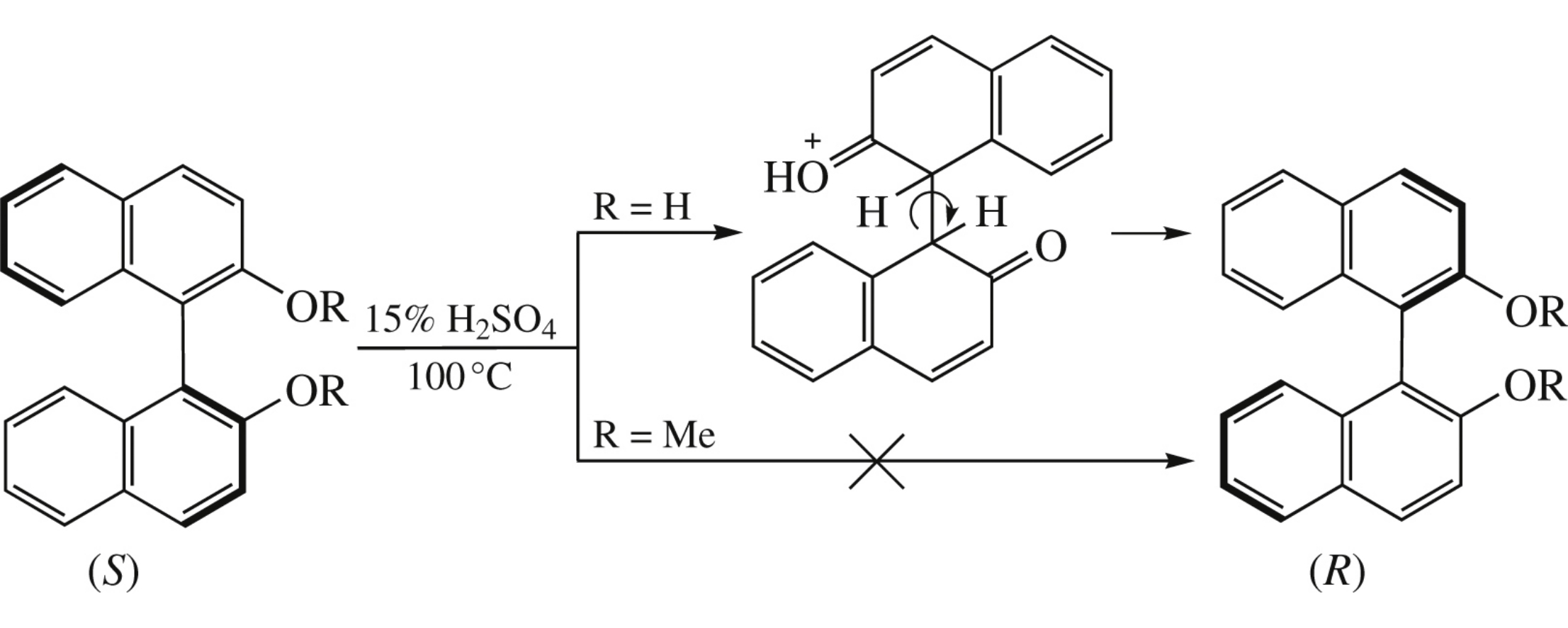
Abstract
Compared with the parent 1,1’-bi-2-naphthol (BINOL), its dimethyl ether is highly resistant towards racemization under moderate acidic conditions. This finding confirms the hypothetical mechanism of BINOL atropisomerization including internal rotation around the C1sp3 –C1'sp3 bond in its protonatedforms.
References
1.

Parmar D., Sugiono E., Raja S., Rueping M.
Chemical Reviews,
2014
2.

Schenker S., Zamfir A., Freund M., Tsogoeva S.B.
European Journal of Organic Chemistry,
2011
3.

4.

Chen Y., Yekta S., Yudin A.K.
Chemical Reviews,
2003
5.

Kočovský P., Vyskočil Š., Smrčina M.
Chemical Reviews,
2003
6.
10.1016/j.mencom.2018.01.007_sbref0005f
Pu
1 1’-Binaphthyl-Based Chiral Materials: Our Journey,
2010
7.

Kyba E.P., Gokel G.W., De Jong F., Koga K., Sousa L.R., Siegel M.G., Kaplan L., Sogah G.D., Cram D.J.
Journal of Organic Chemistry,
1977
8.

Yudin A.K., Martyn L.J., Pandiaraju S., Zheng J., Lough A.
Organic Letters,
1999
9.

Alkorta I., Cancedda C., Cocinero E.J., Dávalos J.Z., Écija P., Elguero J., González J., Lesarri A., Ramos R., Reviriego F., Roussel C., Uriarte I., Vanthuyne N.
Chemistry - A European Journal,
2014
10.

Genaev A.M., Salnikov G.E., Shernyukov A.V., Zhu Z., Koltunov K.Y.
Organic Letters,
2017
11.
Koltunov K.Y., Chernov A.N.
Mendeleev Communications,
2015
12.

Gavrilov K.N., Zheglov S.V., Novikov I.M., Chuchelkin I.V., Gavrilov V.K., Lugovsky V.V., Zamilatskov I.A.
Russian Chemical Bulletin,
2015
13.

14.

Salvadori P., Pini D., Rosini C., Bertucci C., Uccello-Barretta G.
Chirality,
1992
15.
10.1016/j.mencom.2018.01.007_bib0040
Repinskaya
Russ. J. Org. Chem.,
1992