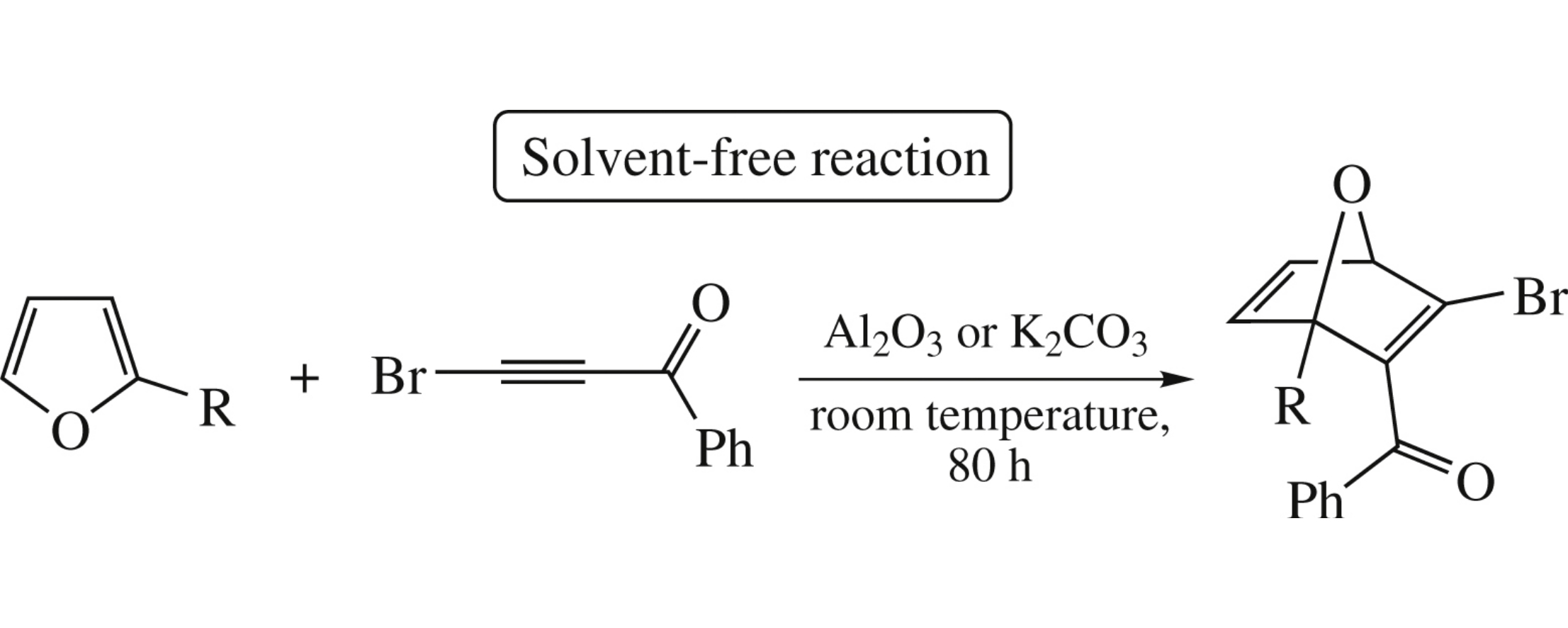
Abstract
1-(Het)aryl-3-bromoprop-2-ynones react with 2-(2-furyl)pyrrole in the Al2O3 or K2CO3 dispersion (room temperature, 1 h) to afford the cross-coupling products either at the furan or pyrrole rings. Furan and 2-methylfuran with 3-bromo-1-phenylprop-2-ynone under the same conditions give the Diels–Alder cycloadducts, while furan-2-carbaldehyde andits acetals are inactive.
References
1.

Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Ushakov I.A.
Tetrahedron Letters,
2004
2.
Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Sinegovskaya L.M., Potekhin K.A., Fedyanin I.V.
Mendeleev Communications,
2005
3.

Gotsko M.D., Sobenina L.N., Tomilin D.N., Ushakov I.A., Dogadina A.V., Trofimov B.A.
Tetrahedron Letters,
2015
4.

Tomilin D.N., Pigulski B., Gulia N., Arendt A., Sobenina L.N., Mikhaleva A.I., Szafert S., Trofimov B.A.
RSC Advances,
2015
5.

Tomilin D.N., Gotsko M.D., Sobenina L.N., Ushakov I.A., Afonin A.V., Soshnikov D.Y., Trofimov A.B., Koldobsky A.B., Trofimov B.A.
Journal of Fluorine Chemistry,
2016
6.

Pigulski B., Arendt A., Tomilin D.N., Sobenina L.N., Trofimov B.A., Szafert S.
Journal of Organic Chemistry,
2016
7.
Tomilin D.N., Soshnikov D.Y., Trofimov A.B., Gotsko M.D., Sobenina L.N., Ushakov I.A., Afonin A.V., Koldobsky A.B., Vitkovskaya N.M., Trofimov B.A.
Mendeleev Communications,
2016
8.
10.1016/j.mencom.2018.01.004_bib0040
Mendling
Arzneimittelforschung,
2002
9.
E. Friedman, M.K. Holloway, G.A. Rodan, A. Schmidt and R. L. Vogel, Patent EP 0786995 B1, 2003.
10.
10.1016/j.mencom.2018.01.004_bib0050
Sperry
Curr. Opin. Drug Discov. Devel.,
2005
11.

Bern C., Montgomery S.P., Herwaldt B.L., Rassi A., Marin-Neto J.A., Dantas R.O., Maguire J.H., Acquatella H., Morillo C., Kirchhoff L.V., Gilman R.H., Reyes P.A., Salvatella R., Moore A.C.
JAMA - Journal of the American Medical Association,
2007
12.

Schneider K., Keller S., Wolter F., Röglin L., Beil W., Seitz O., Nicholson G., Bruntner C., Riedlinger J., Fiedler H., Süssmuth R.
Angewandte Chemie - International Edition,
2008
13.

Machado R.S., Silva M.R., Viriato A.
Jornal de Pediatria,
2008
14.

Dong Y., Shi Q., Liu Y., Wang X., Bastow K.F., Lee K.
Journal of Medicinal Chemistry,
2009
15.
![Design, synthesis and anti-Parkinsonian evaluation of 3-alkyl/aryl-8-(furan-2-yl)thiazolo[5,4-e][1,2,4]triazolo[1,5-c]pyrimidine-2(3H)-thiones against neuroleptic-induced catalepsy and oxidative stress in mice](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Azam F., El-gnidi B.A., Alkskas I.A., Ahmed M.A.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2010
16.

Diana P., Carbone A., Barraja P., Kelter G., Fiebig H., Cirrincione G.
Bioorganic and Medicinal Chemistry,
2010
17.
![Synthesis and antitumor activity of 3-[(methyl)bis(5-trialkylsilyl-furan-2-yl)silyl]propylamines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Ignatovich L., Romanov V., Spura J., Popelis J., Domracheva I., Shestakova I.
Chemistry of Heterocyclic Compounds,
2012
18.
10.1016/j.mencom.2018.01.004_bib0090
Banerjee
Int. J. Rev. Life Sci.,
2012
19.
![Synthesis and antitumor activities of novel dibenzo[b,d]furan–imidazole hybrid compounds](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Liu L., Wang X., Yan J., Li Y., Sun C., Chen W., Zhou B., Zhang H., Yang X.
European Journal of Medicinal Chemistry,
2013
20.
![Anticancer properties of new synthetic hybrid molecules combining naphtho[2,3-b]furan-4,9-dione or benzo[f]indole-4,9-dione motif with phosphonate subunit](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gach K., Modranka J., Szymański J., Pomorska D., Krajewska U., Mirowski M., Janecki T., Janecka A.
European Journal of Medicinal Chemistry,
2016
21.
![Anti-cancer effect of N-(3,5-bis(trifluoromethyl)phenyl)-5-chloro-2,3-dihydronaphtho[1,2-b]furan-2-carboxamide, a novel synthetic compound](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kwon S.M., Jung Y.Y., Hwang C.J., Park M.H., Yoon N.Y., Kim T.M., Yu J.M., Kim D.H., Seo D.W., Youn H.S., Seo H.O., Chung I.S., Han S.B., Hwang B.Y., Yoo H., et. al.
Molecular Carcinogenesis,
2015
22.
![Recent Advances in the Synthesis of Biologically Active Compounds Containing Benzo[b]Furans as a Framework](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
M. Heravi M., Zadsirjan V.
Current Organic Synthesis,
2015
23.

Sobenina L.N., Petrova O.V., Tomilin D.N., Gotsko M.D., Ushakov I.A., Klyba L.V., Mikhaleva A.I., Trofimov B.A.
Tetrahedron,
2014
24.
10.1016/j.mencom.2018.01.004_bib0120
Trofimov
2009