Abstract
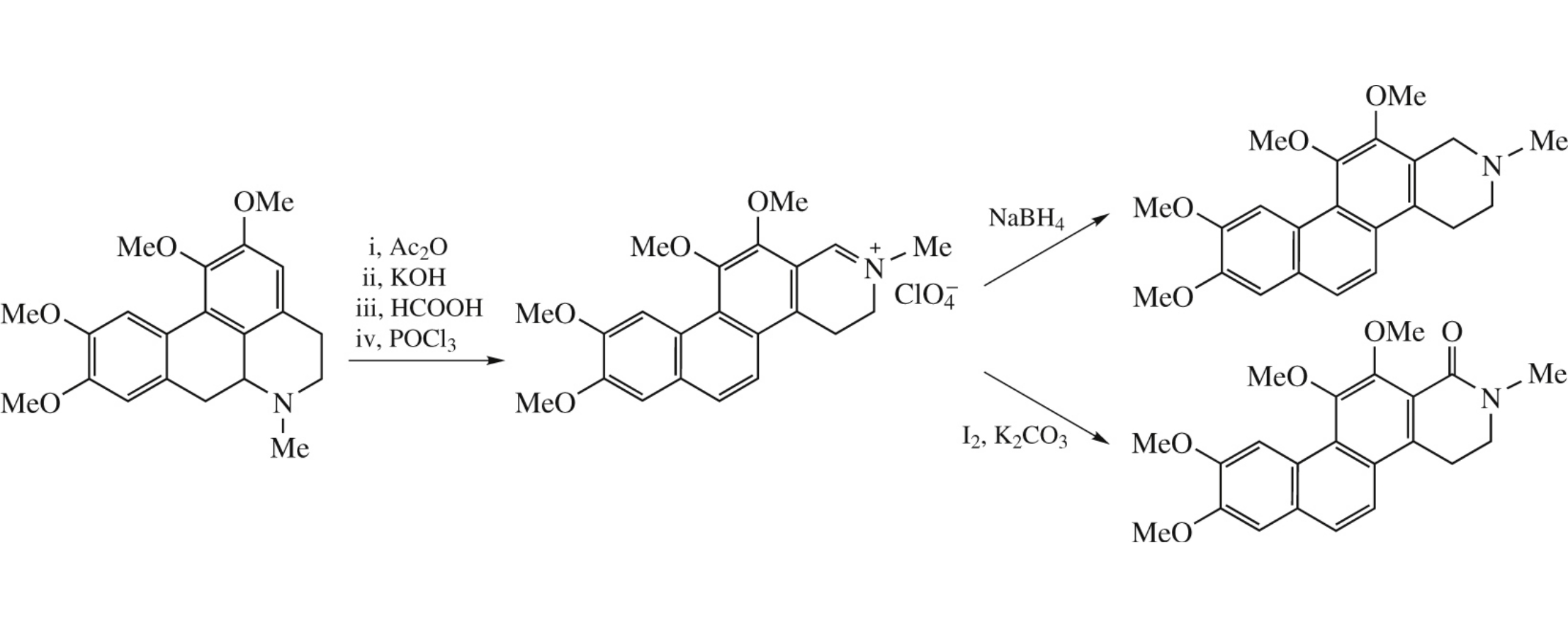
Using glaucine as an example, a convenient access to naphtho[2,1-f]isoquinoline (litebamine) derivatives is proposed. The synthetic scheme involves a stepwise pyridine–pyridine recyclization of the intermediate N-acylsecoaporphine analogues.
References
1.

Spasova M., Philipov S., Milkova T.
Advances in Experimental Medicine and Biology,
2009
2.

Nabavi S., Uriarte E., Fontenla J., Rastrelli L., Sobarzo-Sanchez E.
Current Organic Chemistry,
2017
3.
10.1016/j.mencom.2018.01.019_sbref0005c
Pinto
Pharm. Pharmacol. Commun.,
1998
4.

Remichkova M., Dimitrova P., Philipov S., Ivanovska N.
Fitoterapia,
2009
5.

Kang H., Jang S., Pak J.H., Shim S.
Molecular and Cellular Biochemistry,
2015
6.

Eid S.Y., El-Readi M.Z., Wink M.
Phytomedicine,
2012
7.

Chi T., Lee S., Su M.
Planta Medica,
2006
8.

Dargan P.I., Button J., Hawkins L., Archer J.R., Ovaska H., Lidder S., Ramsey J., Holt D.W., Wood D.M.
European Journal of Clinical Pharmacology,
2008
9.

Asencio M., Hurtado-Guzmán C., López J.J., Cassels B.K., Protais P., Chagraoui A.
Bioorganic and Medicinal Chemistry,
2005
10.

Cortijo J., Villagrasa V., Pons R., Berto L., Martí-Cabrera M., Martinez-Losa M., Domenech T., Beleta J., Morcillo E.J.
British Journal of Pharmacology,
1999
11.

Spasova M., Philipov S., Nikolaeva-Glomb L., Galabov A.S., Milkova T.
Bioorganic and Medicinal Chemistry,
2008
12.

Gyurkovska V., Philipov S., Kostova N., Ivanovska N.
Immunopharmacology and Immunotoxicology,
2014
13.

Castro J.L., Castedo L., Riguera R.
Journal of Organic Chemistry,
1987
14.

Rao N.S., Lee S.
Journal of the Chinese Chemical Society,
2000
15.

Castedo L., A. Seijas J., Rodr暖uez de Lera A., Villaverde C.
Heterocycles,
1985
16.

Voskressensky L.G., Borisova T.N., Listratova A.V., Kulikova L.N., Titov A.A., Varlamov A.V.
Tetrahedron Letters,
2006
17.

WHALEY W.M., MEADOW M.
Journal of Organic Chemistry,
1954
18.
![Photochemically induced cyclization of N-[2-(o-styryl)phenylethyl]acetamides and 5-styryl-1-methyl-1,2,3,4-tetrahydroisoquinolines: new total syntheses of 1-methyl-1,2,3,4-tetrahydronaphtho[2,1-f]isoquinolines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Martı́nez E., Estévez J.C., Estévez R.J., Castedo L.
Tetrahedron,
2001
19.
![First total syntheses of the 1,2,3,4-tetrahydronaphtho[2,1-f]isoquinolines annoretine and litebamine](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
First total syntheses of the 1,2,3,4-tetrahydronaphtho[2,1-f]isoquinolines annoretine and litebamine
Pampı́n M.C., Estévez J.C., Estévez R.J., Suau R., Castedo L.
Tetrahedron,
2003
20.

Wu Y., Liou J., Duh C., Lee S., Lu S.
Tetrahedron Letters,
1991
21.

Egydio A.P., Valvassoura T.A., Santos D.Y.
Biochemical Systematics and Ecology,
2013
22.

Vitorović-Todorović M.D., Juranić I.O., Mandić L.M., Drakulić B.J.
Bioorganic and Medicinal Chemistry,
2010
23.

Teng C., Hsueh C., Chang Y., Ko F., Lee S., Liu K.C.
Journal of Pharmacy and Pharmacology,
1997
24.

Huang C., Huang W., Wang S., Wu P., Wu W.
European Journal of Pharmacology,
2008
25.

Wu Y., Chang G., Chang-Yih D., Shang-Kwei W.
Phytochemistry,
1993
26.

Shoei-Sheng L., Yi-Jen L., Mei-Zu C., Yang-Chang W., Chung-Hsiung C.
Tetrahedron Letters,
1992
27.

Chiou C., Kang J., Lee S.
Journal of Natural Products,
1998
28.

Castedo L., L. Castro J., Riguera R.
Heterocycles,
1982
29.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
30.
10.1016/j.mencom.2018.01.019_bib0075
Sheldrick
Acta Crysallogr.,
2015
31.
10.1016/j.mencom.2018.01.019_bib0080
Sheldrick
Acta Crystallogr.,
2015
32.
Pampín M.C., Estévez J.C., Estévez R.J., Castedo L.
Arkivoc,
2003
33.

Fletcher S., Gunning P.T.
Tetrahedron Letters,
2008