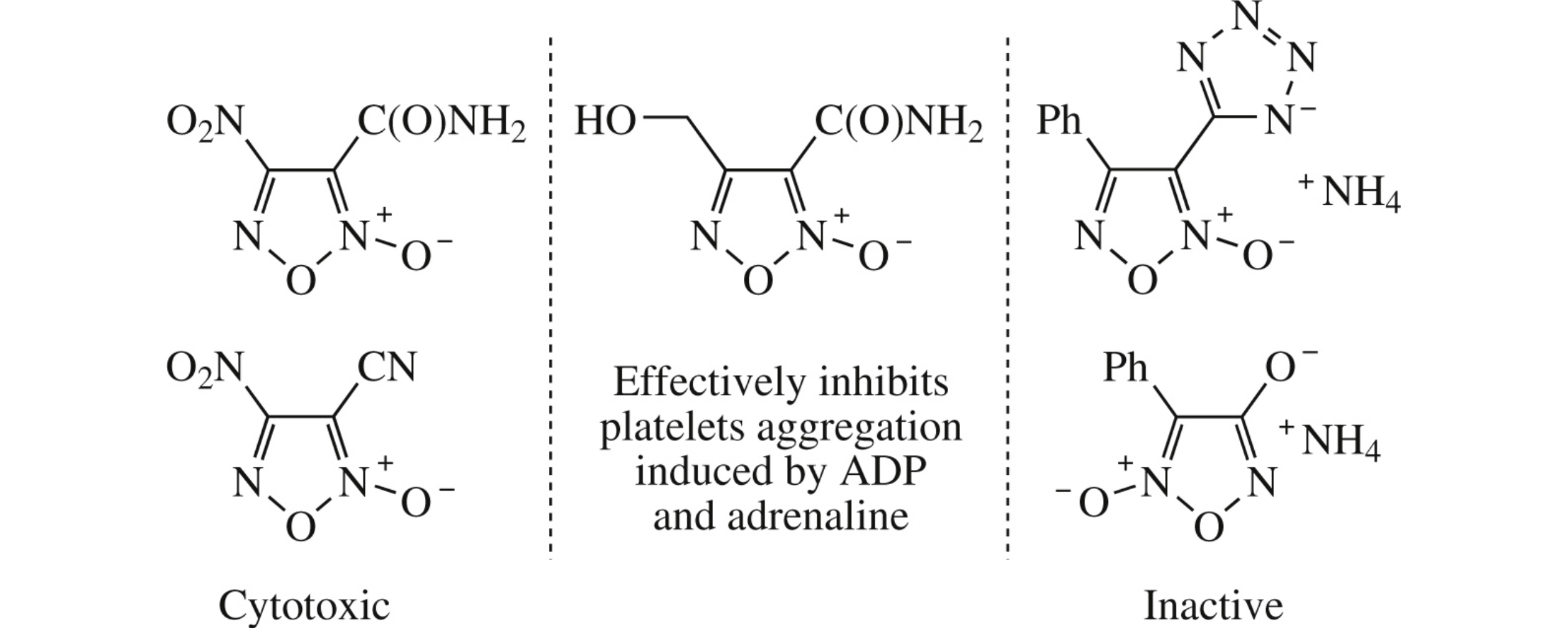
Abstract
The effect of the water-soluble furoxan derivatives on platelet aggregation has been estimated using a series of inducers. The most active compound, 3-carbamoyl-4-(hydroxymethyl)furoxan (CAS-1609), effectively inhibits aggregate formation induced by adenosine diphosphate and adrenaline. This result is a good basis for the search of new structures with antiaggregant activity among furoxancarboxylic acid amides.
References
1.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
2.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
3.
Ananikov V.P., Eremin D.B., Yakukhnov S.A., Dilman A.D., Levin V.V., Egorov M.P., Karlov S.S., Kustov L.M., Tarasov A.L., Greish A.A., Shesterkina A.A., Sakharov A.M., Nysenko Z.N., Sheremetev A.B., Stakheev A.Y., et. al.
Mendeleev Communications,
2017
4.

Serafim R.A., Pernichelle F.G., Ferreira E.I.
Expert Opinion on Drug Discovery,
2017
5.
10.1016/j.mencom.2018.01.016_bib0015
Cheng
Nitric Oxide and Cancer. Prognosis, Prevention and Therapy,
2010
6.

Fershtat L.L., Makhova N.N.
ChemMedChem,
2017
7.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
8.

Chegaev K., Federico A., Marini E., Rolando B., Fruttero R., Morbin M., Rossi G., Fugnanesi V., Bastone A., Salmona M., Badiola N.B., Gasparini L., Cocco S., Ripoli C., Grassi C., et. al.
Bioorganic and Medicinal Chemistry,
2015
9.

Bertinaria M., Orjuela-Sanchez P., Marini E., Guglielmo S., Hofer A., Martins Y.C., Zanini G.M., Frangos J.A., Gasco A., Fruttero R., Carvalho L.J.
Journal of Medicinal Chemistry,
2015
10.

Guglielmo S., Cortese D., Vottero F., Rolando B., Kommer V.P., Williams D.L., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2014
11.

Borretto E., Lazzarato L., Spallotta F., Cencioni C., D’Alessandra Y., Gaetano C., Fruttero R., Gasco A.
ACS Medicinal Chemistry Letters,
2013
12.
10.1016/j.mencom.2018.01.016_bib0030
Gasco
Nitric Oxide Donors: For Pharmaceutical and Biological Applications,
2005
13.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2016
14.

Kulikov A.S., Ovchinnikov I.V., Molotov S.I., Makhova N.N.
Russian Chemical Bulletin,
2003
15.
![An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron,
2015
16.
K. Schönafinger and H. Bohn, Patent US 5486531 A, 1996.
17.

Bohn H., Brendel J., Martorana P.A., Schönafinger K.
British Journal of Pharmacology,
1995
18.

Balbo S., Lazzarato L., Di Stilo A., Fruttero R., Lombaert N., Kirsch-Volders M.
Toxicology Letters,
2008
19.

Lai Y., Shen L., Zhang Z., Liu W., Zhang Y., Ji H., Tian J.
Bioorganic and Medicinal Chemistry Letters,
2010
20.
10.1016/j.mencom.2018.01.016_sbref0060b
Aguirre
Pharmazie,
2006
21.

Zhao J., Gou S., Sun Y., Fang L., Wang Z.
Inorganic Chemistry,
2012
22.
Kulikov A.S., Larin A.A., Fershtat L.L., Anikina L.V., Pukhov S.A., Klochkov S.G., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
23.

Puri R.N., Colman R.W., Liberman M.A.
Critical Reviews in Biochemistry and Molecular Biology,
1997
24.

Ueno M., Kodali M., Tello-Montoliu A., Angiolillo D.J.
Journal of Atherosclerosis and Thrombosis,
2011
25.

Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Tosco P., Di Stilo A., Fruttero R., Gasco A.
Il Farmaco,
2003
26.

Fershtat L.L., Epishina M.A., Ovchinnikov I.V., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2016