Abstract
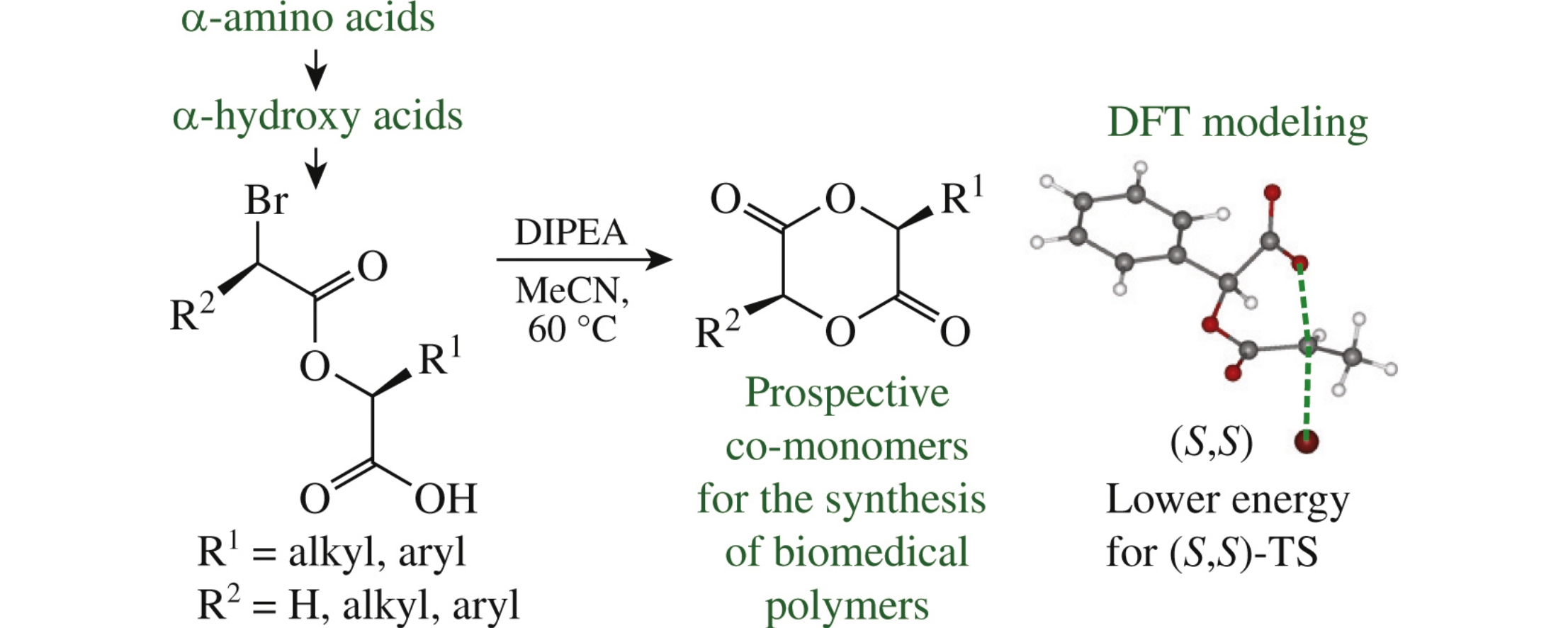
Intramolecular cyclization of α-bromoacyl derivatives of α-hydroxy acids affords 1,4-dioxane-2,5-diones. The method is suitable to obtain mono- and disubstituted products, the latter being formed stereoselectively as (S,S)-diastereomers, as confirmed by DFT modeling.
References
1.

Farah S., Anderson D.G., Langer R.
Advanced Drug Delivery Reviews,
2016
2.

Castro-Aguirre E., Iñiguez-Franco F., Samsudin H., Fang X., Auras R.
Advanced Drug Delivery Reviews,
2016
3.

Santoro M., Shah S.R., Walker J.L., Mikos A.G.
Advanced Drug Delivery Reviews,
2016
4.

Raquez J., Habibi Y., Murariu M., Dubois P.
Progress in Polymer Science,
2013
5.

Narayanan G., Vernekar V.N., Kuyinu E.L., Laurencin C.T.
Advanced Drug Delivery Reviews,
2016
6.

Brannigan R.P., Dove A.P.
Biomaterials Science,
2017
7.
Nifant’ev I.E., Minyaev M.E., Shlyakhtin A.V., Ivchenko P.V., Churakov A.V.
Mendeleev Communications,
2017
8.
Sedush N.G., Izraylit V.V., Mailyan A.K., Savinov D.V., Kiryukhin Y.I., Chvalun S.N.
Mendeleev Communications,
2017
9.
Ivchenko P.V., Shlyakhtin A.V., Nifant’ev I.E.
Mendeleev Communications,
2017
10.

Ruengdechawiwat S., Somsunan R., Molloy R., Siripitayananon J., Franklin V.J., Topham P.D., Tighe B.J.
Advanced Materials Research,
2014
11.

Wang L., Poirier V., Ghiotto F., Bochmann M., Cannon R.D., Carpentier J., Sarazin Y.
Macromolecules,
2014
12.
10.1016/j.mencom.2018.01.020_bib0060
Raquez
2012
13.
10.1016/j.mencom.2018.01.020_bib0065
Suwandi
Clin. Exp. Rheumatol.,
2015
14.

Coumes F., Darcos V., Domurado D., Li S., Coudane J.
Polymer Chemistry,
2013
15.

Synthesis and Organocatalytic Ring-Opening Polymerization of Cyclic Esters Derived from l-Malic Acid
Pounder R.J., Dove A.P.
Biomacromolecules,
2010
16.

Olejniczak J., Chan M., Almutairi A.
Macromolecules,
2015
17.

Leemhuis M., Akeroyd N., Kruijtzer J.A., van Nostrum C.F., Hennink W.E.
European Polymer Journal,
2008
18.

Trimaille T., Möller M., Gurny R.
Journal of Polymer Science, Part A: Polymer Chemistry,
2004
19.

Delle Chiaie K.R., Yablon L.M., Biernesser A.B., Michalowski G.R., Sudyn A.W., Byers J.A.
Polymer Chemistry,
2016
20.
10.1016/j.mencom.2018.01.020_bib0100
Reichardt
Solvents and Solvent Effects in Organic Chemistry,
2010
21.

Deechongkit S., You S., Kelly J.W.
Organic Letters,
2004
22.

Nifant'ev I.E., Shlyakhtin A.V., Tavtorkin A.N., Ivchenko P.V., Borisov R.S., Churakov A.V.
Catalysis Communications,
2016