Abstract
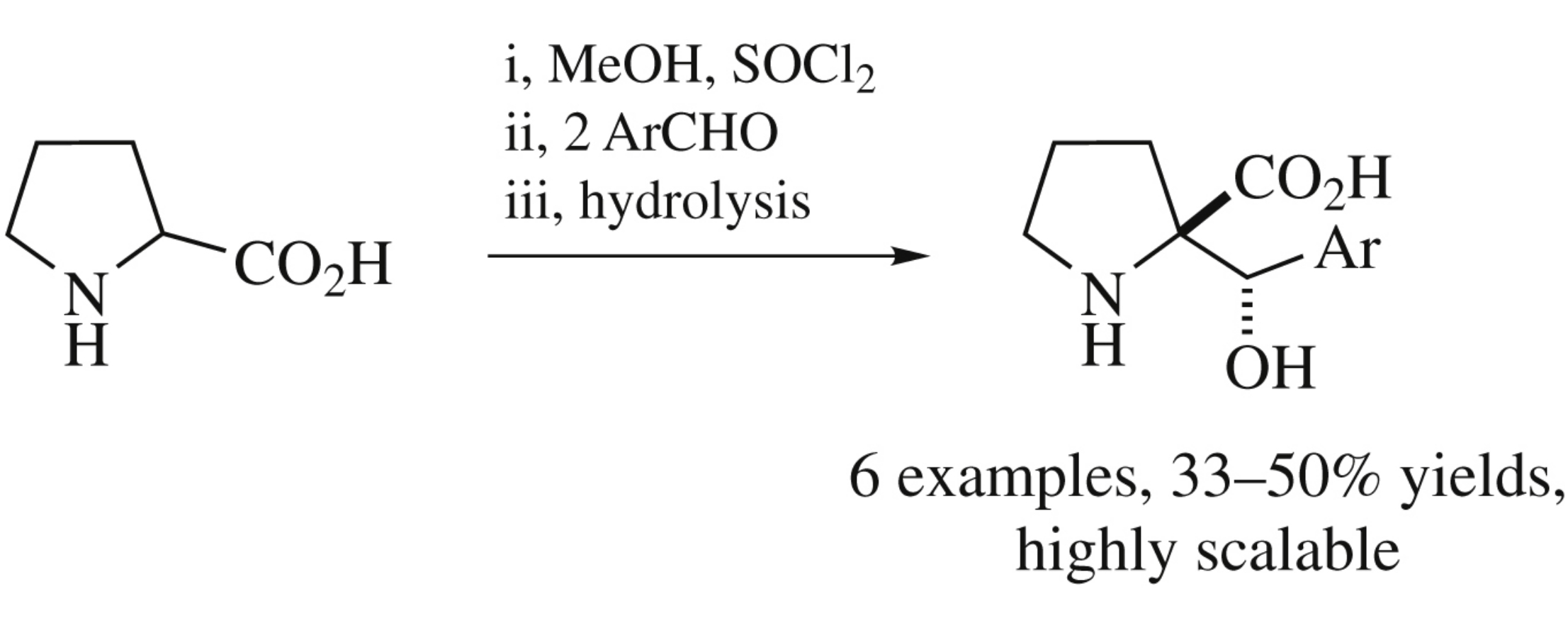
2-Oxapyrrolizidines formed in reactions of methyl prolinate with two molecules of aromatic aldehyde upon gradual hydrolysis give α-(α-hydroxybenzyl)prolines. The products are preferentially formed as one diastereomer (91–100%) in overall yield 33–50% (five stages starting from proline).
References
1.

Tsuge O., Kanemasa S.
Advances in Heterocyclic Chemistry,
1989
2.
10.1016/j.mencom.2017.11.031_sbref0005b
Harwood
2002
3.

Najera C., Sansano J.
Current Organic Chemistry,
2003
4.

Coldham I., Hufton R.
Chemical Reviews,
2005
5.
![Construction of Enantiopure Pyrrolidine Ring System via Asymmetric [3+2]-Cycloaddition of Azomethine Ylides](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Pandey G., Banerjee P., Gadre S.R.
Chemical Reviews,
2006
6.
![Intramolecular [3 + 2]-Cycloadditions of Azomethine Ylides Derived from Secondary Amines via Redox-Neutral C–H Functionalization](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Mantelingu K., Lin Y., Seidel D.
Organic Letters,
2014
7.
8.

Singh M.S., Chowdhury S., Koley S.
Tetrahedron,
2016
9.

Rizzi G.P.
Journal of Organic Chemistry,
1970
10.
![Use of N-[(trimethylsilyl)methyl]amino ethers as capped azomethine ylide equivalents](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Padwa A., Dent W.
Journal of Organic Chemistry,
1987
11.
10.1016/j.mencom.2017.11.031_sbref0010c
Nyerges
Synthesis,
2001
12.

Nair V., Mathai S., Augustine A., Viji S., Radhakrishnan K.V.
Synthesis,
2004
13.

Ryan J.H., Spiccia N., Wong L.S., Holmes A.B.
Australian Journal of Chemistry,
2007
14.

Spiccia N., Basutto J., Jokisz P., Wong L.S., Meyer A.G., Holmes A.B., White J.M., Ryan J.H.
Organic Letters,
2011
15.

Lee S., Diab S., Queval P., Sebban M., Chataigner I., Piettre S.R.
Chemistry - A European Journal,
2013
16.

Gayen B., Banerji A.
Monatshefte fur Chemie,
2014
17.

Santos H., Distiller A., D'Souza A.M., Arnoux Q., White J.M., Meyer A.G., Ryan J.H.
Organic Chemistry Frontiers,
2015
18.

19.
Moshkin V.S., Sosnovskikh V.Y.
Mendeleev Communications,
2015
20.

Orsini F., Pelizzoni F., Forte M., Destro R., Gariboldi P.
Tetrahedron,
1988
21.

Felluga F., Pitacco G., Visintin C., Valentin E.
Helvetica Chimica Acta,
1997
22.

Harwood L.M., Macro J., Watkin D., Williams C.E., Wong L.F.
Tetrahedron Asymmetry,
1992
23.

Alker D., Hamblett G., Harwood L.M., Robertson S.M., Watkin D.J., Eleri Williams C.
Tetrahedron,
1998
24.
10.1016/j.mencom.2017.11.031_sbref0030c
Aldous
Synthesis,
2005
25.

Brome V.A., Harwood L.M., Osborn H.M.
Canadian Journal of Chemistry,
2006
26.

Seashore-Ludlow B., Torssell S., Somfai P.
European Journal of Organic Chemistry,
2010
27.

Danielsson J., Toom L., Somfai P.
European Journal of Organic Chemistry,
2010
28.

Sansano J., Nájera C., Castelló L.
Synthesis,
2014
29.

Moshkin V.S., Sosnovskikh V.Y.
Tetrahedron Letters,
2013
30.

Moshkin V.S., Sosnovskikh V.Y.
Tetrahedron Letters,
2013
31.

Moshkin V.S., Sosnovskikh V.Y.
Tetrahedron Letters,
2013
32.

Moshkin V.S., Sosnovskikh V.Y.
Tetrahedron Letters,
2014
33.
![3-Cyanochromones in [3+2] cycloadditions with an azomethine ylide derived from sarcosine and formaldehyde. A short synthesis of 1-benzopyrano[2,3-c:3,4-c′]dipyrrolidines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sosnovskikh V.Y., Kornev M.Y., Moshkin V.S.
Tetrahedron Letters,
2014
34.
![Substituted chromones in [3+2] cycloadditions with nonstabilized azomethine ylides: synthesis of 1-benzopyrano[2,3-c]pyrrolidines and 1-benzopyrano[2,3-c:3,4-c′]dipyrrolidines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sosnovskikh V.Y., Kornev M.Y., Moshkin V.S., Buev E.M.
Tetrahedron,
2014
35.

Moshkin V.S., Buev E.M., Sosnovskikh V.Y.
Tetrahedron Letters,
2015
36.

Buev E.M., Moshkin V.S., Sosnovskikh V.Y.
Tetrahedron Letters,
2015
37.

Buev E.M., Moshkin V.S., Sosnovskikh V.Y.
Organic Letters,
2016
38.

Horwitz D., Pettinger W.A., Orvis H., Thomas R.E., Sjoerdsma A.
Clinical Pharmacology and Therapeutics,
1967
39.

Mathias C.J.
Clinical Autonomic Research,
2008