Abstract
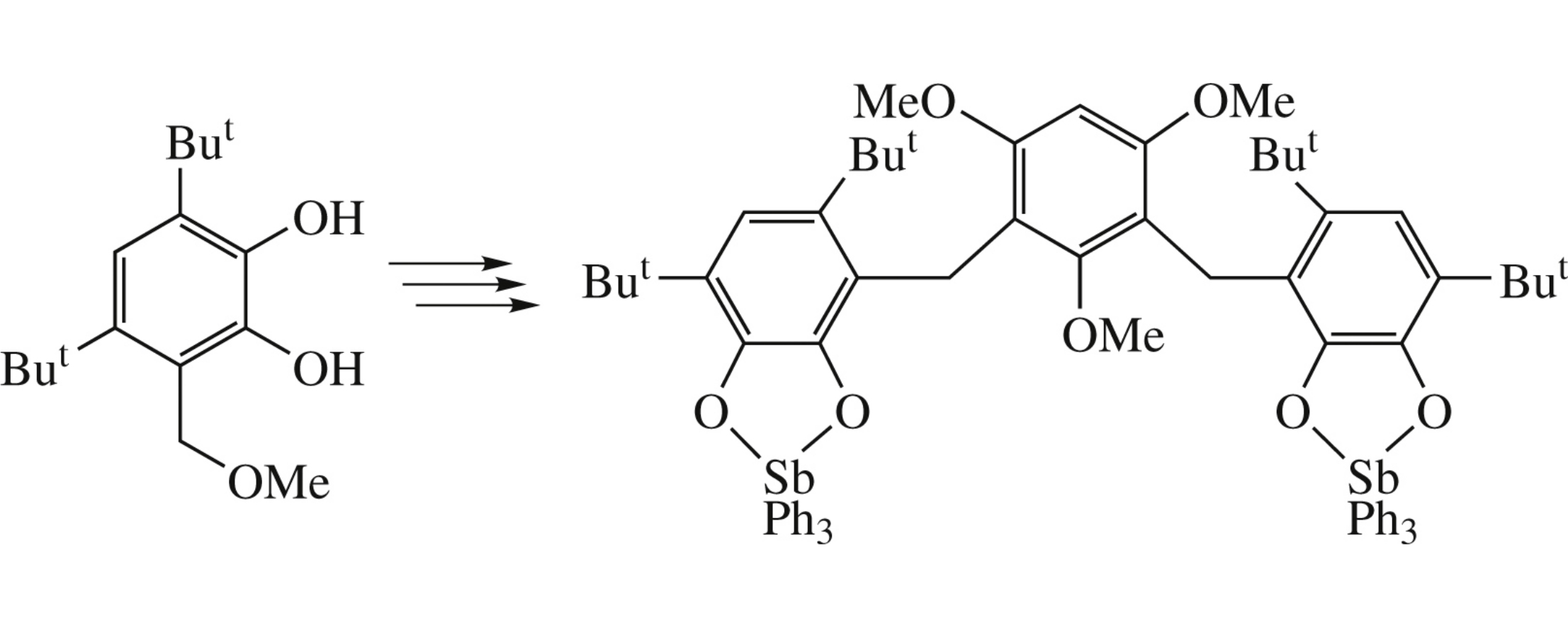
Alkylation of 1,3,5-trimethoxybenzene with 3,5-di-tert-butyl-6-methoxymethylcatechol affords new sterically hindered biscatechol, whose oxidation gives the corresponding bis-o-quinone. Its treatment with triphenylstibine leads to bis-triphenylantimony(v) bis-catecholate.
References
1.
10.1016/j.mencom.2018.01.025_sbref0005a
Levason
2003
2.

Kishore P.V., Baskar V.
Inorganic Chemistry,
2014
3.

Jami A.K., Prabhu M.S., Baskar V.
Organometallics,
2010
4.

Huang Y.
Accounts of Chemical Research,
1992
5.
10.1016/j.mencom.2018.01.025_sbref0015a
Farrell
2003
6.

Liu R., Ma Y., Yu L., Li J., Cui J., Wang R.
Applied Organometallic Chemistry,
2003
7.

Qiu J., Unruh D.K., Cozzolino A.F.
Journal of Physical Chemistry A,
2016
8.

Hirai M., Gabbaï F.P.
Chemical Science,
2014
9.

Abakumov G.A., Poddel'sky A.I., Grunova E.V., Cherkasov V.K., Fukin G.K., Kurskii Y.A., Abakumova L.G.
Angewandte Chemie - International Edition,
2005
10.

Arsenyev M.V., Shurygina M.P., Poddel’sky A.I., Druzhkov N.O., Chesnokov S.A., Fukin G.K., Cherkasov V.K., Abakumov G.A.
Journal of Polymer Research,
2013
11.

Chesnokov S.A., Lenshina N.A., Arsenyev M.V., Kovylin R.S., Baten'kin M.A., Poddel'sky A.I., Abakumov G.A.
Applied Organometallic Chemistry,
2016
12.

Lenshina N.A., Shurygina M.P., Arsenyev M.V., Poddel’sky A.I., Zaitsev S.D., Chesnokov S.A., Abakumov G.A.
Journal of Coordination Chemistry,
2015
13.

Smolyaninov I.V., Poddel’sky A.I., Korchagina E.O., Smolyaninova S.A., Berberova N.T.
Doklady Physical Chemistry,
2015
14.

Poneti G., Mannini M., Cortigiani B., Poggini L., Sorace L., Otero E., Sainctavit P., Sessoli R., Dei A.
Inorganic Chemistry,
2013
15.

Shultz D.A., Bodnar S.H., Kumar R.K., Lee H., Kampf J.W.
Inorganic Chemistry,
2000
16.

Arsenyev M.V., Baranov E.V., Chesnokov S.A., Cherkasov V.K., Abakumov G.A.
Russian Chemical Bulletin,
2013
17.
Arsenyev M.V., Baranov E.V., Fedorov A.Y., Chesnokov S.A., Abakumov G.A.
Mendeleev Communications,
2015
18.

Arsenyev M.V., Khamaletdinova N.M., Baranov E.V., Chesnokov S.A., Cherkasov V.K.
Russian Chemical Bulletin,
2016
19.

Druzhkov N.O., Egorova E.N., Arsen’ev M.V., Baranov E.V., Cherkasov V.K.
Russian Chemical Bulletin,
2016
20.

Shultz D.A., Boal A.K., Driscoll D.J., Kitchin J.R., Tew G.N.
Journal of Organic Chemistry,
1995
21.

Bencini A., Daul C.A., Dei A., Mariotti F., Lee H., Shultz D.A., Sorace L.
Inorganic Chemistry,
2001
22.

Caneschi A., Dei A., Mussari C.P., Shultz D.A., Sorace L., Vostrikova K.E.
Inorganic Chemistry,
2002
23.

Sloop J.C., Shultz D.A., Coote T., Shepler B., Sullivan U., Kampf J.W., Boyle P.D.
Journal of Physical Organic Chemistry,
2011
24.

Poddel’sky A., Piskunov A., Druzhkov N., Fukin G., Cherkasov V., Abakumov G.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2009
25.
![5-{5-(bicyclo[2.2.1]hept-2-enyl)hydroxymethyl}-3,6-di-tert-butylo-benzoquinone and related polymers. Synthesis and some properties](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Safronova A.V., Bochkarev L.N., Druzhkov N.O., Kurskii Y.A., Baranov E.V., Abakumov G.A.
Russian Journal of General Chemistry,
2012
26.
Arsenyev M.V., Baranov E.V., Shurygina M.P., Chesnokov S.A., Abakumov G.A.
Mendeleev Communications,
2016
27.

Poddel'sky A.I., Arsenyev M.V., Astaf'eva T.V., Chesnokov S.A., Fukin G.K., Abakumov G.A.
Journal of Organometallic Chemistry,
2017
28.

Dalpozzo R., Bartoli G., Sambri L., Melchiorre P.
Chemical Reviews,
2010
29.

Singh M.S., Nagaraju A., Anand N., Chowdhury S.
RSC Advances,
2014
30.

Osipov D.V., Osyanin V.A., Klimochkin Y.N.
Russian Chemical Reviews,
2017
31.

Iwanek W., Stefańska K., Szumna A., Wierzbicki M.
Tetrahedron,
2015
32.
10.1016/j.mencom.2018.01.025_bib0085
Bukharov
Zh. Org. Khim.,
2004
33.

Tangdenpaisal K., Phakhodee W., Ruchirawat S., Ploypradith P.
Tetrahedron,
2013
34.
![6,6'-[piperazine-1,4-diylbis(methylene)]bis[3,5-di(tert-butyl)-1,2- benzoquinone]: Synthesis and properties](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Sayapin Y.A., Tupaeva I.O., Tkachev V.V., Shilov G.V.
Russian Journal of Organic Chemistry,
2016
35.

Sobczyk L., Grabowski S.J., Krygowski T.M.
Chemical Reviews,
2005
36.

Schaefer T.
The Journal of Physical Chemistry,
1975
37.

Denisov E.T., Denisova T.G.
Russian Chemical Reviews,
2009
38.
![Synthesis, structure, and spectroscopic properties of copper(II) compounds containing nitrogen–sulphur donor ligands; the crystal and molecular structure of aqua[1,7-bis(N-methylbenzimidazol-2′-yl)-2,6-dithiaheptane]copper(II) perchlorate](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Addison A.W., Rao T.N., Reedijk J., van Rijn J., Verschoor G.C.
Journal of the Chemical Society Dalton Transactions,
1984
39.

Poddel'sky A.I., Baranov E.V., Fukin G.K., Cherkasov V.K., Abakumov G.A.
Journal of Organometallic Chemistry,
2013
40.
10.1016/j.mencom.2018.01.025_bib0115
SAINT, v. 8.27B, Bruker AXS,
2012
41.
10.1016/j.mencom.2018.01.025_bib0120
SADABS, v. 2014/2, Bruker AXS,
2014
42.
10.1016/j.mencom.2018.01.025_bib0125
Sheldrick
Acta Crystallogr.,
2015
43.
10.1016/j.mencom.2018.01.025_bib0130
Sheldrick
SHELXTL, v. 6.14, Bruker AXS,
2003