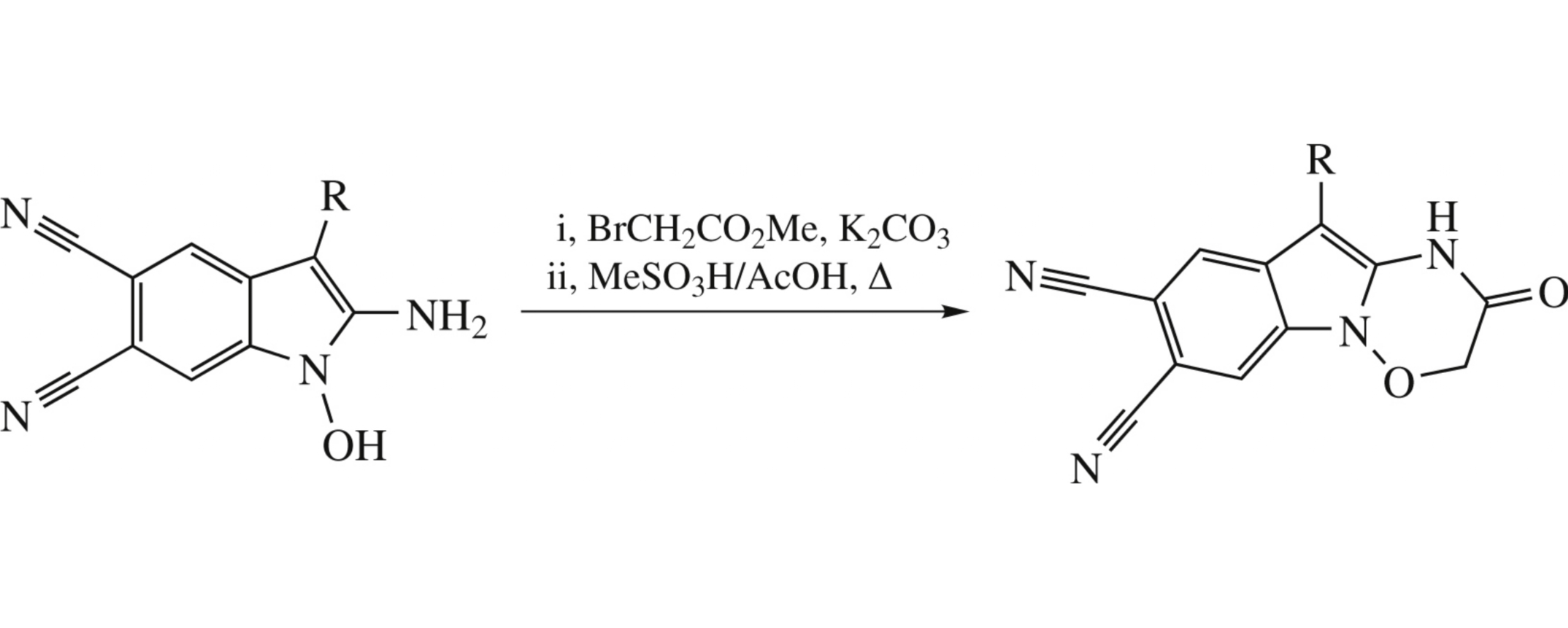
10-Aryl-2-oxo-2,3-dihydro-1H-[1,2,4]oxadiazino[2,3-a]indole-7,8-dicarbonitriles were obtained in two steps from the corresponding 2-amino-1-hydroxyindoles and methyl bromoacetate.
References
1.

Belley M., Sauer E., Beaudoin D., Duspara P., Trimble L.A., Dubé P.
Tetrahedron Letters,
2006
2.

Belley M., Beaudoin D., Duspara P., Sauer E., St-Pierre G., Trimble L.
Synlett,
2007
3.

Filimonov S.I., Chirkova Z.V., Sharunov V.S., Abramov I.G., Stashina G.A., Firgang S.I., Suponitsky K.Y.
Chemistry of Heterocyclic Compounds,
2012
4.

Xing R., Tian Q., Liu Q., Li Y.
Chinese Journal of Chemistry,
2012
5.

Liu T., Zhu H., Luo D., Yan S., Lin J.
Molecules,
2016
6.

Argade N., Vaidya S.
Synthesis,
2016
7.

Ishikura M., Itoh T., Abe T., Choshi T.
Heterocycles,
2016
8.
![Benzo[g]indoles](/storage/images/resized/9Mus3KG1Tkd7Bwaurt8H3RwWh0CxRlGoO6ng9UK1_small_thumb.webp)
Pozharskii A.F., Kachalkina S.G., Gulevskaya A.V., Filatova E.A.
Russian Chemical Reviews,
2017
9.
Gvozdev V.D., Shavrin K.N., Baskir E.G., Egorov M.P., Nefedov O.M.
Mendeleev Communications,
2017
10.

Chirkova Z.V., Kabanova M.V., Filimonov S.I., Abramov I.G., Petzer A., Petzer J.P., Suponitsky K.Y.
Bioorganic and Medicinal Chemistry Letters,
2016
11.

Berkowitz P.T., Long R.A., Dea P., Robins R.K., Matthews T.R.
Journal of Medicinal Chemistry,
1977
12.

N. Weller H., V. Miller A., E. J. Dickinson K., Anders Hedberg S., L. Delaney C., P. Serafino R., Moreland S.
Heterocycles,
1993
13.

Ürögdi L., Kisfaludy L., Patthy Á., Vezér C.
Journal of Heterocyclic Chemistry,
1989
14.

Trindade A.F., Bode J.W.
Organic Letters,
2016
15.
![Base-controlled [3+3] cycloaddition of isoquinoline N-oxides with azaoxyallyl cations](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
An Y., Xia H., Wu J.
Chemical Communications,
2016
16.
![Facile access to novel 1,2,4-oxadiazinan-5-ones via [3 + 3] cycloaddition of in situ generated azaoxyallyl cations with nitrones](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Zhao H., Zhao Y., Liu Y., Zhao L., Feng N., Pang H., Chen X., Song X., Du J.
RSC Advances,
2017
17.

Chirkova Z.V., Filimonov S.I.
Russian Chemical Bulletin,
2017
18.
![Synthesis of N-substituted 1-hydroxypyrrolo[3,4-f]indol-5,7-diones](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Chirkova Z.V., Kabanova M.V., Filimonov S.I., Sergeev S.S., Smirnova E.A., Sudzilovskaya T.N.
Russian Journal of Organic Chemistry,
2017
19.

Esser F., Pook K., Carpy A.
Synthesis,
1990
20.

Zhao D., Hughes D.L., Bender D.R., DeMarco A.M., Reider P.J.
Journal of Organic Chemistry,
1991