Abstract
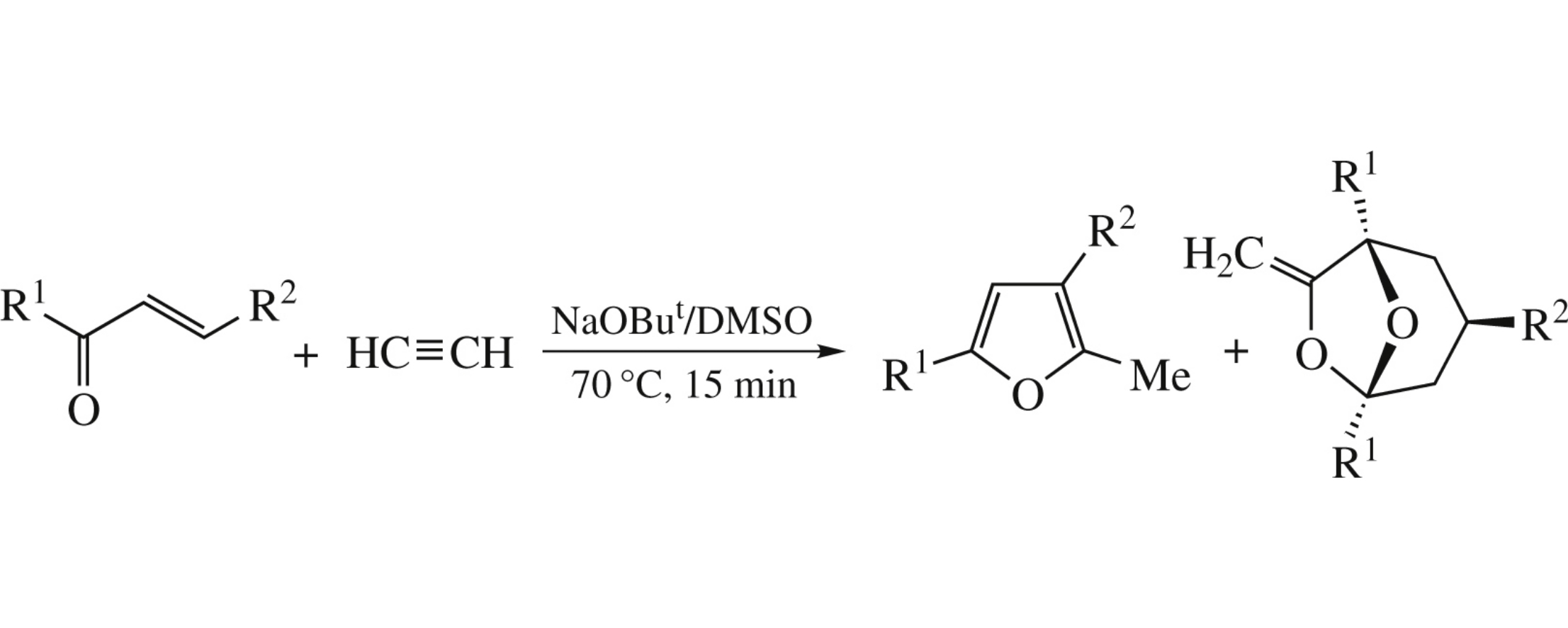
Acetylene reacts with chalcones in the NaOBut / DMSO superbase system (3–12 atm, 70°C, 15 min) to give, along with substituted furans, functionalized 3-exo-aryl-7-methylidene- 6,8-dioxabicyclo[3.2.1]octanes, novel frontalin congeners, which are diastereoselectively assembled from two molecules of chalcones and one molecule of acetylene.
References
1.

Schobert H.
Chemical Reviews,
2013
2.

Trotuş I., Zimmermann T., Schüth F.
Chemical Reviews,
2013
3.
Rodygin K.S., Kostin A.A., Ananikov V.P.
Mendeleev Communications,
2015
4.

Huang Z., Lim H.N., Mo F., Young M.C., Dong G.
Chemical Society Reviews,
2015
5.

Galkin K.I., Ananikov V.P.
Russian Chemical Reviews,
2016
6.
Fershtat L.L., Ashirbaev S.S., Kulikov A.S., Kachala V.V., Makhova N.N.
Mendeleev Communications,
2015
8.
Shabalin D.A., Dvorko M.Y., Schmidt E.Y., Protsuk N.I., Ushakov I.A., Mikhaleva A.I., Trofimov B.A.
Mendeleev Communications,
2015
9.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
10.
Trofimov B.A., Belyaeva K.V., Andriyankova L.V., Nikitina L.P., Mal’kina A.G.
Mendeleev Communications,
2017
11.

Alyabyev S.B., Beletskaya I.P.
Russian Chemical Reviews,
2017
12.
![One-Pot Assembly of 7-Methylene-6,8-dioxabicyclo[3.2.1]octanes, Congeners of Frontalin, from Ketones and Acetylene](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Trofimov B.A., Schmidt E.Y., Ushakov I.A., Mikhaleva A.I., Zorina N.V., Protsuk N.I., Senotrusova E.Y., Skital'tseva E.V., Kazheva O.N., Alexandrov G.G., Dyachenko O.A.
European Journal of Organic Chemistry,
2009
13.
![A Highly Efficient and Selective AuI-Catalyzed Tandem Synthesis of Diversely Substituted Pyrrolo[1,2-a]quinolines in Aqueous Media](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Liu X., Che C.
Angewandte Chemie - International Edition,
2008
14.
![Gold-Catalyzed One-Pot Cascade Construction of Highly Functionalized Pyrrolo[1,2-a]quinolin-1(2H)-ones](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Zhou Y., Feng E., Liu G., Ye D., Li J., Jiang H., Liu H.
Journal of Organic Chemistry,
2009
15.

Schmidt E.Y., Bidusenko I.A., Cherimichkina N.A., Ushakov I.A., Borodina T.N., Smirnov V.I., Trofimov B.A.
Chemistry - A European Journal,
2015
16.
10.1016/j.mencom.2018.01.015_bib0025
Tarasova
Zh. Org. Khim.,
1991
17.

Rasmussen L.E., Riddle H.S., Krishnamurthy V.
Nature,
2002
18.

Yus M., Ramón D., Prieto O.
European Journal of Organic Chemistry,
2003
19.

Trofimov B.A., Schmidt E.Y., Skitaltseva E.V., Zorina N.V., Protsuk N.I., Ushakov I.A., Mikhaleva A.I., Dyachenko O.A., Kazheva O.N., Aleksandrov G.G.
Tetrahedron Letters,
2011
20.

Schmidt E.Y., Trofimov B.A., Bidusenko I.A., Cherimichkina N.A., Ushakov I.A., Protzuk N.I., Gatilov Y.V.
Organic Letters,
2014
21.

Clarke P.A., Santos S., Martin W.H.
Green Chemistry,
2007
22.

Clarke P., Ermanis K.
Current Organic Chemistry,
2013
23.
![Pot, atom and step economic synthesis: a diversity-oriented approach to construct 2-substituted pyrrolo[2,1-f][1,2,4]triazin-4(3H)-ones](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Xiang H., Chen Y., He Q., Xie Y., Yang C.
RSC Advances,
2013
24.

Jyoti Kalita S., Mecadon H., Deka D.C.
Tetrahedron Letters,
2015
25.

Bidusenko I.A., Cherimichkina N.A., Schmidt E.Y., Trofimov B.A.
Russian Journal of Organic Chemistry,
2017
26.
![Superbase-Promoted Selective Cascade Cyclization Reaction of 1,5-Diketones with Acetylenes to Methylene-6,8-dioxabicyclo[3.2.1]octanes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Schmidt E.Y., Bidusenko I.A., Protsuk N.I., Ushakov I.A., Trofimov B.A.
European Journal of Organic Chemistry,
2013