Abstract
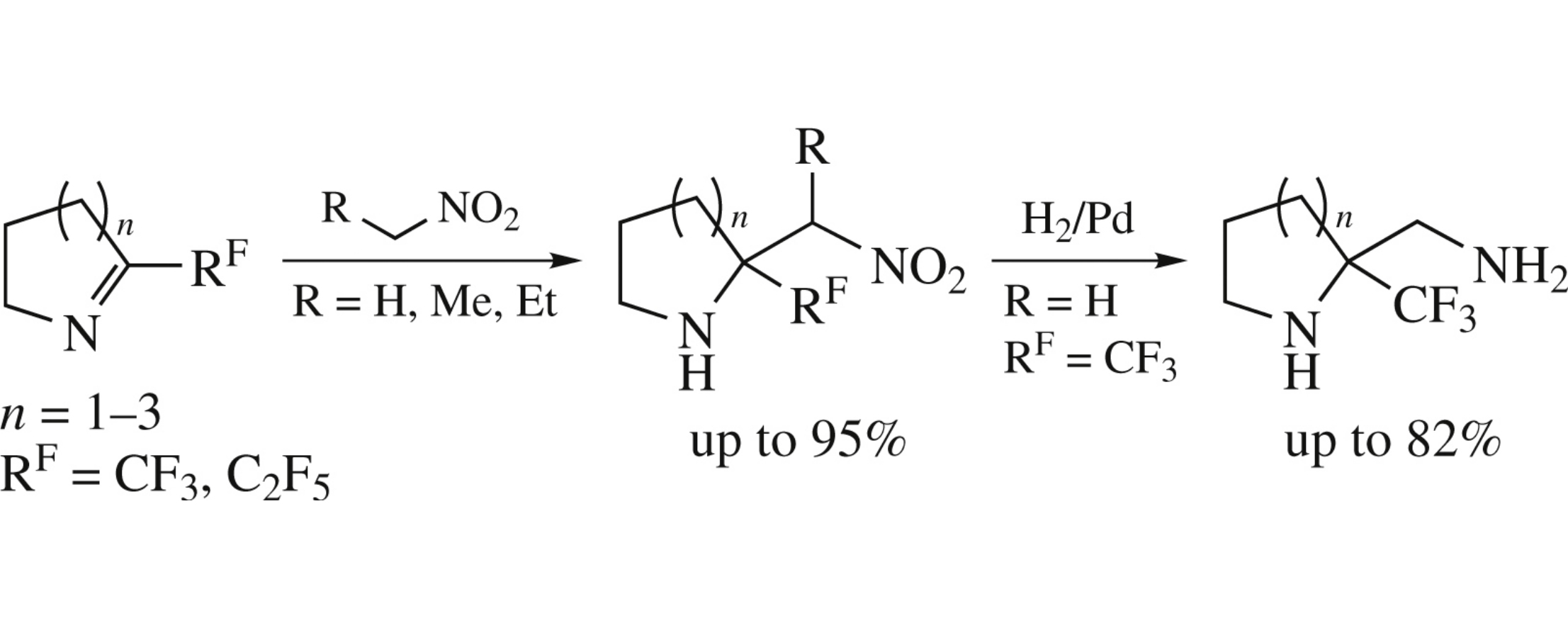
Aza-Henry reaction of 5-, 6- and 7-membered cyclic imines bearing perfluorinated group with nitroalkanes affords the corresponding nitro amines in up to 95% yield. Subsequent reduction of nitro group gives perfluoroalkyl-equipped vicinal diamines.
References
1.

Salwiczek M., Nyakatura E.K., Gerling U.I., Ye S., Koksch B.
Chemical Society Reviews,
2012
2.

Ahrens T., Kohlmann J., Ahrens M., Braun T.
Chemical Reviews,
2014
3.

Wang J., Sánchez-Roselló M., Aceña J.L., del Pozo C., Sorochinsky A.E., Fustero S., Soloshonok V.A., Liu H.
Chemical Reviews,
2013
4.

Zhou Y., Wang J., Gu Z., Wang S., Zhu W., Aceña J.L., Soloshonok V.A., Izawa K., Liu H.
Chemical Reviews,
2016
5.
Maienfisch P., Hall R.G.
Chimia,
2004
6.

Boibessot T., Bénimélis D., Meffre P., Benfodda Z.
Amino Acids,
2016
7.
K Andra K.
Biochemistry & Analytical Biochemistry,
2015
8.

Oishi M., Kondo H., Amii H.
Chemical Communications,
2009
9.
Shmatova O.I., Shevchenko N.E., Balenkova E.S., Röschenthaler G., Nenajdenko V.G.
Mendeleev Communications,
2013
10.
Romanov A.R., Rulev A.Y., Ushakov I.A., Muzalevskiy V.M., Nenajdenko V.G.
Mendeleev Communications,
2014
11.

Smart B.E.
Journal of Fluorine Chemistry,
2001
12.

Shmatova O.I., Nenajdenko V.G.
Journal of Organic Chemistry,
2013
13.
Levin V.V., Struchkova M.I., Dilman A.D.
Mendeleev Communications,
2017
14.
Filyakova V.I., Boltachova N.S., Pervova M.G., Palysaeva N.V., Slepukhin P.A., Sheremetev A.B., Charushin V.N.
Mendeleev Communications,
2017
15.

Jensen A.A., Gharagozloo P., Birdsall N.J., Zlotos D.P.
European Journal of Pharmacology,
2006
16.

Lazareno S., Gharagozloo P., Kuonen D., Popham A., Birdsall N.J.
Molecular Pharmacology,
1998
17.
![Asymmetric Synthesis of (S,S,S)-2-Aza-bicyclo-[3.3.0]-octane-3-carboxylic Acid Benzyl Ester: Formal Synthesis of Ramipril](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Kondaiah G.C., Vivekanandareddy M., Reddy L.A., Anurkar S.V., Gurav V.M., Ravikumar M., Bhattacharya A., Bandichhor R.
Synthetic Communications,
2011
18.

Malakondaiah G.C., Gurav V.M., Reddy L.A., Babu K.S., Bhaskar B.V., Reddy P.P., Bhattacharya A., Anand R.V.
Synthetic Communications,
2008
19.
10.1016/j.mencom.2018.01.027_sbref0025a
Shevchenko
Synthesis,
2010
20.

Shevchenko N.E., Nenajdenko V.G., Röschenthaler G.
Journal of Fluorine Chemistry,
2008
21.

Shmatova O.I., Nenajdenko V.G.
European Journal of Organic Chemistry,
2013
22.

Shmatova O.I., Shevchenko N.E., Nenajdenko V.G.
European Journal of Organic Chemistry,
2015
23.

Kutovaya I.V., Shmatova O.I., Tkachuk V.M., Melnichenko N.V., Vovk M.V., Nenajdenko V.G.
European Journal of Organic Chemistry,
2015