Abstract
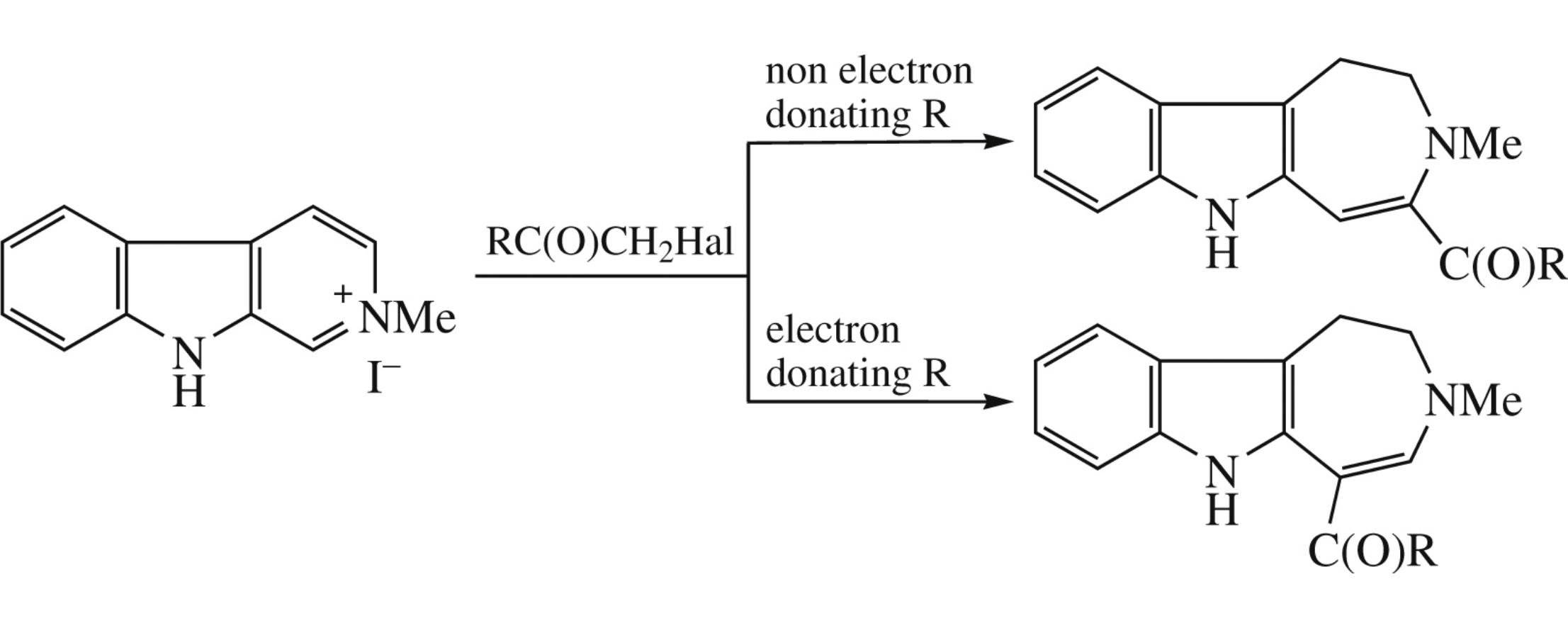
2-Methyl-3,4-dihydro-β-carbolin-2-ium iodide in the reaction with aryl halomethyl ketones is converted into 4- or/and 5-acyl-1,2-dihydroazepino[4,5-b]indoles depending on the electron donating effect of substituents in the aryl moiety of the halomethyl ketone.
References
1.

Bartlett M.F., Dickel D.F., Taylor W.I.
Journal of the American Chemical Society,
1958
2.

Kam T., Sim K., Pang H., Koyano T., Hayashi M., Komiyama K.
Bioorganic and Medicinal Chemistry Letters,
2004
3.

Jana G.K., Sinha S.
Tetrahedron,
2012
4.
![Synthese von 7,12-Dihydro-indolo[3,2-d][1]benzazepin-6-(5H)-onen und 6,11-Dihydro-thieno-[3′,2′:2,3]azepino[4,5-b]indol-5(4H)-on](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kunick C.
Archiv der Pharmazie,
1992
5.

Tolle N., Kunick C.
Current Topics in Medicinal Chemistry,
2011
6.
10.1016/j.mencom.2018.01.028_bib0030
Inhibitors of Cyclin-Dependent Kinases as Anti-Tumor Agents, CRC Enzyme Inhibitor Series,
2006
7.
J. Rutka and C. Faria, Patent WO 2016201581 A1, 2016.
8.
P. Hammerman, K.-K. Wong and N. S. Gray, Patent WO 2016201370 A1, 2016.
9.
![Synthesis of Azepino[4,5-b]indole Analogues via 7-endo-Selective Cyclization of Isocyanoacetates and Indole-1,2-alkynylaldehydes: An Approach towards the Chromoazepinone Core](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
10.

Ferrer C., Amijs C. ., Echavarren A.
Chemistry - A European Journal,
2007
11.

Ferrer C., Echavarren A.M.
Angewandte Chemie - International Edition,
2006
12.

Flatt B., Martin R., Wang T., Mahaney P., Murphy B., Gu X., Foster P., Li J., Pircher P., Petrowski M., Schulman I., Westin S., Wrobel J., Yan G., Bischoff E., et. al.
Journal of Medicinal Chemistry,
2009
13.
![Azepinoindoles. I. Hexahydroazepino[4,5-b]indoles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Hester J.B., Tang A.H., Keasling H.H., Veldkamp W.
Journal of Medicinal Chemistry,
1968
14.

Liu K.G., Lo J.R., Comery T.A., Zhang G.M., Zhang J.Y., Kowal D.M., Smith D.L., Di L., Kerns E.H., Schechter L.E., Robichaud A.J.
Bioorganic and Medicinal Chemistry Letters,
2008
15.
![Formation and reactions of azepino[4,5-b]indoles: an unprecedented ozone reaction in the formation of novel benzo[c]naphthyridinones.](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Stewart S.G., Ghisalberti E.L., Skelton B.W., Heath C.H.
Organic and Biomolecular Chemistry,
2010
16.

Montgomery T.D., Nibbs A.E., Zhu Y., Rawal V.H.
Organic Letters,
2014
17.
R. Martin, T.-L. Wang, B.T. Flatt, X.-H. Gu and R. Griffith, Patent WO 2003099821 A1, 2003.
18.
T. Baik, C.A. Buhr, B.B. Busch, D.S.-M. Chan, B.T. Flatt, X.H. Gu, V. Jammalamadaka, R.G. Khoury, K. Lara, S. Ma, R. Martin, R. Mohan, J.M. Nuss, J.J. Parks, L. Wang, T.L. Wang, J.H. Wu, W. Xu and B. K. S. Yeung, Patent WO 2007070796 A1, 2007.
19.

Lundquist J.T., Harnish D.C., Kim C.Y., Mehlmann J.F., Unwalla R.J., Phipps K.M., Crawley M.L., Commons T., Green D.M., Xu W., Hum W., Eta J.E., Feingold I., Patel V., Evans M.J., et. al.
Journal of Medicinal Chemistry,
2010
20.
![Practical syntheses of hexahydroazepino[4,5-b]- and hexahydroazocino[4,5-b]indoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Diker K., de Maindreville M.D., Lévy J.
Tetrahedron Letters,
1995
21.
![Synthesis of 3-benzazepines and azepino[4,5-b]heterocyclic ring systems via intramolecular Friedel–Crafts cyclization](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kargbo R.B., Sajjadi-Hashemi Z., Roy S., Jin X., Herr R.J.
Tetrahedron Letters,
2013
22.
![Synthesis of β-carbolines and azepino[4,5-b]indols from azidoacrylates](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Moody C.J., Ward J.G.
Journal of the Chemical Society Chemical Communications,
1982
23.
![[2,3] Fused indoles. Synthesis of β-carbolines and azepino[4,5-b]indoles from 3-(2-alkylindol-3-yl)-2-azidoacrylates1](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Moody C.J., Ward J.G.
Journal of the Chemical Society Perkin Transactions 1,
1984
24.

Bosch J., Bennasar M.-., Zulaica E., Massiot G., Massoussa B.
Tetrahedron Letters,
1987
25.

Kuehne M.E., Bohnert J.C., Bornmann W.G., Kirkemo C.L., Kuehne S.E., Seaton P.J., Zebovitz T.C.
Journal of Organic Chemistry,
1985
26.

Zubenko A.A., Kartsev V.G., Morkovnik A.S., Divaeva L.N., Suponitsky K.Y.
ChemistrySelect,
2016
27.
10.1016/j.mencom.2018.01.028_bib0130
Sheldrick
SHELXL-97, Program for the Refinement of Crystal Structure,
1997