Abstract
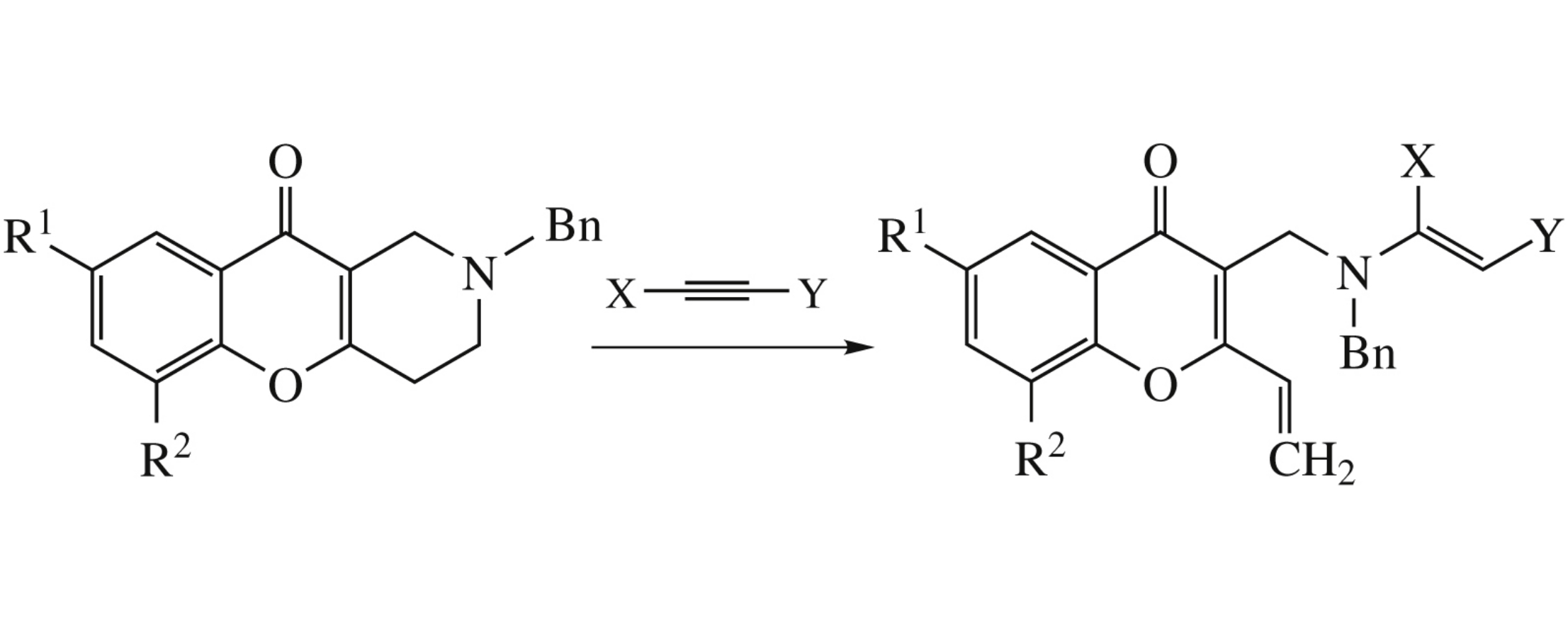
2-Vinyl substituted chromones were obtained by the reaction between 1,2,3,4-tetrahydrochromeno[3,2-c]pyridines and electron-deficient alkynes.
References
1.

Middleton E., Kandaswami C., Theoharides T.C.
Pharmacological Reviews,
2000
2.

Galietta L.J., Springsteel M.F., Eda M., Niedzinski E.J., By K., Haddadin M.J., Kurth M.J., Nantz M.H., Verkman A.S.
Journal of Biological Chemistry,
2001
3.

Kahnberg P., Lager E., Rosenberg C., Schougaard J., Camet L., Sterner O., Nielsen E.Ø., Nielsen M., Liljefors T.
Journal of Medicinal Chemistry,
2002
4.

Veitch N.C., Grayer R.J.
Natural Product Reports,
2008
5.

Fujita Y., Yonehara M., Tetsuhashi M., Noguchi-Yachide T., Hashimoto Y., Ishikawa M.
Bioorganic and Medicinal Chemistry,
2010
6.

Eleya N., Malik I., Reimann S., Wittler K., Hein M., Patonay T., Villinger A., Ludwig R., Langer P.
European Journal of Organic Chemistry,
2012
7.

Kaur K., Jain M., Kaur T., Jain R.
Bioorganic and Medicinal Chemistry,
2009
8.

Larget R., Lockhart B., Renard P., Largeron M.
Bioorganic and Medicinal Chemistry Letters,
2000
9.

Gaspar A., Matos M.J., Garrido J., Uriarte E., Borges F.
Chemical Reviews,
2014
10.

Ryabukhin D.S., Vasilyev A.V.
Russian Chemical Reviews,
2016
11.
Kornev M.Y., Moshkin V.S., Eltsov O.S., Sosnovskikh V.Y.
Mendeleev Communications,
2016
12.

Kaldas R.S., Hughes C.L.
Reproductive Toxicology,
1989
13.

Ranatunga S., Tang C.A., Kang C.W., Kriss C.L., Kloppenburg B.J., Hu C.A., Del Valle J.R.
Journal of Medicinal Chemistry,
2014
14.
![The reaction of tetrahydrochromeno[3,4-c]pyridines with activated alkynes. The first synthesis of tetrahydrochromeno[4,3-d]azocines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Voskressensky L.G., Kulikova L.N., Gozun S.V., Khrustalev V.N., Borisova T.N., Listratova A.V., Ovcharov M.V., Varlamov A.V.
Tetrahedron Letters,
2011
15.

Sliwa H., Cordonnier C.
Journal of Heterocyclic Chemistry,
1977
16.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
17.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
18.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
19.

Spek A.L.
Journal of Applied Crystallography,
2003
20.

Shaw A.Y., Chang C., Liau H., Lu P., Chen H., Yang C., Li H.
European Journal of Medicinal Chemistry,
2009
21.

Lee K.Y., Nam D.H., Moon C.S., Seo S.H., Lee J.Y., Lee Y.S.
European Journal of Medicinal Chemistry,
2006