Abstract
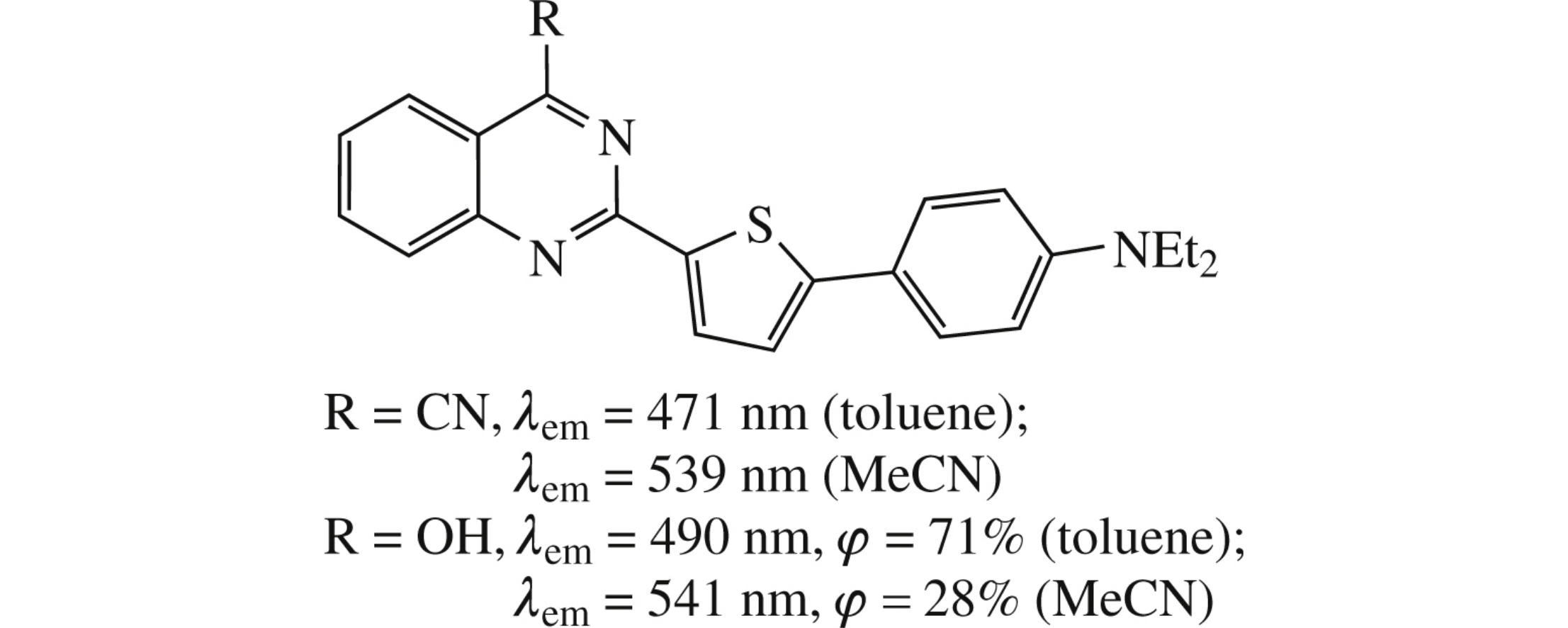
Two novel 2-[5-(4-diethylaminophenyl)thiophen-2-yl]quinazoline derivatives were obtained using Suzuki cross-coupling as the key step. Optical studies have demonstrated that 4-cyanoquinazoline derivative possesses low photolumine-scence, whereas its 3H-quinazolin-4-one counterpart represents promising photoluminescent molecule.
References
1.

Achelle S., Rodrı́guez-López J., Robin-le Guen F.
Journal of Organic Chemistry,
2014
2.

Mphahlele M., Paumo H., El-Nahas A., El-Hendawy M.
Molecules,
2014
3.

Liu D., Zhang Z., Zhang H., Wang Y.
Chemical Communications,
2013
4.

Zhang Z., Xie J., Wang H., Shen B., Zhang J., Hao J., Cao J., Wang Z.
Dyes and Pigments,
2016
5.

Ortiz R.P., Casado J., Hernández V., Navarrete J.L., Letizia J., Ratner M., Facchetti A., Marks T.
Chemistry - A European Journal,
2009
6.

Achelle S., Barsella A., Caro B., Robin-le Guen F.
RSC Advances,
2015
7.

Cvejn D., Achelle S., Pytela O., Malval J., Spangenberg A., Cabon N., Bureš F., Robin-le Guen F.
Dyes and Pigments,
2016
8.
Verbitskiy E.V., Baranova A.A., Lugovik K.I., Khokhlov K.O., Cheprakova E.M., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Arkivoc,
2016
9.
Dmitriev A.V., Yusupov A.R., Irgashev R.A., Kazin N.A., Mal’tsev E.I., Lypenko D.A., Rusinov G.L., Vannikov A.V., Charushin V.N.
Mendeleev Communications,
2016
10.

Liang M., Chen J.
Chemical Society Reviews,
2013
11.

Yan H., Lee P., Armstrong N.R., Graham A., Evmenenko G.A., Dutta P., Marks T.J.
Journal of the American Chemical Society,
2005
12.

Nosova E.V., Moshkina T.N., Lipunova G.N., Kopchuk D.S., Slepukhin P.A., Baklanova I.V., Charushin V.N.
European Journal of Organic Chemistry,
2016
13.
Nosova E.V., Moshkina T.N., Kopchuk D.S., Lipunova G.N., Slepukhin P.A., Charushin V.N.
Mendeleev Communications,
2016
14.
W Hu, G. Zhong, L. Yang and H. Xu, Patent US 0245033 A1, 2013.
15.

OHBA S., KONNO S., YAMANAKA H.
Chemical and Pharmaceutical Bulletin,
2011
16.

Layeva A.A., Nosova E.V., Lipunova G.N., Trashakhova T.V., Charushin V.N.
Russian Chemical Bulletin,
2007
17.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
18.

Abdel-Jalil R.J., Voelter W., Saeed M.
Tetrahedron Letters,
2004
19.

Ooyama Y., Oda Y., Mizumo T., Ohshita J.
Tetrahedron,
2013
20.
10.1016/j.mencom.2018.01.002_bib0060
Borisevich
Dokl. Akad. Nauk SSSR,
1954
21.

Hrast M., Rožman K., Jukič M., Patin D., Gobec S., Sova M.
Bioorganic and Medicinal Chemistry Letters,
2017